Clear Sky Science · en
Coagulation pretreatment could deteriorate reverse osmosis membrane fouling
Why cleaner water is not always simple
Modern industries are under growing pressure to reuse water and release almost no liquid waste. A key technology for this "zero liquid discharge" approach is reverse osmosis, in which water is forced through a thin membrane that blocks salts and pollutants. To protect these delicate membranes, engineers commonly add chemicals that clump tiny particles together before filtration. This study shows that such pretreatment, long assumed to help, can surprisingly make membrane clogging worse and weaken the overall system.
How factories try to squeeze out every drop
Many power plants and chemical factories now rely on multi-step treatment lines to clean salty, dirty wastewater. After large debris is removed, a common next step is coagulation, where iron- or aluminum-based salts are mixed into the water so that fine particles and natural organic matter stick together and can be filtered out. The remaining water then passes through low-pressure filters and finally through high-pressure reverse osmosis units that separate clean water from concentrated brine. In traditional seawater desalination, this strategy works well. But in industrial systems that use membranes instead of sand filters upfront, some of the metal coagulants slip through, and operators have noticed puzzling, faster-than-expected fouling of the reverse osmosis membranes.
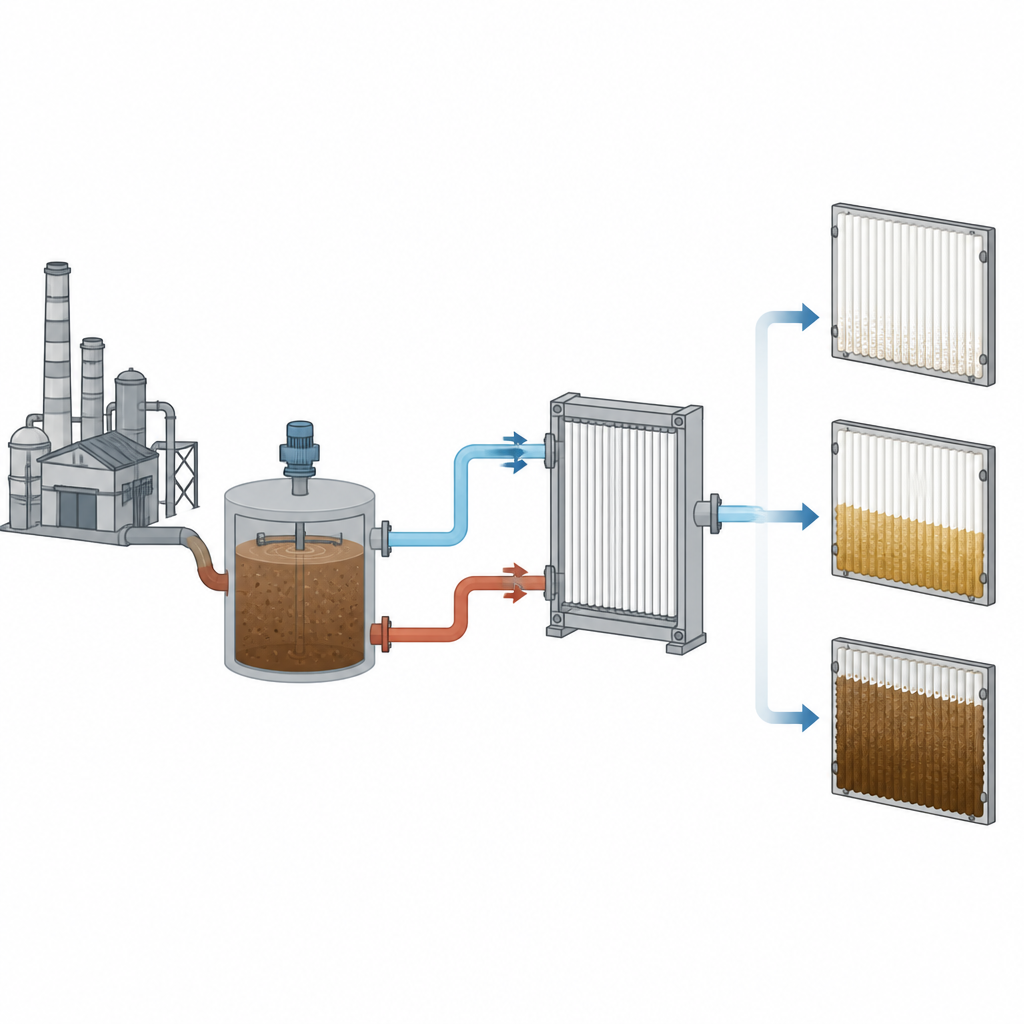
Testing different pretreatment choices side by side
The researchers built a pilot-scale treatment line using real desulfurization wastewater from a coal-fired power plant. They compared three setups: one with no coagulant, one using an aluminum salt, and one using an iron salt, while keeping pressure, flow, and starting performance the same. Over 20 days, all membranes gradually lost efficiency, but the loss was much more severe when coagulants were used. The control system without coagulation lost about one quarter of its water flow, the aluminum-treated system lost about half, and the iron-treated system lost more than two thirds. Microscopic images revealed that the fouling layers were thinner and more open in the control case, thicker under aluminum treatment, and thickest and most compact under iron treatment.
