Clear Sky Science · en
Long-term brain pressure monitoring via a discrete microimplant; a first-in-human safety and initial efficacy trial in adults and children with hydrocephalus
Why Watching Brain Pressure at Home Matters
For people living with hydrocephalus—a condition where fluid builds up in the brain—every bad headache can feel like an emergency. Families often rush to the hospital fearing that a tiny drainage tube, or shunt, has failed, yet most of these visits turn out to be false alarms. This study describes a new rice‑grain–sized brain implant that quietly measures pressure inside the skull and sends readings wirelessly to a phone and then to doctors. It aims to turn guesswork into data, letting care happen earlier, at home, and with less fear.
A Tiny Sensor for a Big Problem
Hydrocephalus is usually treated by surgically placing a shunt to drain excess brain fluid. These shunts fail at strikingly high rates—about half within two years—most often signaled only by vague symptoms like headache or irritability. Children, especially those who cannot yet talk, are hardest to assess. Today, decisions rely on hospital scans and cautious observation rather than continuous measurements of what really matters: the pressure inside the skull. The team set out to build a very small, long‑lasting sensor that could sit safely in brain tissue, track pressure over years, and send readings from a patient’s home to their clinical team.
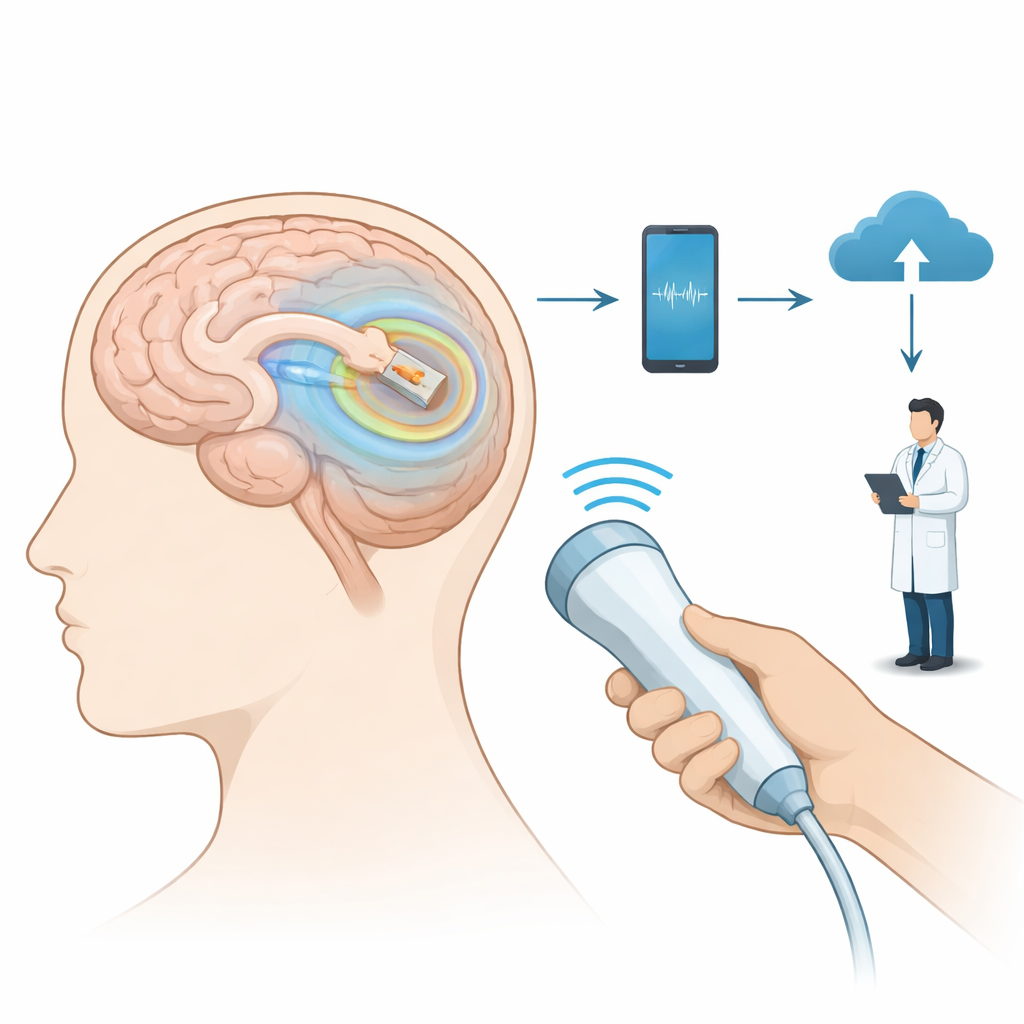
How the Implant and Home System Work
The device is a slender glass‑encased capsule, just a few millimeters wide, designed to be inserted into the brain’s outer layer during routine shunt surgery through a small additional opening in the skull. All surfaces that touch tissue are smooth borosilicate glass, and the electronics are sealed in a separate cavity to keep fluid out. Instead of using a battery, the implant is powered wirelessly by a handheld wand placed on the scalp. When the wand is activated, it briefly sends energy into the implant and then listens while the sensor beams back pressure readings several dozen times per second. The wand passes this information to a smartphone app and then to a secure cloud portal where doctors can review long‑term trends.
