Clear Sky Science · en
Friction-differentiated separation of alkali ions through two-dimensional nanochannels based on niobate perovskite
Why Separating Simple Salts Suddenly Matters
The batteries that power electric cars and store renewable energy depend heavily on lithium, a metal that is usually trapped in salty water alongside very similar neighbors like sodium and potassium. Today’s filters struggle to tease these nearly identical ions apart, wasting energy and resources. This paper reports a new kind of ultra-thin membrane that separates these ions not by their size or charge, but by how “slippery” they are as they slide through angstrom-scale channels—offering a fresh route to cleaner, more efficient lithium production from brines.
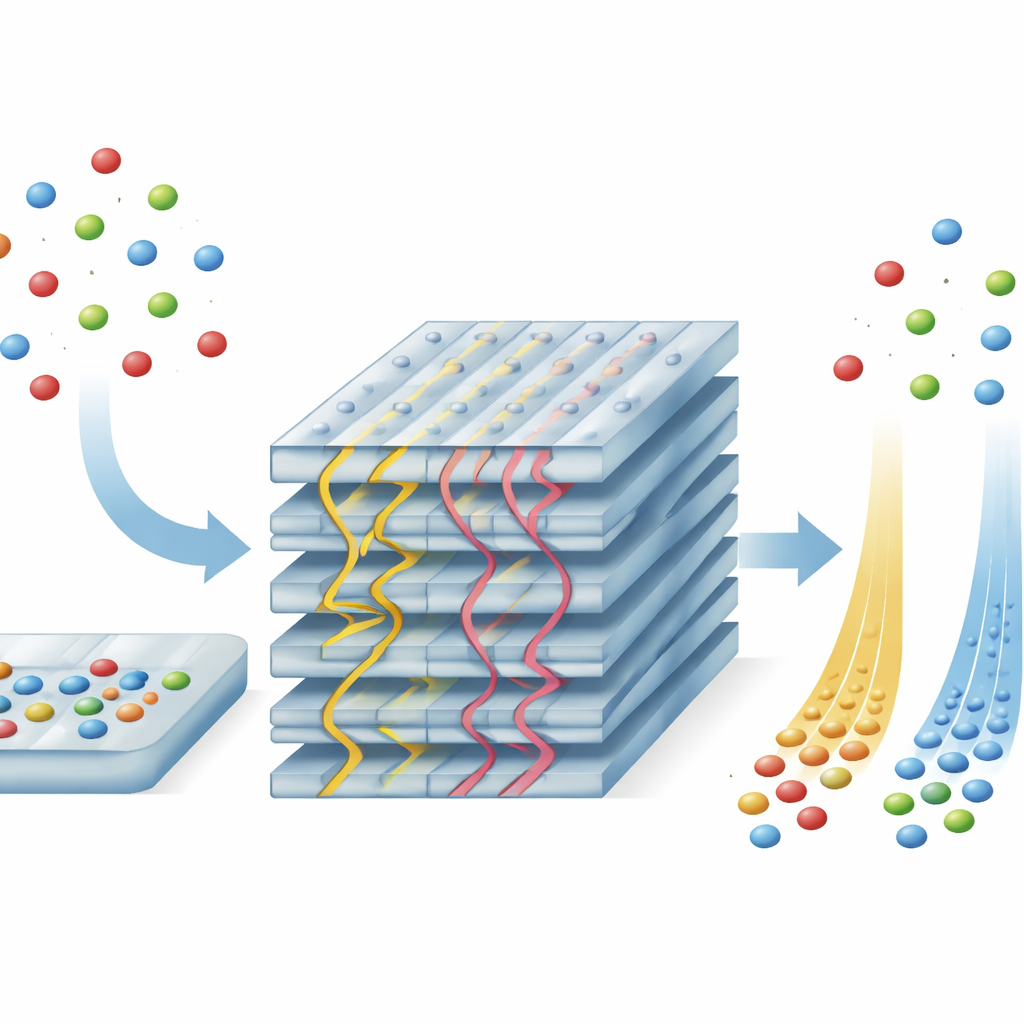
From Static Sieves to Moving-World Filters
Most ion-separation technologies work like carefully sized sieves or charged nets: they exploit how big, how strongly charged, or how water-loving each ion is. That strategy fails for alkali ions such as lithium, sodium, and potassium, which share the same charge and have almost indistinguishable hydrated sizes—differences smaller than the natural jitter of atoms in a channel wall. As a result, many of today’s artificial nanochannels achieve selectivity ratios under ten when asked to distinguish among these ions. The authors argue that instead of focusing on static properties, we should design membranes around a dynamic one: the friction that ions experience as they move through confined spaces.
Building Ultra-Ordered Sliding Lanes
To turn friction into a sorting tool, the researchers created membranes from a layered material called niobate perovskite. By exfoliating this crystal into ultra-thin, highly uniform nanosheets and stacking them carefully, they formed membranes whose internal channels are just 6–9 angstroms wide—only a few atoms across—and aligned over long distances. Within this lattice, two distinct channel shapes naturally emerge: a herringbone pattern filled with thin layers of water, and a zigzag pattern that is essentially dry. Because the atomic arrangement and chemical groups along these channels are exquisitely regular, they provide a controlled landscape in which tiny differences in how each ion interacts with the walls translate into different levels of sliding resistance.
