Clear Sky Science · en
The role of hydration in the removal of glyphosate (GLY) and aminomethylphosphonic acid (AMPA) by nanofiltration membranes
Why caring about hidden herbicides in water matters
Herbicides such as glyphosate, the world’s most widely used weed killer, and its breakdown product AMPA regularly end up in rivers, lakes, and even drinking water. Although present at tiny concentrations, both chemicals are linked to a range of health concerns. Many treatment plants rely on special membranes to strain them out, yet these small, highly water-loving molecules can slip through more easily than expected. This study explores a subtle but crucial player in their removal: the thin shell of water that clings to each molecule.

How modern filters try to stop tiny pollutants
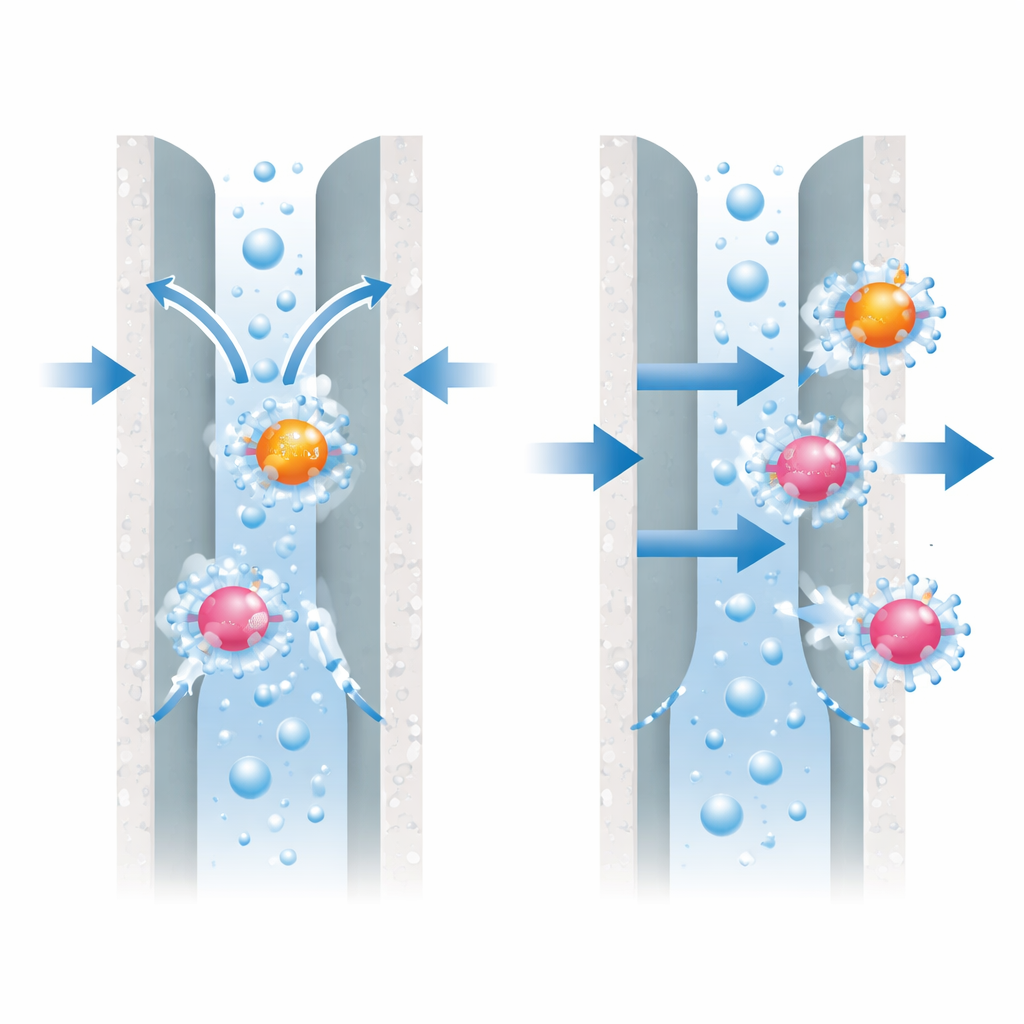
The researchers focus on nanofiltration membranes, thin barriers with pores so small they can separate individual molecules. These membranes can block pollutants in three main ways. First, by size: if a molecule is larger than a pore, it simply cannot pass. Second, by charge: if both the pollutant and the membrane surface carry similar electrical charges, they repel each other. Third, by hydration: molecules in water are wrapped in layers of water molecules, and forcing them to shed some of that shell costs energy, which can discourage them from entering narrow pores. The team set out to untangle how much this hydration effect actually helps stop glyphosate and AMPA.
Testing different membranes under real-world conditions
The authors filtered water spiked with realistic levels of glyphosate and AMPA through six commercial membranes that ranged from very tight to relatively open. As expected, the tightest membranes, with the smallest pore sizes, removed around 85–90 percent of both chemicals, mostly by simple size blocking, with a small contribution from charge and hydration. The more open membranes, however, had pores larger than the bare size of these pollutants and almost no tendency to adsorb them onto their surface. Yet they still managed to remove a significant share, especially when the pollutants carried an electrical charge. This indicated that charge-based repulsion and hydration, not just pore size, were doing much of the work.
The quiet power of water shells and pH
To see how water shells change, the team altered the acidity (pH) of the water. At very low pH, glyphosate and AMPA are mostly neutral, so charge-based repulsion is weak. Even then, some removal (around 50–80 percent for glyphosate and less for AMPA) persisted with open membranes, suggesting that the hydrated size of the molecules was effectively larger than the pores. As pH increased and the molecules became more negatively charged, both removal and the thickness and structure of their hydration shells increased. Using infrared spectroscopy, the researchers could detect subtle changes in the way water molecules vibrate around the pollutants, a sign of tighter hydrogen bonding. Computer simulations backed this up, revealing dense clusters of water molecules around the charged groups on glyphosate and AMPA, especially around the phosphate end of each molecule.
When pressure helps pollutants slip through
The study also tested what happens when the driving pressure across the membrane is turned up. For the tighter membranes, raising the pressure had little effect on removal: the molecules stayed mostly blocked. For a looser membrane, however, higher pressure dramatically reduced removal, dropping glyphosate rejection from about 86 percent to under 30 percent and AMPA rejection to below 10 percent. The authors interpret this as the hydration shell being partly stripped away under strong pressure; once the protective water layer is shredded, the now “leaner” molecules can squeeze through the pores more easily, weakening the hydration-based barrier.
What this means for safer drinking water
Together, the experiments and simulations show that the thin shell of water around glyphosate and AMPA is not just a curiosity of chemistry but a practical control knob for water treatment. For small, highly charged pollutants, removal by nanofiltration depends not only on pore size and charge repulsion but also on how strongly the surrounding water clings to them, and how easily that shell can be peeled off under pressure. Understanding and tuning this hydration effect can help engineers design membranes and operating conditions that keep more of these herbicide residues out of drinking water, without always needing the highest pressures and energy use.
Citation: Trinh, P.B., Nguyen, M.N., Futera, Z. et al. The role of hydration in the removal of glyphosate (GLY) and aminomethylphosphonic acid (AMPA) by nanofiltration membranes. Nat Commun 17, 3741 (2026). https://doi.org/10.1038/s41467-026-71492-y
Keywords: glyphosate, nanofiltration, water purification, hydration shell, pesticide removal