Clear Sky Science · en
Abiotic CO2 reduction promoted by carbonate and phyllosilicate minerals on the primitive seafloor
Rocks That Helped Seed Life
The seafloor of the early Earth was far from quiet stone; it may have been a vast natural factory turning simple gas into the building blocks of life. This study shows that very common seafloor minerals, lightly coated with trace metals, can turn carbon dioxide into a range of organic molecules without any help from biology. Understanding this rocky chemistry not only offers clues to how life began on Earth, but also points to ways similar processes could be shaping potentially habitable worlds like Mars and the icy moons of the outer solar system.
Electricity From Stone and Water
On the young Earth, water percolating through hot, iron-rich rocks would have created strong chemical contrasts between fluids and surrounding minerals. These contrasts can generate natural electrical currents, a phenomenon known as geoelectrochemistry. Earlier work showed that certain rare sulfide minerals can use this rock-powered electricity to reduce carbon dioxide. The new research asked a broader question: can far more abundant minerals, such as carbonates and sheet-like phyllosilicates that blanket much of the seafloor, also act as catalysts for turning carbon dioxide into useful organics under such conditions?
Common Minerals With a Metal “Boost”
The team tested a wide range of carbonate minerals containing elements like magnesium, calcium, iron, and manganese, as well as natural carbonate and phyllosilicate samples. On their own, most of these minerals did little more than split water to make hydrogen gas. The picture changed dramatically when tiny amounts of transition metal ions, especially copper and zinc, were allowed to adsorb onto the mineral surfaces. Under conditions mimicking early oceans and hydrothermal vents, these metal-decorated minerals converted carbon dioxide into methane, carbon monoxide, formic acid, and simple two-carbon organics such as ethylene and ethanol. Isotope labeling confirmed that the carbon in these products came directly from carbon dioxide. 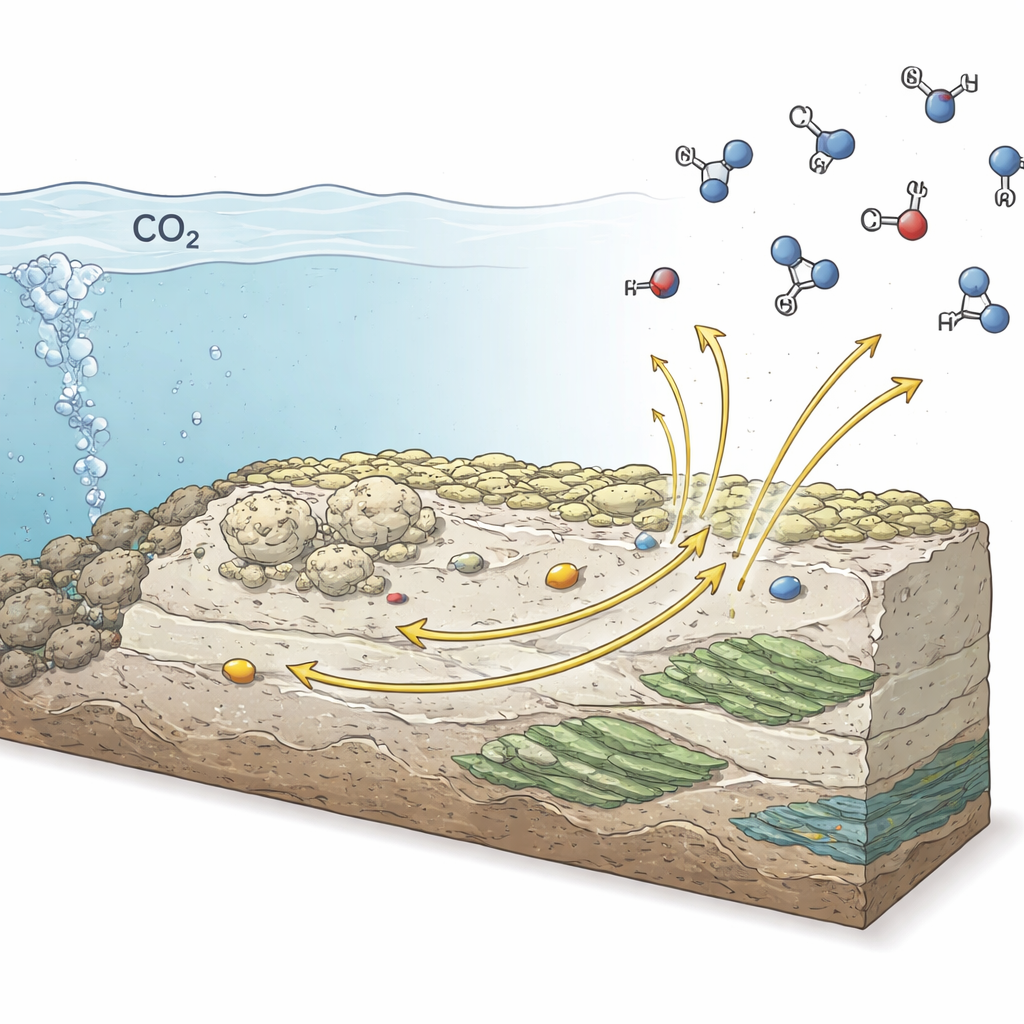
How the Seafloor Acts Like a Subtle Battery
Detailed measurements revealed how this rocky chemistry works. During electrochemical reactions, some of the adsorbed metal ions on the mineral surfaces were partially reduced to tiny patches of native metal, which acted as the main active sites for transforming carbon dioxide. At the same time, the carbonate or phyllosilicate host helped split water and shuttle the resulting protons to the reaction sites, speeding up the stepwise “hydrogenation” of carbon dioxide into more complex molecules. Spectroscopic fingerprints of short-lived intermediates—such as partially hydrogenated carbon species—appeared only when the metal-coated minerals were present, underscoring the partnership between metal specks and mineral substrates in driving the reaction.
