Clear Sky Science · en
Dysfunction of the episodic memory network in the Alzheimer’s disease cascade
Why this memory study matters
Alzheimer’s disease is feared largely because it steals our personal memories. This study asks a simple but critical question: does the brain’s memory network start to fail only after brain tissue is lost, or does it malfunction earlier, while the tissue is still largely intact? By tracking more than a thousand brain scans over time, the researchers show that changes in how the brain switches its memory network on and off may be an early and partly reversible step in the Alzheimer’s chain of events.
Following the long road of Alzheimer’s
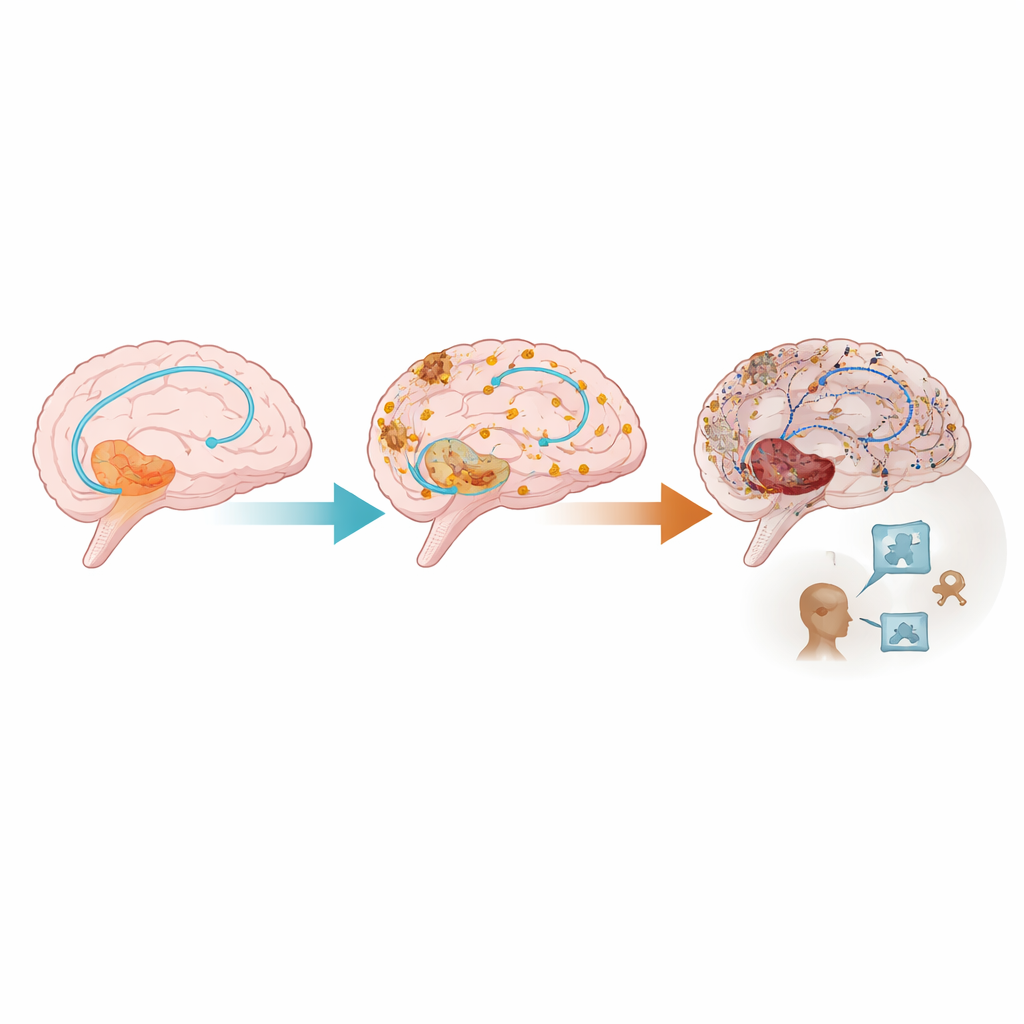
Alzheimer’s disease unfolds slowly, often over two decades. First, sticky amyloid protein builds up, then twisted strands of tau protein spread, brain tissue shrinks, and only later do thinking problems become obvious. The team used data from the large German DELCODE study, which follows older adults ranging from cognitively normal to those with mild impairment or early dementia. They combined spinal fluid measures of amyloid and tau, MRI measures of brain shrinkage in key memory regions, and standard memory tests into a computer model that placed each person on a single “disease timeline,” indicating how far along the Alzheimer’s process they were, even if they did not yet have dementia symptoms.