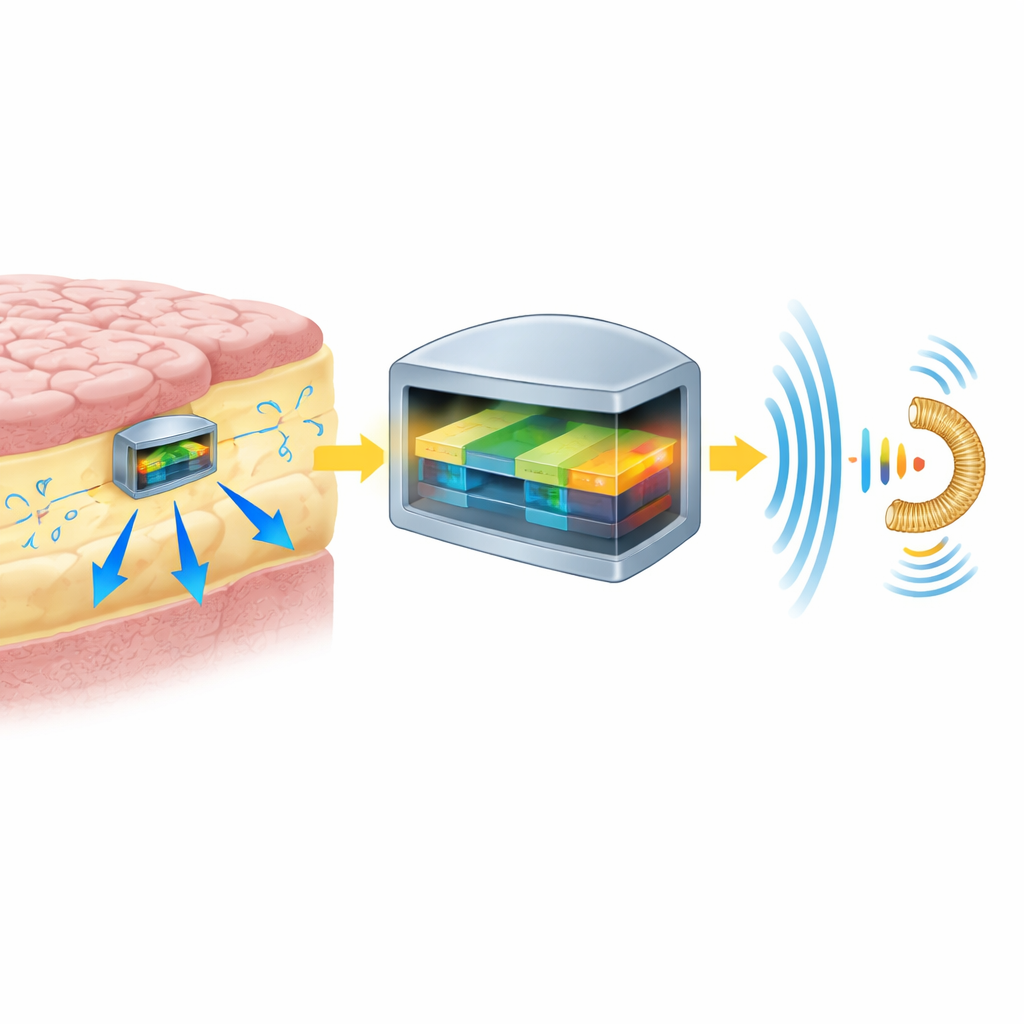
Reliable Signals Without Disturbing the Brain
Making such a tiny device work safely inside the brain required careful engineering. The pressure sensor converts pressure into a fully digital signal, which helps avoid drift—slow changes in readings that would normally require recalibration. Lab tests showed very small changes over a year and projected only modest drift over a decade. The team used low‑frequency wireless power to keep tissue heating well below safety limits and developed a special technique that alternates between power delivery and data transmission so the pressure signals are not drowned out by the power field. Animal studies in sheep showed that the glass implant provoked only a thin, stable scar around it, with no signs of nerve cell loss or toxic effects, and the device stayed in place over many months.
First Experiences in Adults and Children
With encouraging preclinical results, the researchers tested the system in 20 people with hydrocephalus, half of them children as young as 18 months. The sensor was placed during shunt insertion or revision, adding only a few extra minutes to surgery, and no complications related to the device were reported. After going home, participants or their caregivers were asked to perform regular readings using the wand. Over more than 2,500 home measurements and up to 600 days of follow‑up, no sensor failures occurred. In stable patients, pressure values and the small heartbeat‑related pulses riding on each pressure wave remained steady over time and changed predictably with body position, suggesting that the sensor stayed accurate and unobstructed.
Real‑World Cases and Fewer Scares
The most compelling evidence came from individual cases. In one toddler, the sensor showed clearly rising pressure alongside a severe headache; scanning then confirmed a blocked shunt, and surgery to replace it brought both the pressure and symptoms back down. In an older adult, deliberately turning a shunt setting “off” caused both average pressure and pulse amplitude to rise, with symptoms worsening; restoring the setting reversed the pattern. In contrast, another child had two worrying hospital visits for fever and unsteadiness, yet the sensor showed pressures firmly within that child’s normal range, and no shunt surgery was needed. Across the group, families reported feeling less anxious because they could see objective data rather than relying only on how a child seemed.
A New Way to Care for Vulnerable Brains
This early trial shows that a tiny, fully implanted brain pressure sensor can be placed safely in adults and children, function reliably for many months, and provide meaningful information that helps distinguish true shunt problems from false alarms. If adopted more widely, such systems could shift hydrocephalus care from crisis‑driven hospital visits to proactive, data‑guided management at home, easing strain on families and health services alike. Beyond hydrocephalus, the same micro‑implant approach could open the door to long‑term monitoring of pressure in other parts of the body, quietly turning hidden physiologic signals into actionable insight.
Citation: Malpas, S.C., Wright, B.E., Guild, SJ. et al. Long-term brain pressure monitoring via a discrete microimplant; a first-in-human safety and initial efficacy trial in adults and children with hydrocephalus. Nat Commun 17, 3158 (2026). https://doi.org/10.1038/s41467-026-70864-8
Keywords: hydrocephalus, intracranial pressure, brain implant, remote monitoring, shunt failure