Letting Friction Do the Sorting
Experiments showed that, in the pristine niobate membrane, lithium and potassium ions zip through far faster than sodium—by factors of about 30 to 50 in binary mixtures—despite all three carrying the same charge. Computer simulations and nanotribology measurements pinpoint the reason: in the water-filled herringbone channels, potassium and lithium experience substantially lower friction than sodium at the nanonewton scale. The periodic structure of the channel amplifies these friction differences beyond what classical diffusion laws predict, turning small changes in drag into large differences in transport rate. When the team disturbed the channel order or added polymers that disrupted the pathways, the extraordinary selectivity largely vanished, underscoring that controlled friction, not simple size or adsorption, governs the effect.
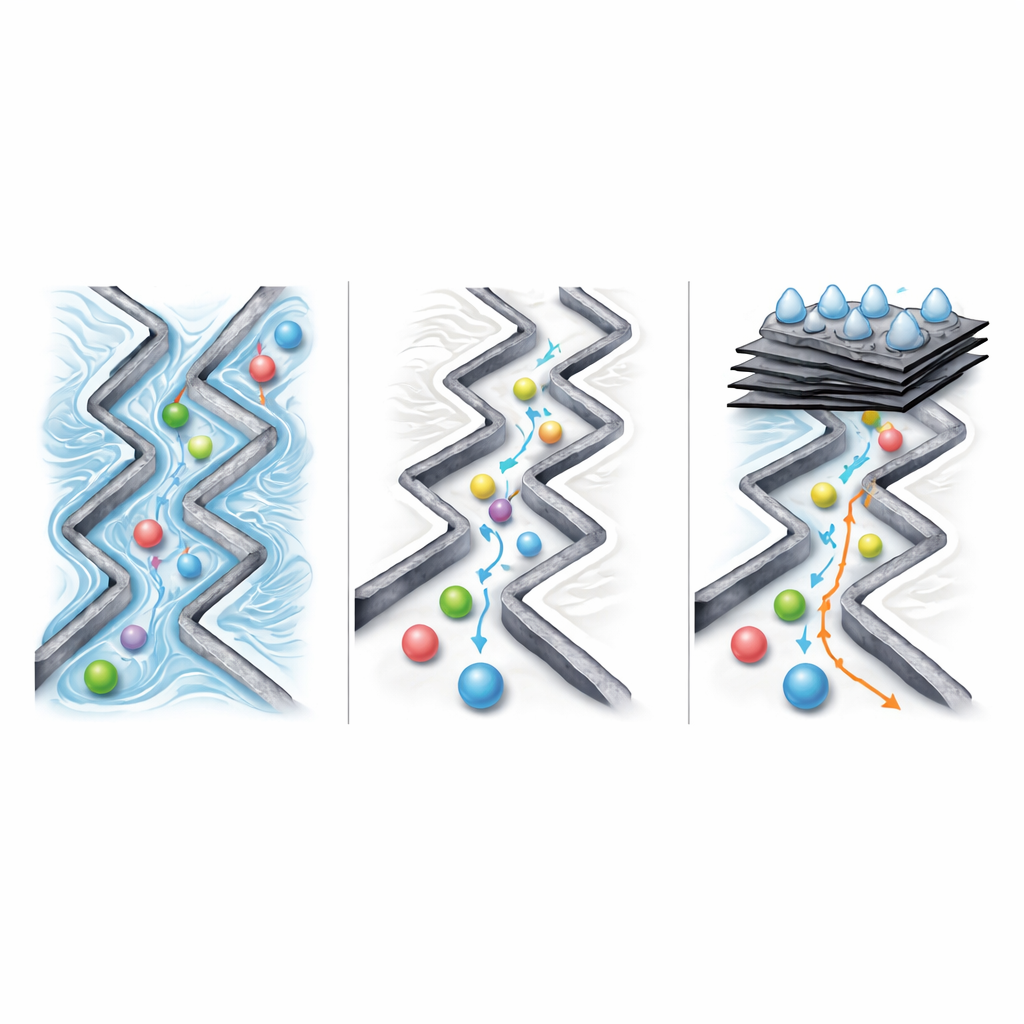
Switching Tracks to Target Lithium Alone
Because the membrane also contains zigzag channels with different interaction patterns, the authors asked whether they could re-route ions to favor lithium over potassium. By inserting thin graphene oxide layers that effectively block the low-friction herringbone paths, they forced ions into the more resistive zigzag channels. There, the friction landscape flips: lithium now faces significantly lower drag than sodium or potassium. Membranes engineered with the right ratio of niobate to graphene oxide achieved lithium-over-potassium separation factors around 28, while still allowing relatively fast ion flow. Structural measurements confirmed that in these composite membranes the herringbone channels are largely sealed, and the zigzag channels expand preferentially when lithium passes through.
Toward Cleaner Lithium from Real-World Brines
To test practical relevance, the team applied their membranes to a simulated brine modeled on Chile’s Salar de Atacama, a major natural source of lithium that also contains large amounts of sodium, potassium, magnesium, and calcium. In a staged process, a commercial nanofiltration step first removed most multivalent ions, and then cascades of niobate and niobate–graphene oxide membranes successively stripped out sodium and potassium. Across these stages, the lithium fraction in the solution rose from under 4% to more than 95%. By surpassing the usual trade-off between throughput and selectivity, and by basing separation on tunable friction rather than nearly indistinguishable sizes, this work outlines a new design principle for membranes that could help secure lithium and other critical ions for a sustainable energy future.
Citation: Ai, X., Zhu, L., Cui, F. et al. Friction-differentiated separation of alkali ions through two-dimensional nanochannels based on niobate perovskite. Nat Commun 17, 3415 (2026). https://doi.org/10.1038/s41467-026-71579-6
Keywords: lithium extraction, ion-selective membranes, nanofluidics, perovskite nanochannels, tribology-based separation