Clear Sky Science · en
A universal atomically dispersed cobalt catalyst for alkylation of ketones alcohols and lignin-derived compounds
Turning Plant Waste into Useful Molecules
Chemists have long relied on fossil fuels and wasteful reagents to build the carbon–carbon bonds that make up medicines, plastics, and countless everyday materials. This study shows how to do much of that work more cleanly, by using a tiny amount of cobalt metal, spread as single atoms on a porous support, to join together simple alcohols and related compounds—including ones made from plant waste—into more valuable products while generating almost no waste.
Why Cleaner Bond-Making Matters
Modern life depends on reactions that connect carbon atoms, yet many of these processes use corrosive halogenated chemicals and metal reagents that must be made and then thrown away. They also draw heavily on oil and gas. The authors aim to support a more circular carbon economy, in which abundant, renewable sources such as biomass replace fossil feedstocks, and in which reactions use every atom efficiently. They focus on reactions between ketones and alcohols, building blocks that already appear in many drugs, agrochemicals, and natural products, and that can be sourced from both petroleum and from biomass such as lignin, the tough aromatic material in wood.
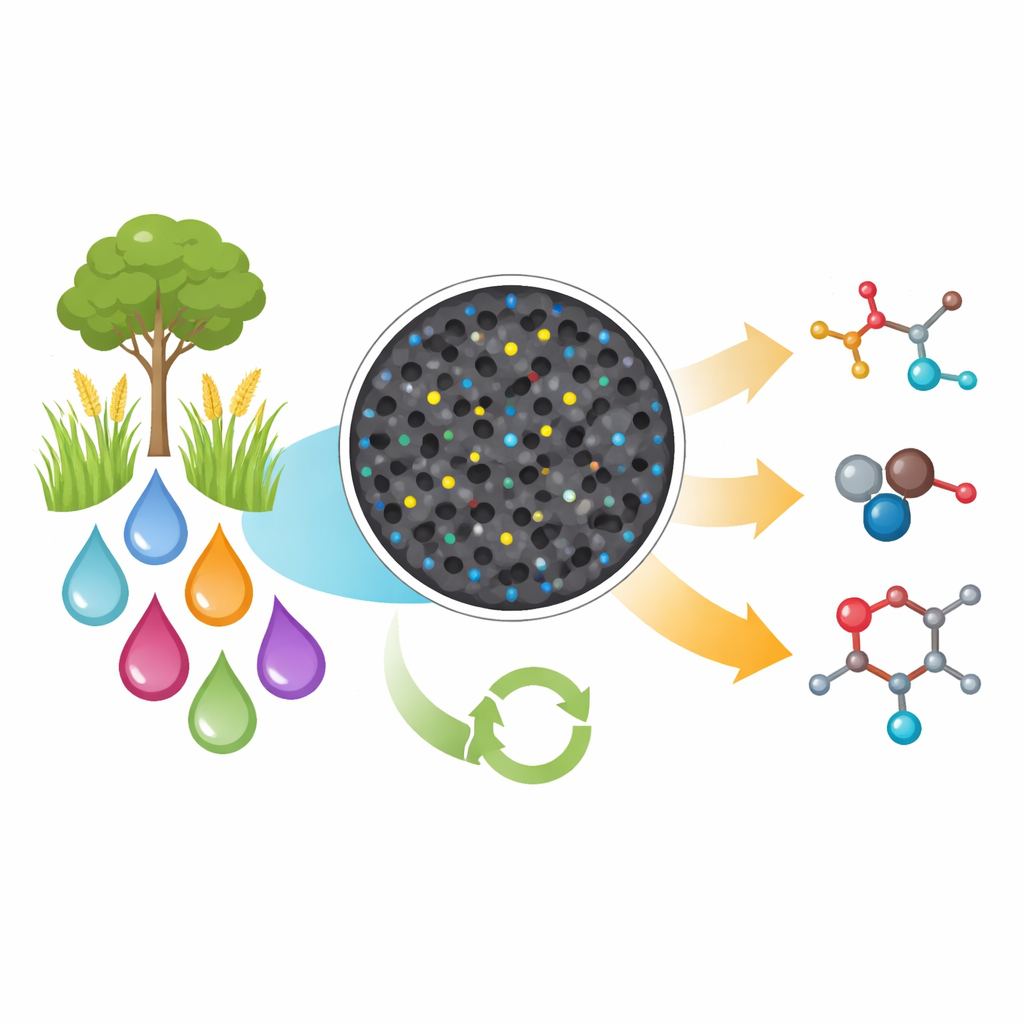
A Tiny but Powerful Cobalt Catalyst
To achieve this goal, the team designed a solid catalyst in which individual cobalt atoms are anchored in a nitrogen-doped carbon framework. They first grow a nitrogen-rich polymer together with cobalt on silica particles, then heat the mixture to high temperature and chemically remove the silica. What remains is a sponge-like carbon material riddled with tiny pores and decorated with isolated cobalt atoms each bound to four nitrogen atoms (so‑called Co–N4 sites). Advanced imaging and spectroscopic methods confirm that the metal does not form larger nanoparticles but is dispersed as single atoms, which turns out to be crucial for activity and selectivity.
How the Reaction Recycles Its Own Hydrogen
The key chemistry uses a strategy known as "borrowing hydrogen." In simple terms, the cobalt site first pulls hydrogen atoms off an alcohol and a lignin-derived fragment, briefly turning them into more reactive partners. These then combine to form a new carbon–carbon bond. Finally, the temporarily stored hydrogen is returned to the product, yielding a stable alkylated ketone or alcohol. Water is the only by-product, and no extra reducing agents are needed. Careful experiments track the appearance and disappearance of intermediate species and show that when the cobalt–nitrogen sites are blocked, the reaction almost stops, underscoring their central role in this hydrogen shuttle.
