Clear Sky Science · en
A high-resolution spatial map of cilia-associated proteins in the human fallopian tube
Why the tiny hairs in the fallopian tube matter
The fallopian tubes play a quiet but essential role in human reproduction: they are the passageway where eggs meet sperm and where the earliest days of pregnancy unfold. Lining these tubes are countless microscopic, hair-like structures called cilia that help move eggs, sperm, and early embryos. When these cilia malfunction, the result can be infertility or even cancer, yet scientists have had only a rough picture of which proteins make them work. This study builds a detailed map of those proteins in the human fallopian tube, offering new clues to reproductive health and disease.
Building a parts list for the fallopian tube
To understand what makes the fallopian tube unique, the researchers first examined large datasets of gene activity from many human organs. They identified 310 genes that are more active in the fallopian tube than in most other tissues. Many of these genes have been linked to moving cilia or the tails of sperm, both of which must beat in a coordinated way to propel cells or fluids. By singling out this group, the team created a starting “parts list” of molecules likely to be important for how the tube moves eggs and fluid.
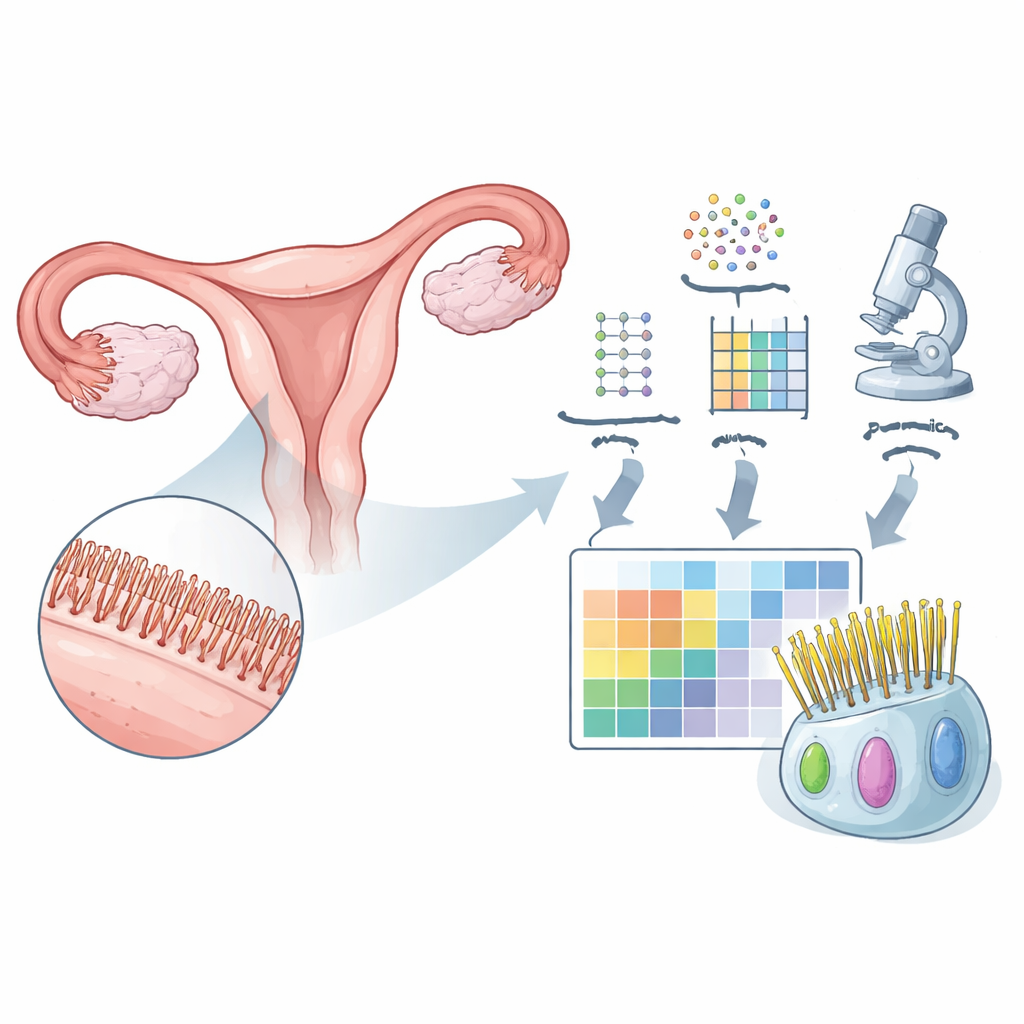
Turning genes into a spatial protein map
Next, the team asked where the corresponding proteins actually appear in human tissue. Using antibody-based imaging, they examined protein patterns in thin slices of fallopian tube and in other ciliated tissues, such as the airways and parts of the brain. Out of the 310 genes, they could reliably visualize 133 proteins. Remarkably, 123 of these were found only in the ciliated cells of the tube’s inner lining, often in specific subregions: at the tips of cilia, along their shafts, near their roots, or within the cell body. This fine-grained map shows not just which proteins are present, but exactly where they sit within the tiny moving structures.
