Clear Sky Science · en
Functional synaptic connectivity shapes spine stability in the hippocampus
How tiny connections help memories last
The brain stores our experiences in networks of neurons that talk to each other through thousands of tiny contact points called synapses. Each synapse often sits on a little bump along a neuron’s branch, known as a spine. These spines are constantly appearing and disappearing, which raises a puzzle: if the hardware that holds memories keeps changing, how can memories remain stable for weeks, months, or years? This study peeks directly at individual spines in the hippocampus—a key memory center—to see how their activity and physical stability are linked over time.
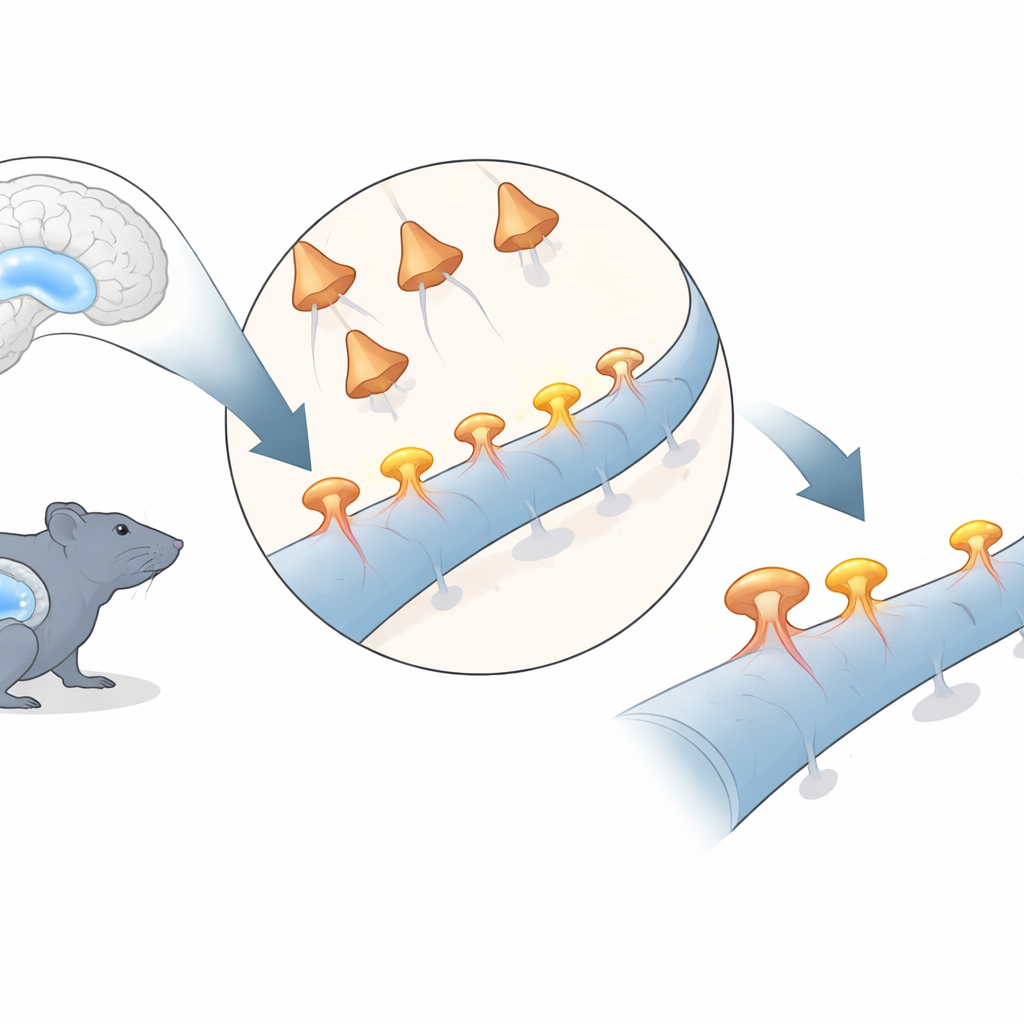
Watching single connections in the memory center
The researchers focused on a well-studied pathway in the hippocampus, where nerve cells in an area called CA3 send signals to cells in an area called CA1. Using genetically encoded light-sensitive proteins, they could trigger activity in selected CA3 cells with flashes of red light. At the same time, they used a fluorescent calcium sensor and high-resolution two-photon microscopy to watch individual spines on CA1 cells in awake, head‑fixed mice for more than two weeks. Each time CA3 cells were stimulated, responsive CA1 spines briefly lit up with a calcium signal, revealing which specific spines were functionally connected to the stimulated inputs.
Active spines are bigger and closer together
The team divided spines into two groups: those that ever showed a light-evoked calcium signal (“responsive” spines) and those that never did (“unresponsive” spines). Spine size is widely used as a readout of synapse strength, because larger spines tend to house more receptors and larger contact zones. The researchers found that responsive spines were, on average, larger than unresponsive ones, indicating that they formed stronger connections. Among responsive spines, those with stronger calcium signals tended to be larger still. In addition, functionally active spines were located closer to one another along the dendritic branch than would be expected by chance, suggesting that inputs from the same or similar CA3 neurons tend to arrive in small local groups rather than being scattered randomly.
