Clear Sky Science · en
Enhancer-mediated Etv4 activation stimulates osteogenic differentiation
Stronger Bones from Our Genes
As people age, fragile bones and fractures become a daily worry, especially for those with osteoporosis. Most current drugs work by slowing bone loss or briefly boosting bone building, but they can have side effects and don’t help everyone. This study explores a different angle: how tiny switches in our DNA control the cells that build bone. By uncovering one such switch and the genetic circuit it controls, the researchers point to new, more precise ways to keep bones strong.
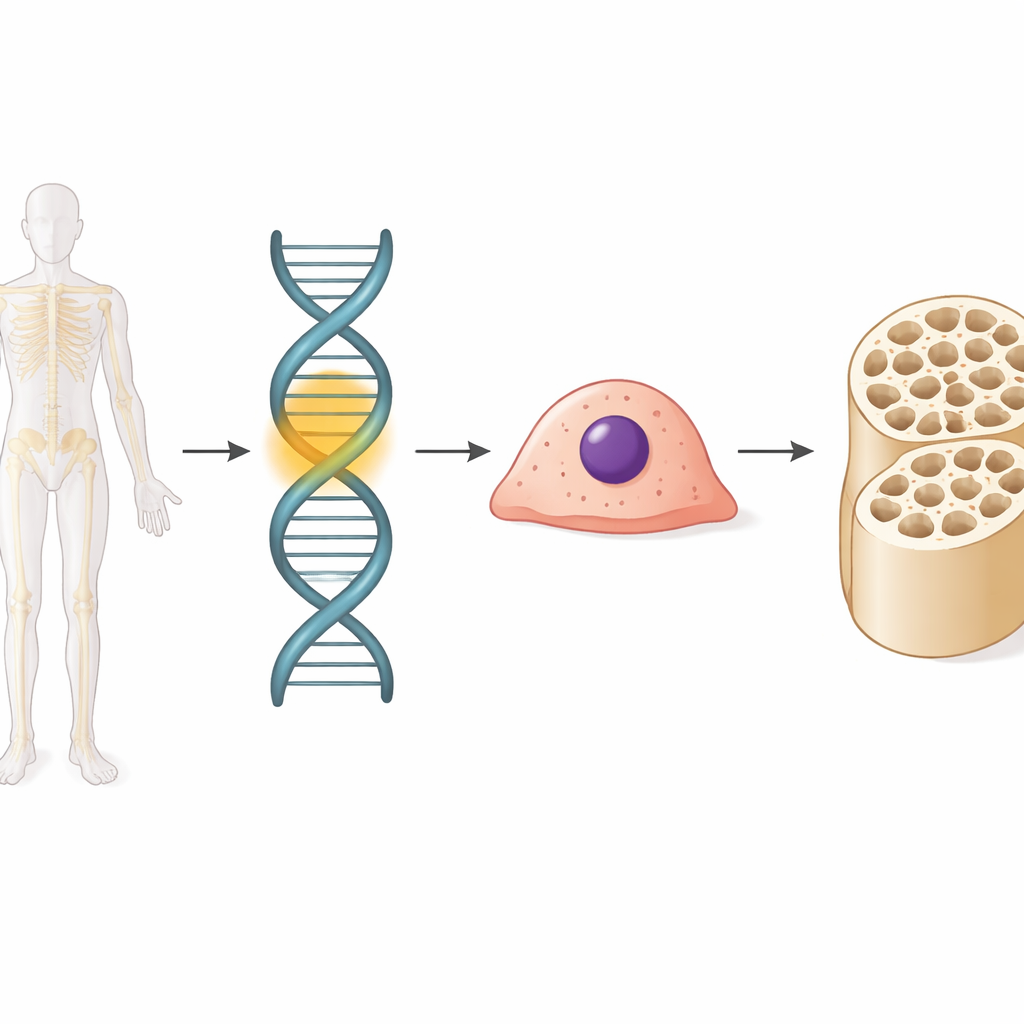
A Hidden Switch in the Genome
Our DNA carries not only genes but also many small control regions that act like dimmer switches for those genes. These switches, called enhancers, don’t make proteins themselves but decide when and how strongly nearby genes turn on. The team sifted through large public datasets that track which DNA regions are active in bone-forming cells. Comparing human and mouse stem cells before and after they turn into bone-building osteoblasts, they identified thousands of enhancers that change activity. Among these, one stood out: a conserved element they named enh11, whose sequence is shared across many mammals and which lights up specifically as stem cells commit to becoming osteoblasts.
From Switch to Weaker Bones in Mice
Finding an interesting enhancer is only the first step; the real test is whether it matters for living bone. Using CRISPR gene-editing in a mouse osteoblast cell line, the researchers deleted the enh11 region. Without this enhancer, cells were much worse at maturing into bone builders: markers of bone mineralization dropped, and the cells deposited less calcium, a key ingredient of hard bone. The team then created mice in which enh11 is removed only in osteoblasts. These animals, both males and females, grew smaller, with shorter thigh bones and visibly thinner, more porous bone structure under high-resolution scans. Importantly, bone-resorbing cells were not changed, pointing to a specific failure of bone formation rather than overactive bone breakdown.