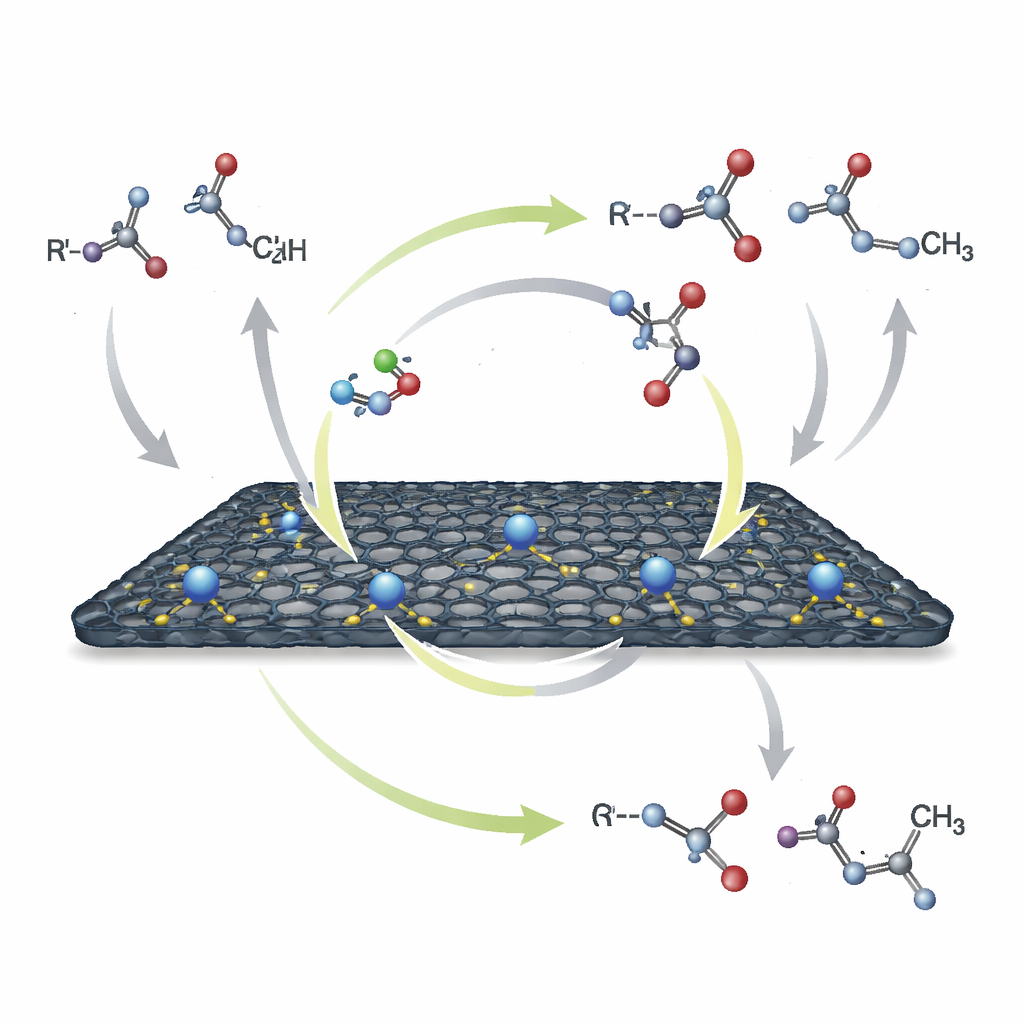
From Model Compounds to Drug-Like Products
Once the optimal material was in hand, the researchers demonstrated how broadly it can be applied. It efficiently breaks specific carbon–oxygen bonds in lignin-like molecules and joins the fragments to a wide variety of primary alcohols, including benzylic, heterocyclic, and even challenging aliphatic examples. The same catalyst joins ordinary ketones with alcohols, and can couple secondary alcohols with primary ones to give either ketones or higher alcohols, depending on the base and temperature. It also performs selective "methylation" using simple methanol instead of hazardous methylating agents. In several cases, the method builds or modifies molecules related to pharmaceuticals, showing that it can handle complex, delicate structures.
Stable, Reusable, and Ready for Industry
Because the cobalt atoms are locked into the carbon framework, they do not leach into solution, and the solid catalyst can be filtered off and reused several times with little loss of performance. Tests on an industrially important mixture called KA oil—a precursor for nylon—demonstrate that the material can process bulk feedstocks as well as fine chemicals, and that it can help connect a renewable route from lignin-derived phenol all the way to nylon building blocks. By using an abundant metal, operating without added hydrogen or stoichiometric reagents, and working with renewable alcohols, the system points toward more sustainable large-scale chemical manufacturing.
What This Means for a Greener Future
In everyday terms, the work provides a "universal" solid cobalt catalyst that can take simple, often plant-based alcohols and convert them into a wide range of more valuable molecules, from fuel-like higher alcohols to drug candidates, while generating almost no chemical waste. The single cobalt atoms act like precision tools on a reusable scaffold, shuttling hydrogen back and forth rather than consuming it. This approach showcases how clever catalyst design can help the chemical industry move away from fossil resources and toxic reagents, and toward cleaner, more efficient ways of making the molecules society depends on.
Citation: Ma, Z., Zhang, B., Cui, Y. et al. A universal atomically dispersed cobalt catalyst for alkylation of ketones alcohols and lignin-derived compounds. Nat Commun 17, 3214 (2026). https://doi.org/10.1038/s41467-026-71275-5
Keywords: single-atom catalysis, borrowing hydrogen, lignin valorization, cobalt catalyst, green chemistry