Clear Sky Science · en
Dual-site single-atom catalysts achieve directional adsorption-oxidation control for enhanced photo-Fenton-like reactions
Cleaning Stubborn Pollutants from Water
Many of today’s medicines, pesticides, and industrial chemicals are so stable that they slip through conventional wastewater treatment and accumulate in rivers, lakes, and even drinking water. This study explores a new kind of light-powered catalyst designed to break down these persistent pollutants more efficiently and with lower environmental cost, potentially offering a practical path toward cleaner, safer water.
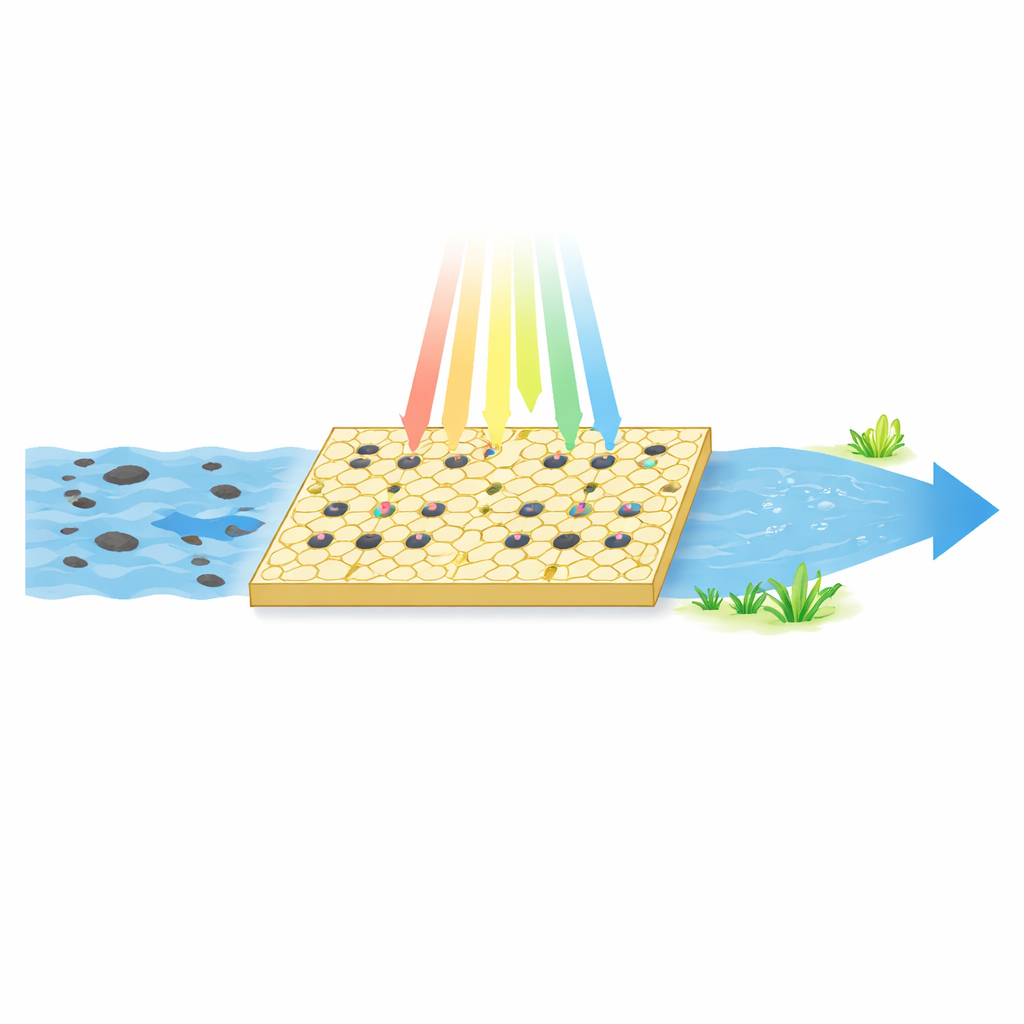
A Tiny Factory on a Solid Surface
The researchers focus on a class of materials called single-atom catalysts, where individual metal atoms are anchored on a solid support. Because every metal atom is exposed and available, these catalysts can be extremely efficient. The team uses a yellow, sheet-like material called graphitic carbon nitride as the support and decorates it with isolated iron atoms. They also deliberately remove some nitrogen atoms from the structure, leaving behind “vacancies.” The result is a dual-site catalyst: one type of site is the iron atom, and the other is the nearby nitrogen vacancy. This pairing is engineered to work together under visible light to attack organic contaminants such as the antibiotic tetracycline in water.
Dividing the Work to Go Faster
In conventional Fenton-like systems, the same active spot on a catalyst must both grab the pollutant and activate an oxidizing agent, which can cause the two processes to compete and slow each other down. In this new design, the roles are split. The nitrogen vacancies preferentially attract and hold pollutant molecules near the surface, while the neighboring iron atoms specialize in activating an oxidant called peroxymonosulfate. When visible light shines on the material, electrons are excited and tend to collect in the vacancies, then flow toward the iron atoms. This directional electron traffic makes it easier for iron to repeatedly convert the oxidant into highly reactive species that can attack the pollutants. Experiments and computer simulations show that this close cooperation between vacancy and metal atom greatly speeds up pollutant breakdown compared with earlier designs.