Comparing cilia across the body
The fallopian tube does not exist in isolation, so the researchers compared its cilia proteins with those in other tissues that also rely on moving cilia or sperm tails. Many of the mapped proteins appeared in the cilia of the airways and in the flagella of sperm, suggesting a shared “core toolkit” for movement across different organs and even species. Yet some proteins were absent from sperm tails or from certain brain structures, hinting at tissue-specific add-ons that may tune cilia for particular jobs, such as moving mucus in the lungs or guiding eggs in the reproductive tract. The team also matched their list to databases of known cilia-related diseases and found that many proteins had never been linked to such disorders before, expanding the pool of candidates that might underlie unexplained infertility or respiratory problems.
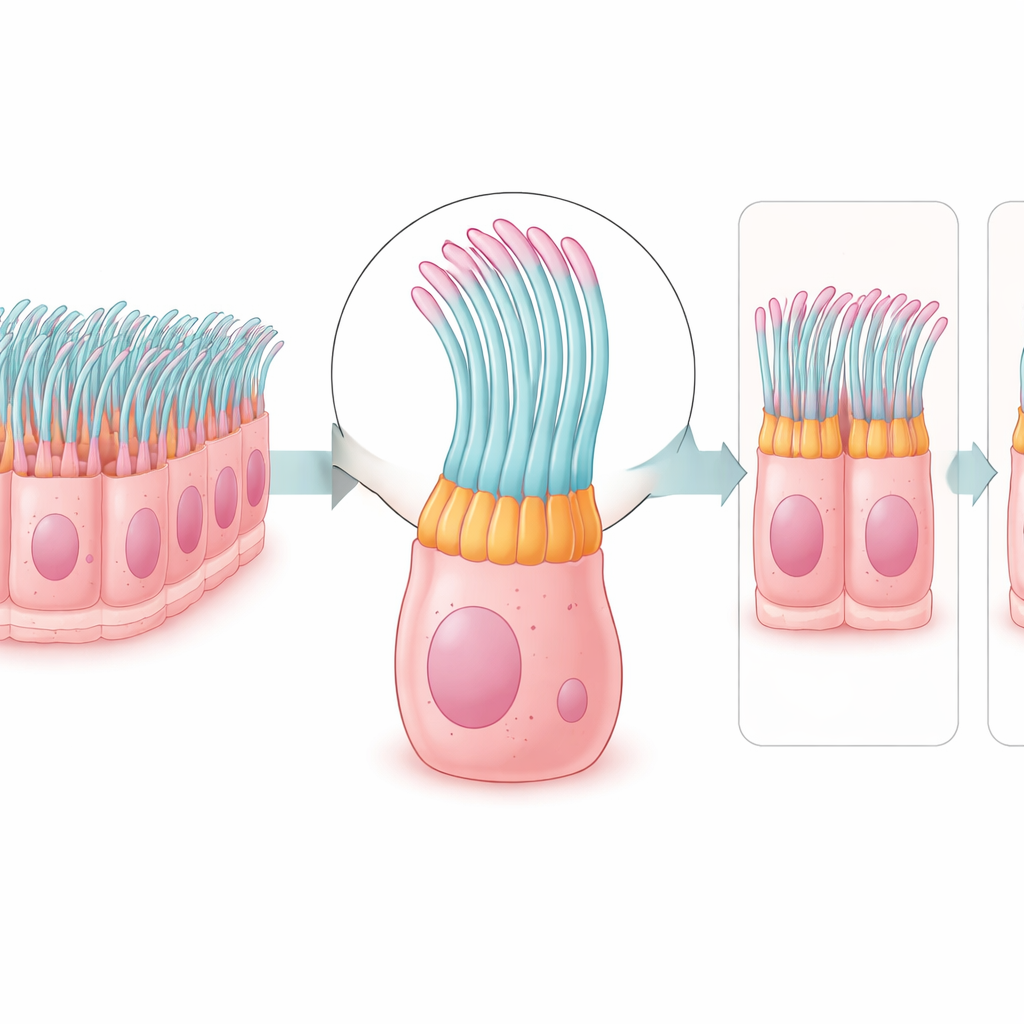
From healthy tubes to damaged ones
To see how this protein map changes in disease, the researchers examined tissue from a patient with hydrosalpinx, a condition where the fallopian tube becomes swollen and fluid-filled and often causes infertility. Compared with healthy tubes, the diseased sample showed a thinner epithelial layer and far fewer ciliated cells, confirmed by reduced levels of a key cilia-regulating factor. Three little-studied proteins—FHAD1, RIIAD1, and C2orf81—were especially diminished in the patient’s cilia. Because these proteins normally sit in the moving parts of the cilium, their loss could weaken ciliary beating and worsen the blockage of fluid and cells in hydrosalpinx.
What this means for fertility and disease
In everyday terms, this study transforms our view of the fallopian tube from a simple “pipe” into a highly specialized conveyor belt built from hundreds of precisely placed proteins. By mapping where these proteins reside within cilia and how they change in a damaged tube, the work offers a molecular blueprint for understanding why some people experience infertility or cilia-related disease without obvious causes. Over time, such maps could help doctors pinpoint which parts of the ciliary machinery are failing in a given patient, guide genetic testing, and eventually suggest targeted treatments to restore the tiny, coordinated movements that are so crucial for human reproduction.
Citation: Hikmet, F., Digre, A., Hansen, J.N. et al. A high-resolution spatial map of cilia-associated proteins in the human fallopian tube. Nat Commun 17, 3616 (2026). https://doi.org/10.1038/s41467-026-71692-6
Keywords: fallopian tube cilia, female infertility, spatial proteomics, motile ciliopathies, reproductive biology