Clear Sky Science · en
Unveiling the synergistic mechanism of C-F and C-Cl bonds in enhancing the triboelectric performance of fluorinated polymers
Turning Everyday Motion into Power
Imagine charging small electronics simply by walking, breathing, or letting the wind blow on a thin sheet of plastic. Triboelectric nanogenerators—devices that turn mechanical motion into electricity—promise exactly that. But to move from clever lab demos to practical power sources, these generators need materials that can grab and hold far more electric charge. This article describes how chemists found a smart way to redesign a well-known plastic so it can capture record levels of charge, and how that breakthrough leads all the way to a breathable, AI-enabled smart insole that recognizes who you are and how you move.
Why Better Plastics Matter for Tiny Power Plants
Triboelectric nanogenerators work a bit like supercharged versions of rubbing a balloon on your hair: when two materials touch and separate, electrons move between them, leaving one side negatively charged and the other positive. The more charge the material can trap on its surface, the more electricity the device can deliver. For over a decade, engineers have relied on fluorinated plastics—materials rich in fluorine atoms—because fluorine strongly attracts electrons. Classic choices like PTFE and PVDF have set the standard for “tribo-negative” materials, but progress has stalled: no new plastic had clearly outperformed these workhorses. The authors set out to understand, at the molecular level, how to push beyond this limit.
Discovering the Winning Chemical Team
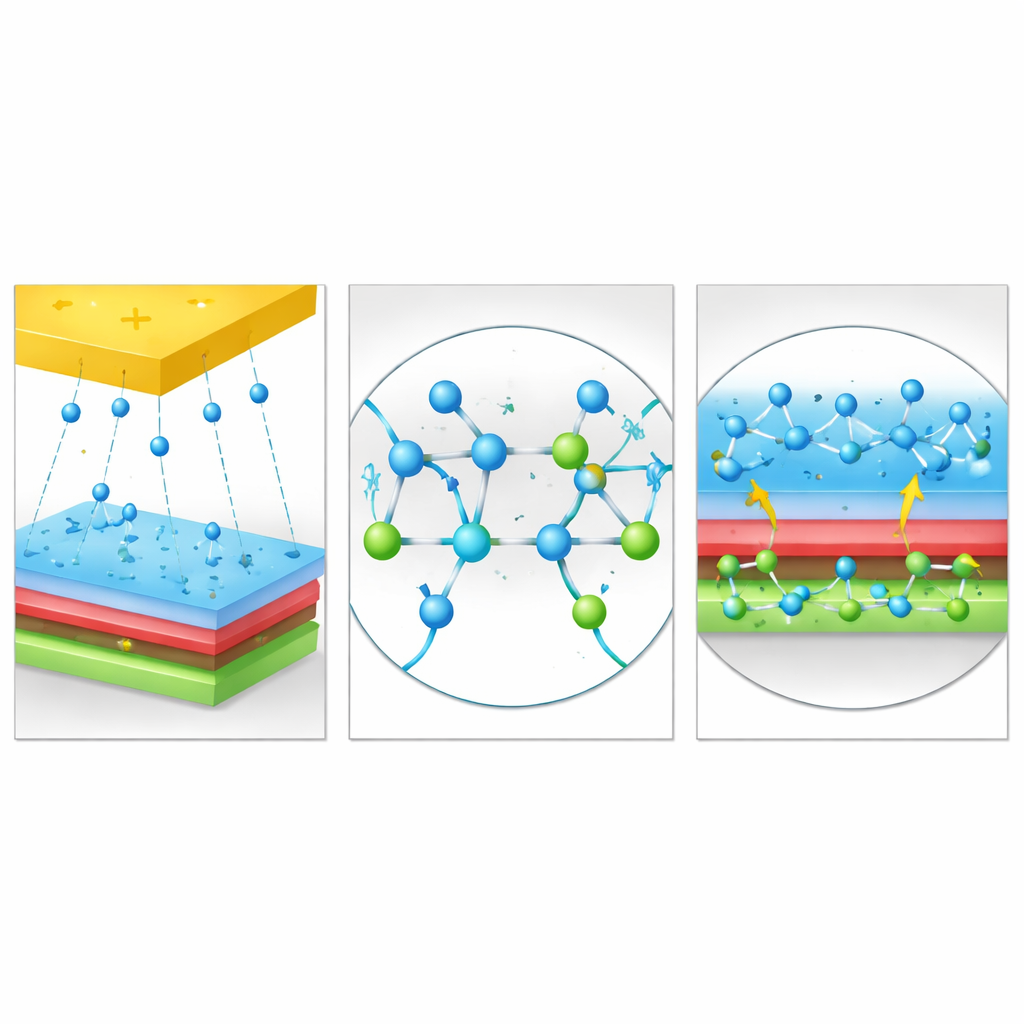
The team focused on a family of plastics based on PVDF, each built from slightly different chemical building blocks. Using quantum-level calculations, they compared how these variants accept extra electrons. They found that when PVDF is combined with a unit called CTFE—containing both fluorine (F) and chlorine (Cl) atoms—the resulting polymer, named PC, has a much lower energy barrier for accepting electrons than the others. In simple terms, the presence of chlorine, working alongside fluorine, creates special spots in the material that are especially eager to grab electrons. Atomic-scale analysis showed that bonds between carbon and chlorine are key contributors to these low-energy “electron landing sites.” Experiments on thin films and nanofibers confirmed the prediction: among all the tested PVDF-based plastics, PC was the most strongly negative, pulling in the most charge when rubbed against a metal.
Finding the Sweet Spot Between Grabbing and Keeping Charge