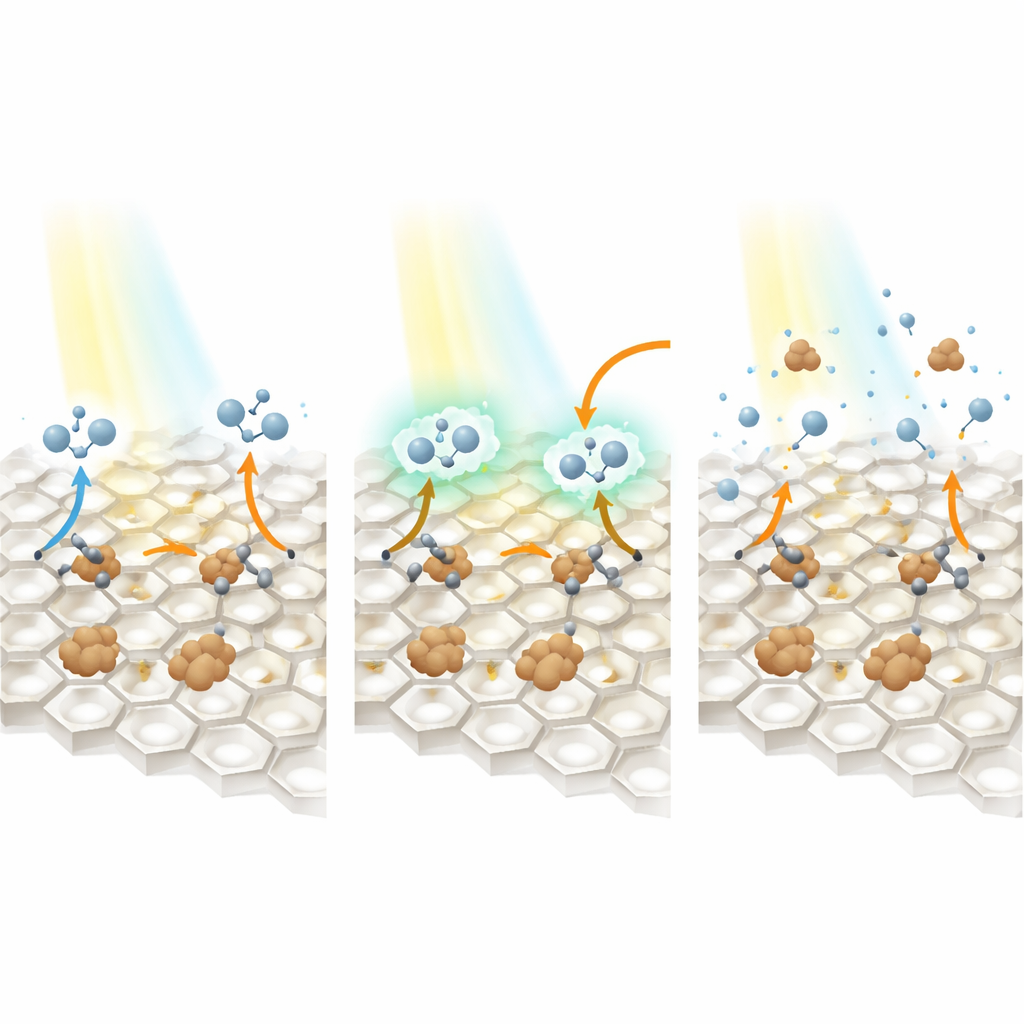
How Light Helps Do the Heavy Lifting
To understand why the material performs so well, the team probes how it absorbs light and manages electric charges. Measurements reveal that adding iron and nitrogen vacancies allows the sheets to harvest more visible light and sharply reduces the tendency of electrons and holes to recombine and cancel each other out. Ultrafast laser techniques show that charge carriers move more quickly and more purposefully in the modified material than in the unmodified carbon nitride. Electrochemical tests confirm that the dual-site catalyst has lower resistance to charge transfer, meaning it can shuttle electrons efficiently between light, oxidant, and pollutant. Together, these effects ensure that a larger fraction of the absorbed light energy is converted into chemical work—namely, generating both radical and non-radical oxidizing species that chew through organic molecules.
Following Pollutants as They Fall Apart
The researchers track how tetracycline is transformed in this system, using advanced calculations and chemical analyses to map out vulnerable positions in the molecule and the likely attack pathways. They identify multiple intermediate fragments and show that different reactive species target different parts of the pollutant, eventually breaking it down into carbon dioxide, water, and small inorganic ions. To test real-world safety, they examine treated water with bacteria, zebrafish embryos, and plant seeds. Compared with untreated samples, the water that passed through the dual-site, light-driven process shows sharply reduced toxicity, suggesting that harmful compounds are not merely being rearranged but are truly detoxified.
Designing Smarter Systems with Data
Because many factors influence performance—such as catalyst composition, pH, contact time, and water source—the team also applies machine learning to their experimental data. They train models to predict how efficiently pollutants will be removed under different conditions and to identify which variables matter most. This analysis highlights reaction time, catalyst modification, and the fine balance of nitrogen content and metal choice as key levers for performance. It also shows that the iron-based, vacancy-rich catalyst offers an advantageous combination of efficiency, stability, and relatively low cost and toxicity compared with some alternatives.
From Lab Concept to Cleaner Water
For non-specialists, the main takeaway is that the authors have built a microscopic “assembly line” where one site on the catalyst concentrates pollutants and another repeatedly activates a powerful oxidant, all driven by visible light. By carefully arranging these sites side by side and tuning how electrons move between them, the system breaks down stubborn contaminants quickly while using very little metal and reducing energy and environmental overhead. With further development and scaling, such dual-site, light-assisted catalysts could become an important tool for turning polluted water into a safer resource in a more sustainable way.
Citation: Bai, CW., Sun, YJ., Huang, XT. et al. Dual-site single-atom catalysts achieve directional adsorption-oxidation control for enhanced photo-Fenton-like reactions. Nat Commun 17, 2958 (2026). https://doi.org/10.1038/s41467-026-70907-0
Keywords: wastewater treatment, single-atom catalysts, advanced oxidation, visible-light photocatalysis, peroxymonosulfate