What happens on the membrane surface
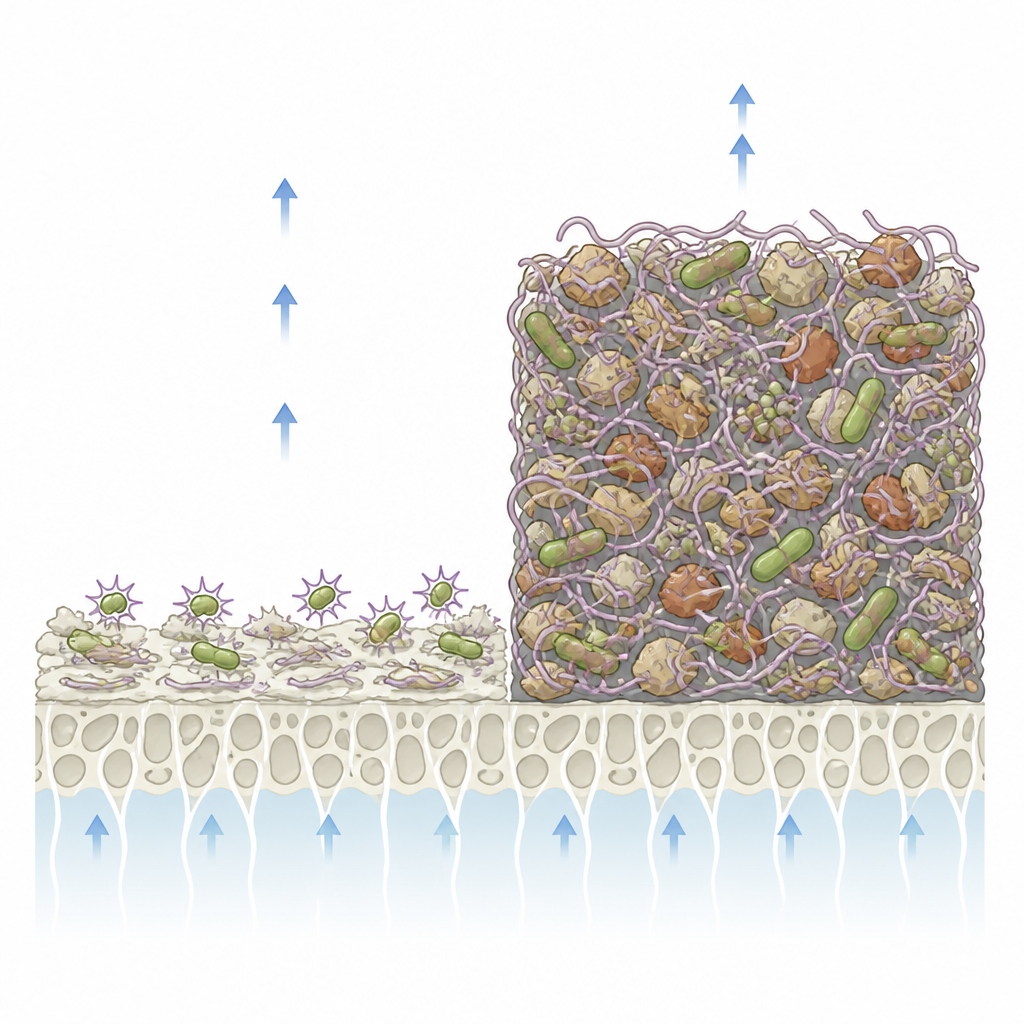
By combining electron microscopy, chemical analysis, and fluorescence methods, the team dissected what built up on the membranes. Aluminum pretreatment favored mainly inorganic scale, with metals such as copper depositing alongside aluminum and other minerals. This created a relatively brittle layer where microbial activity was partly suppressed. In contrast, iron pretreatment produced a rich mixture of inorganic particles, organic matter, and dense biological growth. Iron accumulated strongly on the surface and existed in forms that microorganisms could actively use. This encouraged them to secrete large amounts of sticky polymers, creating a thick, cross-linked slime that trapped more particles and made the layer increasingly water-repellent.
Microbes and metals working together
Genetic sequencing showed that the mix of microbes living in the fouling layer changed markedly with each pretreatment. In the control case, a few familiar bacteria dominated, producing enough slime to foul the membrane but not forming an extremely complex community. Under iron-rich conditions, however, a broader range of species that excel at binding iron, resisting copper, and producing slime-like polymers flourished. Their interaction network was more stable and tightly connected, supporting vigorous biofilm growth. The researchers also found that genes linked to iron uptake, carbohydrate and amino acid use, and production of protective polymers were strongly boosted. In aluminum-rich conditions, microbes faced stronger copper-driven oxidative stress, with higher levels of internal damage and weaker protective systems, which limited but did not prevent fouling.
Rethinking how we prepare water for tight membranes
Overall, the study explains why a pretreatment step that seems helpful at first glance can actually backfire in modern industrial systems. Residual iron in particular turns the membrane surface into a fertile ground for hardy, slime-producing microbes that build thick, stubborn fouling layers, while aluminum shifts the balance toward mineral crusts and stressed communities. For engineers, this means that simply adding more coagulant is not a safe route to cleaner membranes. Instead, designs should limit how much metal reaches the reverse osmosis stage, for example by using tighter filters or sand beds after coagulation, carefully tracking residual metals and organic matter, and possibly choosing alternative coagulants. In clear terms, the work shows that protecting high-performance membranes requires seeing pretreatment, chemistry, and microbiology as one connected system.
Citation: Ding, H., Liang, S., Lin, W. et al. Coagulation pretreatment could deteriorate reverse osmosis membrane fouling. Nat Commun 17, 4168 (2026). https://doi.org/10.1038/s41467-026-70892-4
Keywords: reverse osmosis, membrane fouling, zero liquid discharge, industrial wastewater, coagulation