Clear Sky Science · en
Aging-associated decline of phosphatidylcholine synthesis is a malleable trigger of natural mitochondrial aging
Why the cell’s power plants matter as we grow old
As we age, our bodies slowly lose the ability to bounce back from stress, illness, and changes in diet. A key part of this decline happens in mitochondria, the tiny “power plants” inside our cells. This study asks a simple but important question: what naturally goes wrong with these power plants over time, and can we nudge the process back in our favor with something as accessible as diet?
A closer look at aging power plants
Mitochondria help convert nutrients into usable energy and support many other cellular jobs. When they falter, muscles tire more easily, metabolism slows, and age-related diseases become more likely. While inherited defects in mitochondria are known to cause severe disorders and premature aging, the gradual, everyday decline of mitochondria in otherwise normal aging has been harder to pin down. The researchers turned to the tiny roundworm Caenorhabditis elegans, a standard model for aging, and compared ordinary worms with special long-lived strains whose mitochondria are chronically impaired yet somehow support a longer life.
Finding a hidden weak link in cell membranes
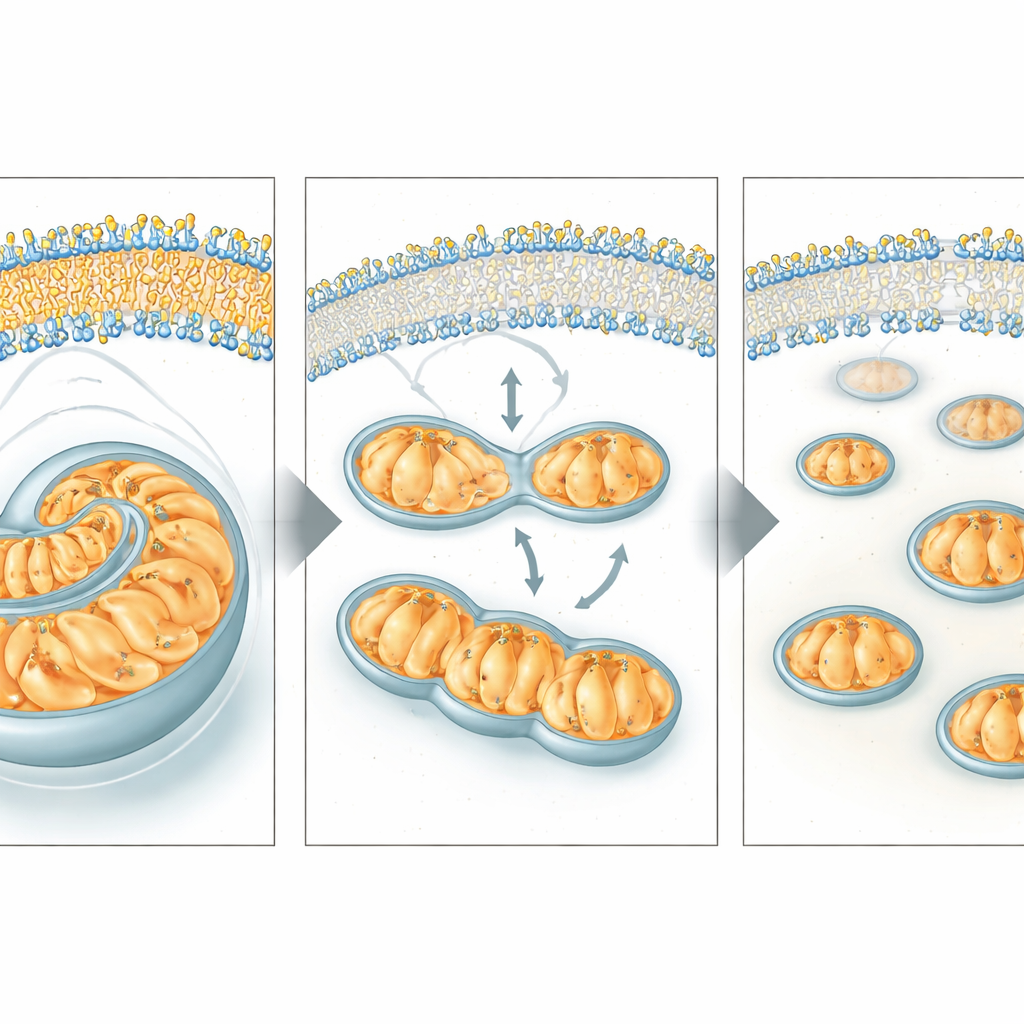
Using large-scale surveys of proteins, fats, and other molecules across the worms’ lifespans, the team found that broad metabolic changes, especially those tied to fats and mitochondria, appear relatively late in life. Among the strongest late-life changes were sharp drops in three related proteins responsible for making phosphatidylcholine, a major building block of cell and mitochondrial membranes. When the scientists deliberately reduced these proteins in young worms, mitochondria rapidly shifted from long, interconnected networks to small, fragmented units and their ability to consume oxygen—a measure of energy production—fell noticeably. This pattern resembles what is normally seen only in old animals.