Adding Nitrogen and Expanding the Recipe
The researchers also explored what happens when nitrogen in the form of ammonia is available, as it likely was in some early Earth and extraterrestrial waters. In the presence of copper-decorated carbonates and ammonia, the system produced noticeable amounts of acetamide, a simple molecule containing carbon, hydrogen, oxygen, and nitrogen. Acetamide is of particular interest because it can help link nucleosides to form nucleotides and is involved in modern cell wall chemistry, hinting at ways early mineral-driven reactions might have fed into primitive metabolic networks. Phyllosilicate minerals, once partially carbonated at their surfaces, showed broadly similar behavior, with their own subtle influence on which organics were favored. 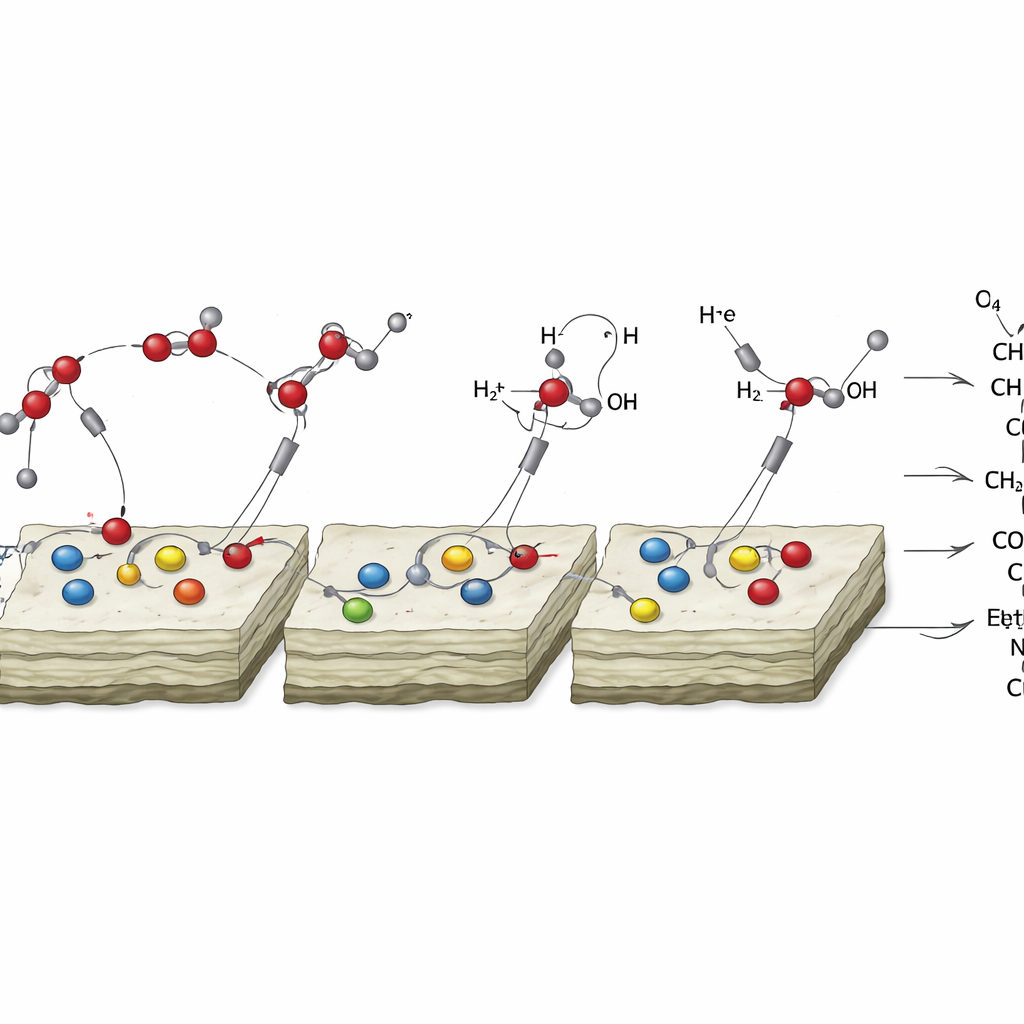
From Ancient Ocean Floors to Other Worlds
By building a vent-like reactor powered by hydrogen oxidation, the team demonstrated that this mineral-catalyzed carbon dioxide reduction can proceed under realistic seafloor temperatures and without an external power supply, relying solely on rock–water chemistry. This suggests that on the early Earth, widespread carbonate and phyllosilicate deposits coated with trace metals like copper and zinc could have continually generated a mix of organic compounds in and around hydrothermal systems and impact-altered crust. Similar settings on Mars and on icy moons with hidden oceans may still host comparable reactions today. In plain terms, the study shows that ordinary rocks, given a sprinkle of metal and a natural electrical push, are capable of turning a simple greenhouse gas into ingredients that life can use.
Citation: Zhong, Y., Zhang, N., Huan, D. et al. Abiotic CO2 reduction promoted by carbonate and phyllosilicate minerals on the primitive seafloor. Nat Commun 17, 3229 (2026). https://doi.org/10.1038/s41467-026-71130-7
Keywords: origin of life, hydrothermal vents, CO2 reduction, mineral catalysis, planetary habitability