Clear Sky Science · en
Amorphous intermediates and discovery of a kinetic polymorph of BiVO4 from heating V+Bi+Zn single-source precursors
Turning Designer Molecules into Useful Solids
Many of the materials that power clean energy technologies—such as solar fuel generators and batteries—are complex metal oxides. This study shows how chemists can start from carefully built molecules that already contain all the needed elements, heat them up, and watch them transform into advanced materials. Along the way, the team discovers hidden intermediate states and even a brand‑new crystal form of a key solar material, opening fresh routes to tune performance for energy applications.
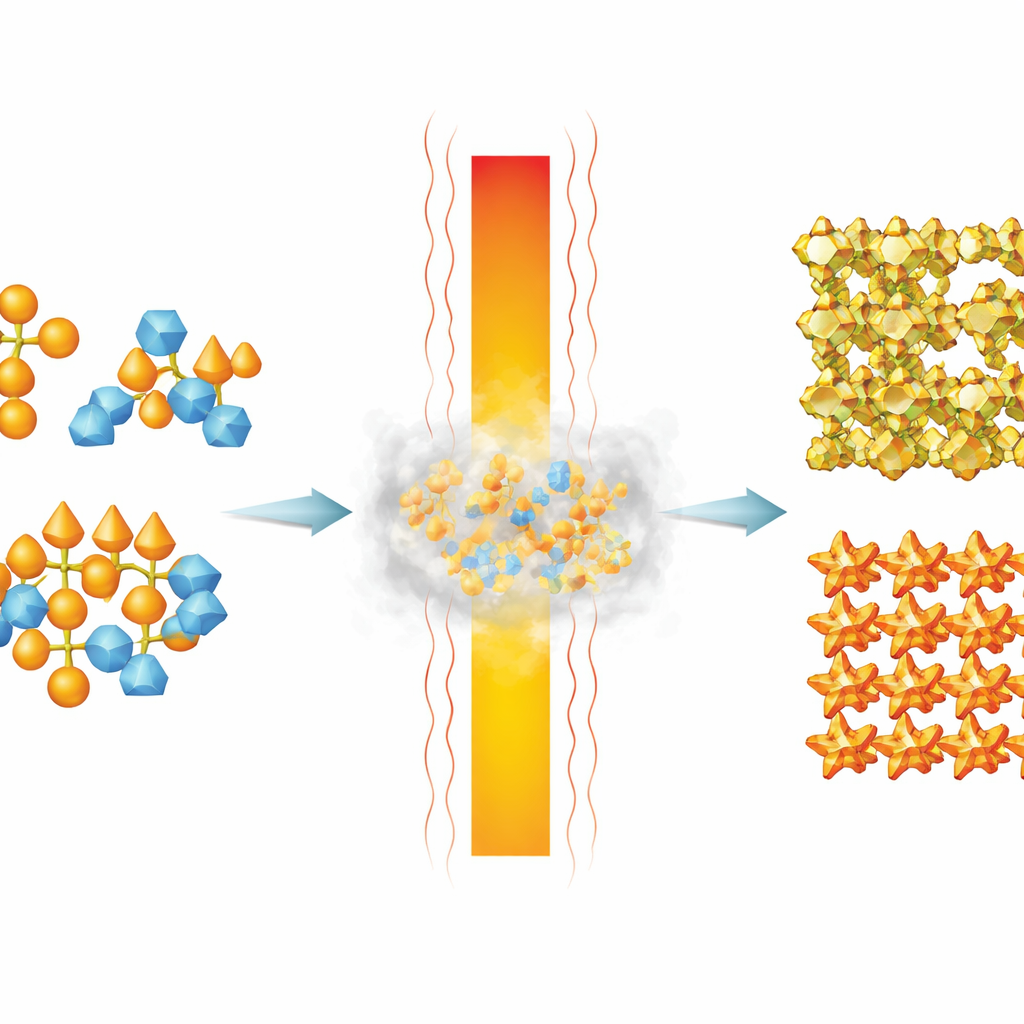
Building Blocks Already Mixed at the Atomic Scale
The researchers begin with so‑called single‑source precursors: molecular clusters that already contain vanadium, bismuth and sometimes zinc in precise ratios. Because all the metals are pre‑mixed in one molecule, heating these clusters can yield very uniform metal‑oxide solids without the high temperatures and long times usually needed to blend separate ingredients. The team studies three related precursors—one with only vanadium, one with vanadium plus bismuth, and one with vanadium, bismuth and zinc—to see exactly how they fall apart and rebuild as solid oxides.
Hidden Amorphous States on the Way to Crystals
Using a combination of advanced tools—solid‑state nuclear magnetic resonance, pair‑distribution function analysis and in‑situ X‑ray diffraction—the authors track structural changes as the precursors are heated. Instead of jumping straight from molecules to well‑ordered crystals, all three systems pass through poorly ordered, “amorphous” stages. For the vanadium‑only precursor, heating produces a black, mixed‑valence vanadium oxide whose local structure resembles known oxides V4O9 and (NH4)V4O10, even though it lacks long‑range order. Further heating finally yields bright orange V2O5 with growing crystal domains. These observations show that apparently featureless black powders can hide distinct local arrangements and oxidation states that matter for function.
A New Crystal Form of a Key Solar Material
The bismuth‑containing precursors ultimately form BiVO4, a leading material for photoelectrochemical water splitting, along with V2O5. But in a narrow temperature window around 350–420 °C, the team observes an extra, transient BiVO4 phase that does not match any known structure. Detailed synchrotron and total‑scattering analysis reveal that this “kinetic” phase adopts a cubic framework similar to a known tin tungstate and to fast oxide‑ion conductors. The authors name it β‑BiVO4. In this structure, vanadium sits in compact tetrahedra while bismuth occupies highly distorted six‑oxygen cages, with the bismuth atoms slightly disordered. Quantum‑mechanical calculations show that β‑BiVO4 has a larger electronic band gap than the usual monoclinic form, arising from more widely spaced vanadium units and altered bismuth‑oxygen bonding. Although less stable than the standard phase, β‑BiVO4 can be frozen in by carefully controlling the heating of the molecular precursors.
