Clear Sky Science · en
Partially π-exposed 3D carbohelicene for mechanical tuning of conductance and thermopower in single-molecule junctions
Why a Tiny Molecular Spring Matters
Imagine an electronic device so small that its key component is a single molecule acting like a tiny metal spring. This study shows how such a spring-shaped carbon molecule can be gently squashed or relaxed to tune how well it carries both electric current and heat. The work turns an abstract theoretical idea into a real, measurable effect, pointing toward future gadgets where signals and waste heat are controlled one molecule at a time.
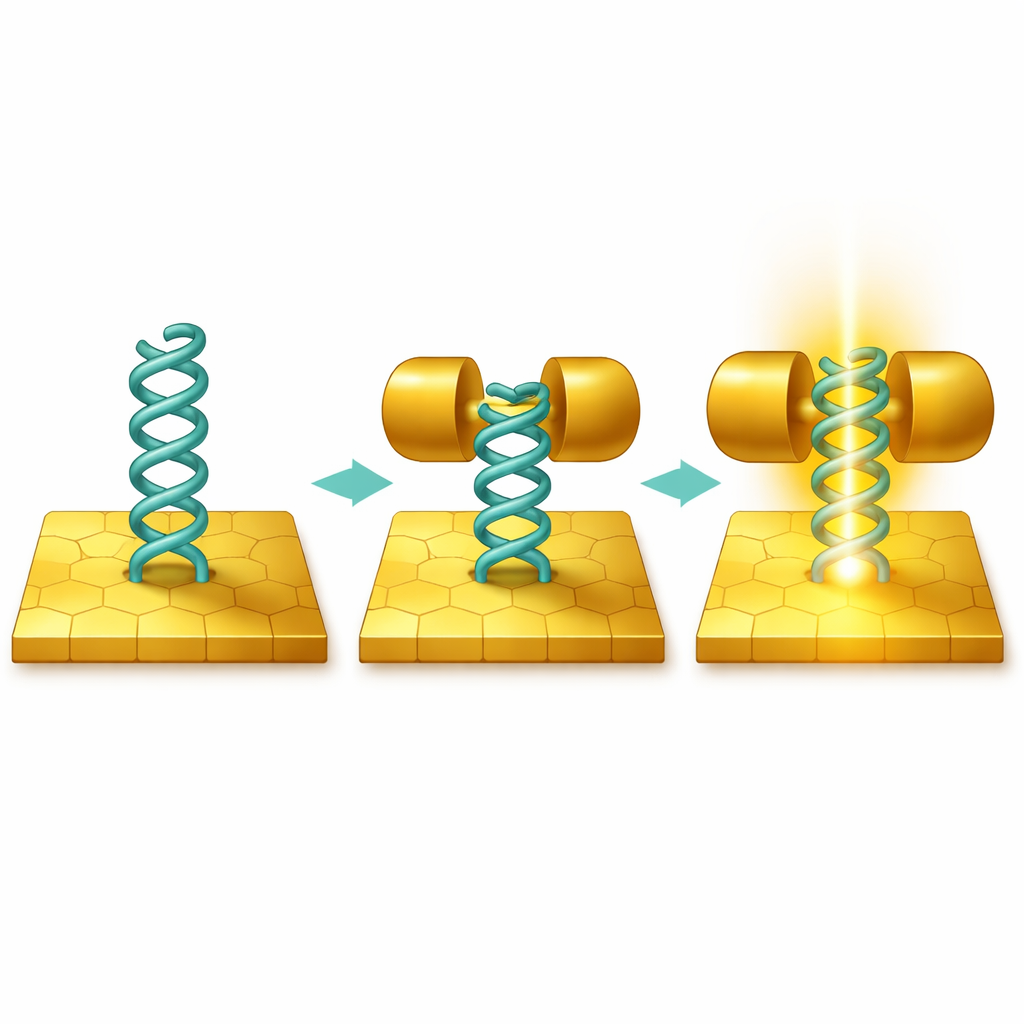
A Corkscrew-Shaped Building Block
The central player is a "helicene" – a corkscrew-like stack of benzene rings that twists into a three-dimensional spiral. Earlier research suggested that this springy shape should allow electrons to flow not only along chemical bonds but also through space between the coils, like shortcuts through the spiral. Theory predicted that stretching or compressing such a molecule between metal contacts could dramatically change both its conductance (how easily current flows) and its thermopower (how much voltage appears when one side is heated). But those predictions had never been tested in a real experiment, because it is hard to grip a bulky, twisted molecule firmly enough at the single-molecule level.