However, a great electron catcher is not useful if it leaks charge quickly. The researchers realized that two things must be balanced: how easily the surface gains electrons and how well it holds onto them over time. To tune this balance, they treated PC with a chlorine-containing plasma, which introduces extra chlorine atoms onto the surface and changes the ratio of chlorine to fluorine. Measurements showed a clear trade-off. As chlorine content increased, the material became better at picking up charge—output initially rose—but its ability to hold that charge fell, because chlorine attracts electrons less strongly than fluorine. At very high chlorine levels, charges escaped too quickly, and performance dropped. An intermediate ratio, where chlorine and fluorine coexist in the right proportion, delivered the best of both worlds: strong electron capture and good charge retention, leading to a record triboelectric charge density for PVDF-based plastics.
From Lab Plastic to Breathing Smart Insoles
To showcase what this tuned material can do, the team spun it into ultrafine fibers, forming a porous membrane that is both highly active electrically and comfortable to wear. They built a flexible triboelectric generator using this chlorine-enhanced PC fiber layer together with nylon fibers. When pressed and released—by a fingertip, a footstep, or a tap—the device produced very high voltages and currents, with power densities sufficient to run small electronics directly or charge capacitors for later use. The fiber mat’s open structure allowed air and moisture to pass through, making it suitable for long-term contact with skin. Integrating the generator into a shoe insole created a self-powered sensor that turns each step into a stream of electrical signals reflecting the wearer’s gait.
Smart Steps and What Comes Next
Finally, the authors fed these electrical footprints into machine-learning and deep-learning models. The AI systems learned to distinguish different people and their activities—walking, running, jumping—with near-perfect accuracy, all from the insole’s passive power supply. For a layperson, the key message is that a subtle tweak in chemical bonding—combining carbon–fluorine and carbon–chlorine links in the right ratio—can dramatically boost how much charge a plastic surface can collect and keep. This not only sets a new performance record for a widely used polymer family but also points to a general design rule for next-generation energy-harvesting and sensing materials that can power and interpret our movements at the same time.
Citation: Liu, J., Zhang, F., Xu, J. et al. Unveiling the synergistic mechanism of C-F and C-Cl bonds in enhancing the triboelectric performance of fluorinated polymers. Nat Commun 17, 3698 (2026). https://doi.org/10.1038/s41467-026-71546-1
Keywords: triboelectric nanogenerator, fluorinated polymers, energy harvesting, wearable sensors, smart insole