Watching the memory network in action
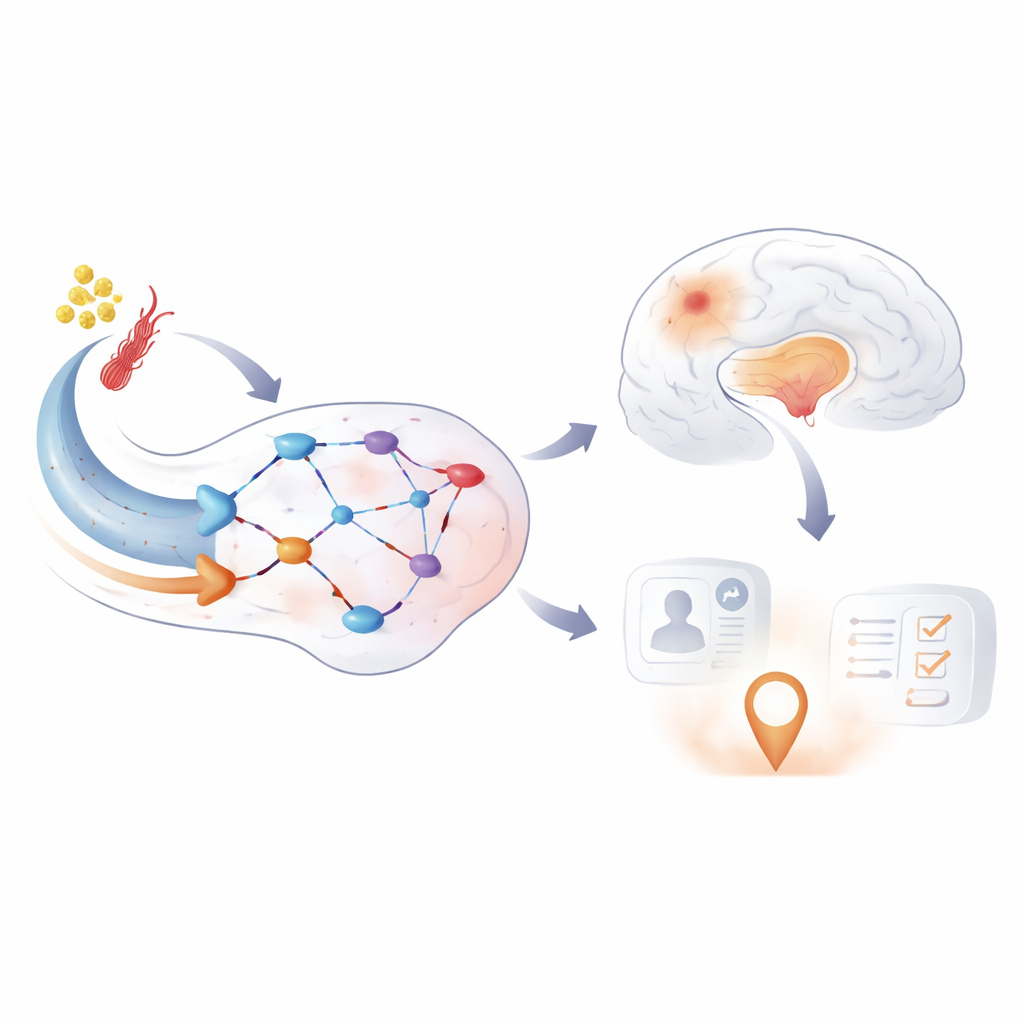
The researchers focused on episodic memory, our ability to remember everyday events, which depends strongly on the hippocampus and a set of regions known to turn down their activity during demanding tasks. Participants lay in an MRI scanner while viewing indoor and outdoor scenes and later took a surprise memory test. By comparing brain activity for scenes that were later remembered versus forgotten, the team measured how strongly memory-related regions “turned on,” and how strongly the usual resting regions “turned down,” during successful memory formation. Across nearly 500 people and multiple years of follow-up, they could then ask how these activation and deactivation patterns changed as individuals moved along the Alzheimer’s timeline.
When the brain fails to power down
A clear pattern emerged: as people progressed along the Alzheimer’s disease timeline, the difference between brain states for remembered and forgotten items shrank. Regions that should quiet down during effortful memory work—especially parts of the posterior cingulate cortex and precuneus—stopped doing so effectively. At the same time, some regions that should become more active during memory encoding showed weaker responses. Importantly, the loss of this “powering down” behavior followed a non‑linear course: it began to change relatively early, then became most abnormal later in the disease and appeared before clear declines on standard cognitive tests. This suggests that the memory network’s switching behavior becomes disrupted before thinking problems are obvious in daily life.
Beyond simple brain shrinkage
The next question was what drives these abnormal activity patterns. By teasing apart the contributions of amyloid, tau, brain volume, small-vessel damage, and communication between regions, the study found that both tau buildup and loss of hippocampal volume were linked to changes in the memory network. Crucially, some of the abnormal activity remained even after accounting for tissue loss, pointing to problems at the level of synapses—the communication points between nerve cells—rather than just dead or shrunken tissue. Activity in the memory network also predicted how well people performed on memory tests, even after controlling for traditional markers of Alzheimer’s damage.
What this means for treatment and prevention
To a layperson, the key message is that Alzheimer’s may harm the brain’s memory circuits in at least two partly independent ways: by killing or shrinking nerve cells, and by disrupting how surviving cells fire together. The study proposes an extended view of the disease cascade in which episodic memory network dysfunction sits between protein buildup and obvious cognitive decline. Because this network disruption appears before full-blown dementia and is not completely explained by irreversible brain loss, it may offer a window of opportunity. Treatments that reduce amyloid and tau, or that directly support synapses and network function through drugs, lifestyle changes, or brain stimulation, might still improve or stabilize memory even after some structural damage has occurred.
Citation: Lattmann, R., Vockert, N., Bernal, J. et al. Dysfunction of the episodic memory network in the Alzheimer’s disease cascade. Nat Commun 17, 3578 (2026). https://doi.org/10.1038/s41467-026-71831-z
Keywords: Alzheimer’s disease, episodic memory, brain networks, functional MRI, synaptic dysfunction