Designing a Molecule That Grabs Gold
To solve this, the researchers crafted a special three-dimensional carbohelicene, labeled molecule 2, with an asymmetric surface. One face of the helix is shielded by bulky tert-butyl groups that keep the molecule soluble and prevent it from stacking into thick piles. The opposite face is left relatively exposed, presenting a broad, flat carbon surface that can sit close to a gold electrode. When gold surfaces are dipped into a solution of this molecule, the exposed face lies down on the metal and forms many weak but cooperative contacts between the carbon network and the gold. Scanning tunneling microscope images reveal that this design produces a neat, upright monolayer of helicenes on a gold (111) surface, unlike a fully shielded reference molecule that adsorbs in a more disordered way.
Building and Squeezing Single-Molecule Bridges
With this ordered layer in place, the team repeatedly brought a sharp gold tip toward and away from the gold surface, so that individual helicene molecules could momentarily bridge the gap between them. By measuring the current as the distance changed, they extracted the characteristic conductance of a single molecule. They found a well-defined conductance peak in the range of about 10⁻³–10⁻² of the conductance quantum, noticeably higher and sharper than in many previous helicene junctions that relied on single-point chemical anchors like sulfur or nitrogen atoms. When the electrodes were pulled apart (the "breaking" process), the conductance stayed nearly constant over distance. When the electrodes were pushed together, partially compressing the helical backbone, the conductance rose and, under strong compression, additional higher-conductance configurations appeared, showing that the molecular spring could be mechanically tuned.

Heat-to-Voltage Conversion Under Mechanical Control
The researchers then explored what happens when one side of the molecular bridge is heated relative to the other. Under a small temperature difference, they measured the tiny voltage that developed across the junction, which reveals the thermopower. They discovered that the way the junction is formed—by pulling apart or by pushing together—matters greatly. In the breaking pathway, with sharper electrode tips and gentler strain on the molecule, the thermopower settled around −15 microvolts per kelvin, comparable to many other carbon-based single-molecule systems. In the pushing pathway, where blunter electrodes compress the helicene and increase its contact area and internal π–π stacking, the thermopower climbed to about −44 microvolts per kelvin, among the highest values reported for π-conjugated single-molecule junctions. This indicates that subtle changes in the molecule’s shape and its alignment with the metal energy levels strongly affect how efficiently it converts a temperature difference into electrical energy.
What This Means for Future Tiny Devices
In simple terms, this work proves that a single, spring-shaped carbon molecule can act as a mechanically tunable element for both electrical conductance and thermoelectric response. By carefully exposing one side of the molecule to form many gentle contacts with gold, the authors created robust, reproducible single-molecule circuits that can be squeezed to boost current and heat-to-voltage conversion. This experimental confirmation of earlier theory suggests that three-dimensional helical carbon frameworks are promising building blocks for ultra-small, mechanically responsive thermoelectric components, and provides a blueprint for future molecular devices that harness not only charge, but also heat and even spin, through smart molecular design.
Citation: Fujii, S., Morita, F., Takahashi, K. et al. Partially π-exposed 3D carbohelicene for mechanical tuning of conductance and thermopower in single-molecule junctions. Nat Commun 17, 3702 (2026). https://doi.org/10.1038/s41467-026-71293-3
Keywords: single-molecule electronics, helicene, thermoelectric nanodevices, metal–pi interactions, molecular junctions