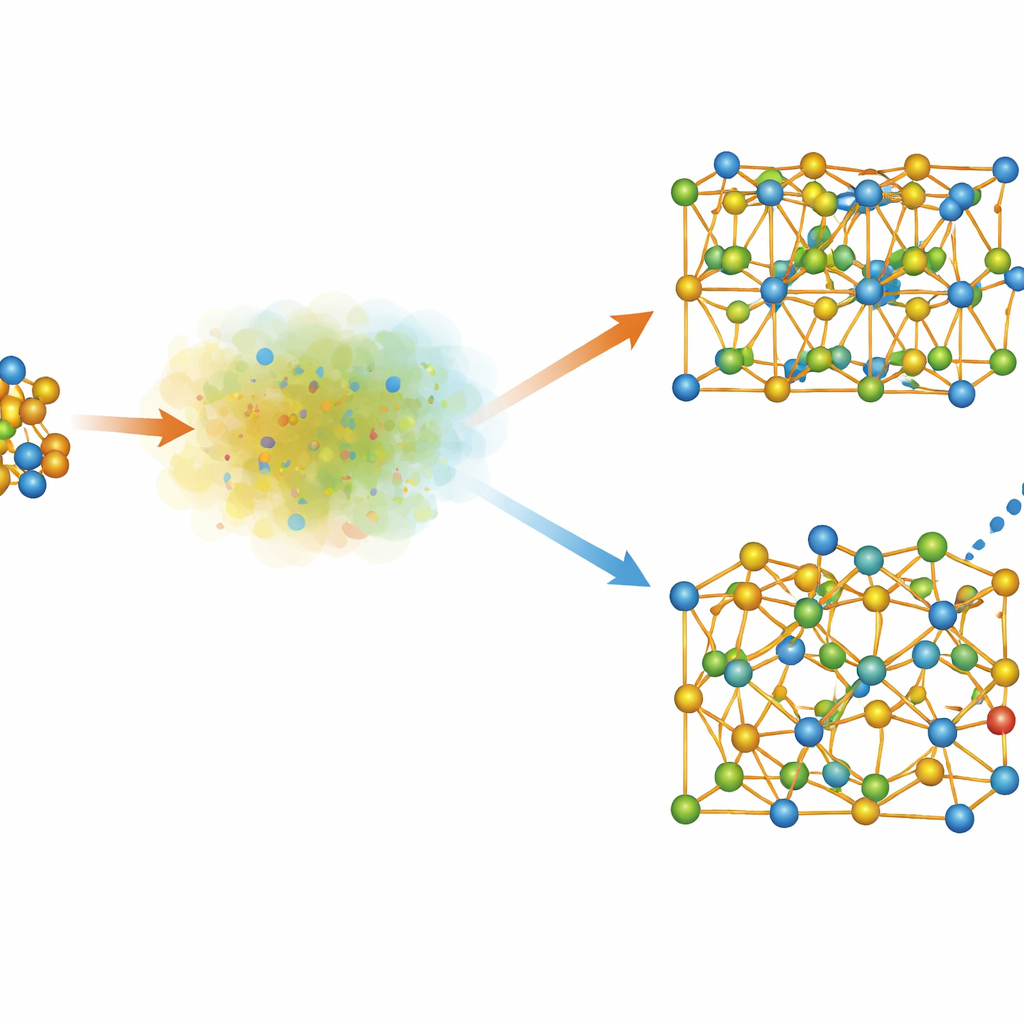
Tuning Composition, Doping and Battery Behavior
Starting from a trimetallic precursor containing zinc, the researchers track how zinc atoms enter the BiVO4 lattice as temperature rises. Subtle shifts in lattice dimensions and loss of separate zinc‑oxide signatures indicate that zinc substitutes into bismuth sites or occupies nearby positions, introducing disorder that broadens the local environment of vanadium. Such zinc‑doped BiVO4 is already known to boost performance as a photoanode by improving conductivity and surface reactions, so linking its formation directly to the decomposition pathway of a single‑source precursor gives a powerful handle for design. Meanwhile, the black amorphous vanadium oxide intermediate from the vanadium‑only route shows promising behavior as a lithium‑ion battery cathode: during cycling, it gradually restructures, allowing it to host an increasing number of lithium ions and reach capacities comparable to engineered mixed‑valence oxides.
Why These Transformations Matter
This work demonstrates that the journey from molecule to solid is rich in structure and opportunity. By monitoring each stage of thermal decomposition, the authors uncover new amorphous states, show how oxidation levels and crystal sizes can be tuned with temperature, and reveal a previously unknown polymorph, β‑BiVO4, with distinct electronic properties. For a general reader, the key message is that starting from carefully designed molecular precursors—and not just from simple oxide powders—can unlock hidden forms of familiar materials and offer new ways to tailor them for solar fuel devices and batteries.
Citation: Hands, A.E., Barnes, T.J., Scarperi, A. et al. Amorphous intermediates and discovery of a kinetic polymorph of BiVO4 from heating V+Bi+Zn single-source precursors. Nat Commun 17, 3739 (2026). https://doi.org/10.1038/s41467-026-71702-7
Keywords: single-source precursors, bismuth vanadate, polymorph discovery, amorphous metal oxides, lithium-ion batteries