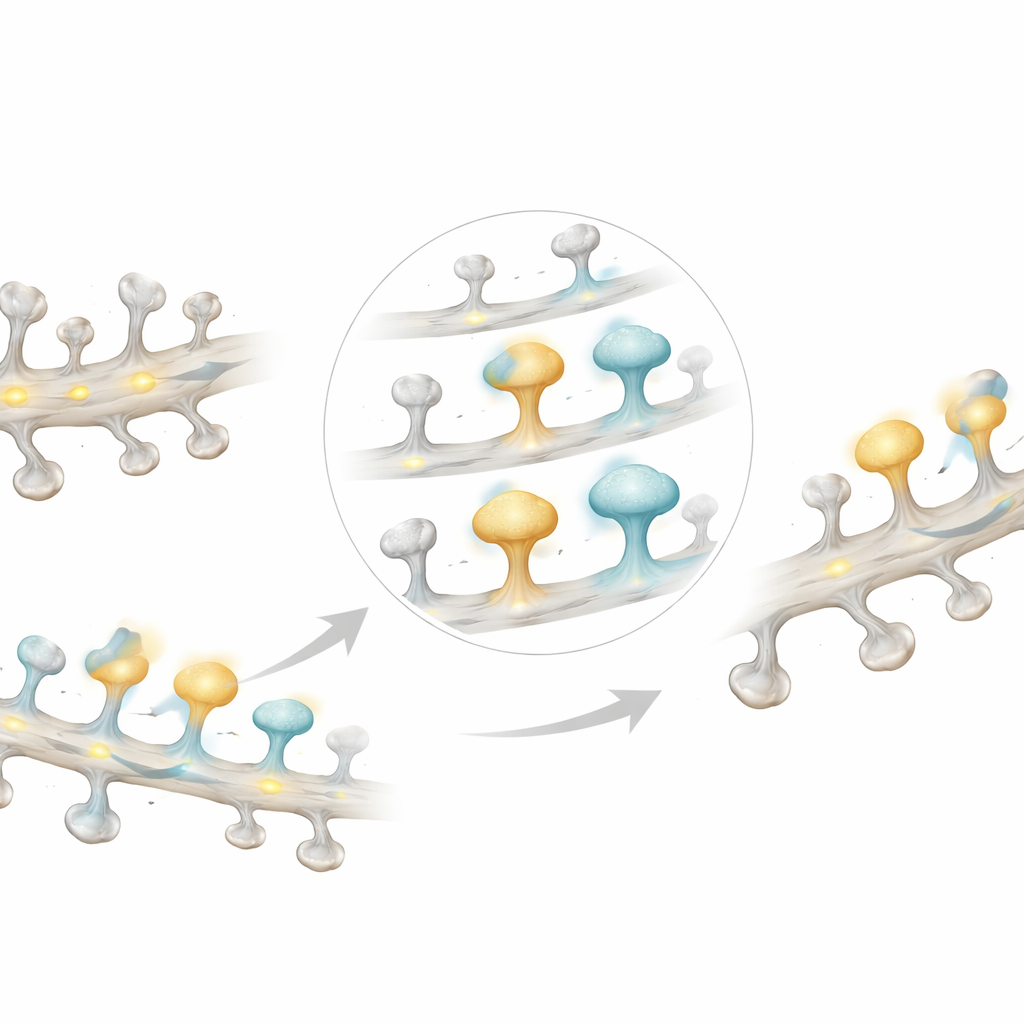
Stable input despite restless single synapses
Although individual spines showed striking variability from session to session—many responded only once across the entire experiment—the overall input to a dendritic branch stayed remarkably stable. The total number of responding spines on a branch, and the average calcium signal measured along the whole dendrite, changed little over the two weeks. This implies that while particular synapses may strengthen, weaken, appear, or disappear, the branch as a whole maintains a fairly constant level of input. In other words, the “wiring diagram” at the level of single spines is fluid, but the broader pattern of connectivity seen by the neuron remains steady.
Strong spines live longer
To quantify how long spines lasted, the authors classified them as persistent (present throughout), transient (appearing and disappearing), newly formed, or eliminated. Responsive spines were much more likely to be persistent and less likely to be transient than unresponsive spines. Even among non‑persistent spines, those that were functionally active tended to survive longer. Looking independently at all spines, regardless of measured activity, showed the same pattern: larger spines had longer lifetimes and were more likely to sit near other long‑lived spines. When the researchers ranked spines by the maximum calcium signal they ever displayed, those with the biggest signals also showed the longest average lifetimes, reinforcing the tight link between functional strength, physical size, and durability.
What this means for memory
These findings suggest that the brain resolves the tension between flexibility and stability by tying the lifespan of a spine to how important its connection is. Strong, active synapses become structurally larger, cluster together, and are maintained for longer periods, forming a stable scaffold of connections that can support lasting memories. Weaker or rarely used spines, by contrast, are more likely to be small, scattered, and short‑lived, allowing neural circuits to be reconfigured as new experiences come in. In this way, the hippocampus can keep a robust backbone of key connections while still constantly remodeling the finer details of its wiring.
Citation: Rais, C., Wiegert, J.S. Functional synaptic connectivity shapes spine stability in the hippocampus. Nat Commun 17, 3218 (2026). https://doi.org/10.1038/s41467-026-71332-z
Keywords: dendritic spines, synaptic plasticity, hippocampus, memory stability, two-photon imaging