Rebuilding membranes to restore function
The researchers then asked whether topping up the missing membrane ingredient could rescue these defects. They provided worms with extra phosphatidylcholine directly, or with choline, a simple nutrient the body can convert into phosphatidylcholine. In worms where the synthesis pathway was blocked, these supplements smoothed out the mitochondrial network, improved energy production, and even corrected stunted body size. Importantly, giving choline to naturally aging worms partially reversed the age-related fragmentation of mitochondria and boosted their respiratory capacity, suggesting that declining phosphatidylcholine production is not just a quirk of mutant strains but a broad contributor to normal mitochondrial aging.
Evidence from human tissues and blood
To see whether a similar story holds in people, the authors mined large public datasets of human tissues and blood samples. They found that a key human enzyme for phosphatidylcholine synthesis tends to be expressed less in older individuals, especially in fat-rich tissues and the ovary. In parallel, measurements from tens of thousands of adults in the UK Biobank showed that circulating phosphatidylcholine levels, and related flexible fatty acids, decline with age. These decreases were most pronounced in postmenopausal women, a group known to experience increased mitochondrial vulnerability. Lower phosphatidylcholine and polyunsaturated fat levels were associated with higher blood lactate, greater obesity and diabetes, slower walking speed, and poorer memory—patterns consistent with stressed, less efficient mitochondria.
From cell cultures to potential interventions
The team also tested whether boosting this lipid could protect human cells under stress. In cultured skin cells treated with high doses of the diabetes drug metformin—which can strain mitochondria—adding choline helped preserve mitochondrial membrane potential, a measure of power-plant health. Combining choline with another mitochondrial fuel provided even better protection. While these experiments do not yet translate directly into medical advice, they suggest that restoring phosphatidylcholine levels can bolster mitochondrial resilience across very different biological systems.
What this means for healthy aging
Overall, the study identifies a gradual drop in phosphatidylcholine synthesis as a natural, reversible driver of mitochondrial aging. As membranes lose this key component, mitochondria become more rigid and fragmented, undermining their ability to produce energy and adapt to metabolic challenges. By replenishing phosphatidylcholine—through nutrients like choline or carefully formulated supplements—the researchers were able to restore healthier mitochondrial networks in worms and improve stress resistance in human cells. For lay readers, the message is that part of our age-related energy decline may stem from an underappreciated weakness in cell membranes, one that might eventually be addressed through targeted dietary or pharmacological strategies.
Citation: Poliezhaieva, T., Li, Y., Chaudhari, P.S. et al. Aging-associated decline of phosphatidylcholine synthesis is a malleable trigger of natural mitochondrial aging. Nat Commun 17, 3589 (2026). https://doi.org/10.1038/s41467-026-71508-7
Keywords: mitochondrial aging, phosphatidylcholine, choline supplementation, cell membrane lipids, metabolic health