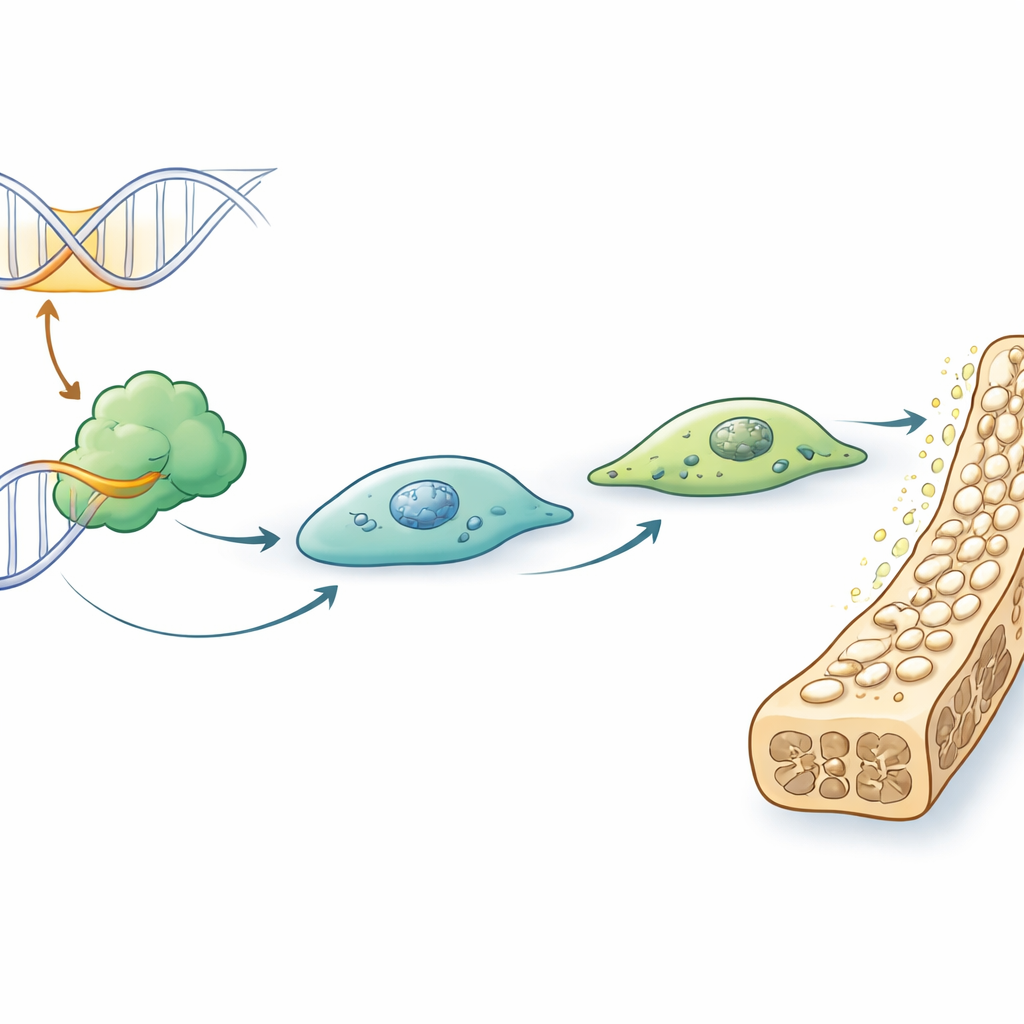
Connecting the Switch to a Key Bone Gene
An enhancer exerts its influence by controlling one or more target genes, sometimes over long stretches of DNA. To find what enh11 talks to, the scientists analyzed three-dimensional genome maps that reveal which distant DNA regions physically touch each other in bone cells. They found that enh11 loops over to the region of a gene called Etv4, known as a transcription factor—essentially a master regulator that can switch many other genes on or off. When enh11 was deleted, Etv4 levels fell. Reporter experiments, in which the enh11 sequence was placed next to the Etv4 control region, showed that it could boost Etv4 activity. Knocking down Etv4 in bone cells produced the same defects in differentiation and mineral deposition seen when enh11 was removed, and mice lacking Etv4 had bones that were lighter, thinner, and structurally weaker, mirroring the enh11-deficient animals.
A Three-Step Control Chain for Bone Building
Digging deeper, the team asked how enh11 itself becomes active. They used mapping techniques to see which proteins bind on top of enh11 and the Etv4 control region, and homed in on a well-known signaling protein called Stat3. Stat3 is activated by various growth factors and hormones and then travels to the nucleus, where it binds DNA. Here, Stat3 was found at enh11 and near Etv4, and its binding was weakened when enh11 was deleted. When Stat3 levels were reduced, Etv4 and bone-related genes dropped, and bone cells again struggled to mature. Removing just the Stat3 binding site within enh11 was enough to blunt osteoblast differentiation. Together, these results outline a chain of command: signals in the body activate Stat3; Stat3 binds to enh11; enh11 boosts Etv4; and Etv4 turns on the genetic program that lets osteoblasts harden and maintain bone.
What This Means for Future Bone Therapies
In simple terms, this work reveals a tiny but powerful control module that helps keep our bones strong. When the enh11 switch and the Stat3–Etv4 circuit work properly, they support the growth and maturation of bone-forming cells and the accumulation of dense, healthy bone. When any part of this chain fails, bones become thinner and more fragile, echoing what happens in osteoporosis. While translating these findings into treatments will take time, enh11, Stat3, and Etv4 now stand out as promising targets. Fine-tuning this natural genetic pathway might one day offer therapies that restore bone strength more precisely and with fewer side effects than today’s drugs.
Citation: Zhang, J., Wang, Q., Cheng, Z. et al. Enhancer-mediated Etv4 activation stimulates osteogenic differentiation. Nat Commun 17, 4096 (2026). https://doi.org/10.1038/s41467-026-70796-3
Keywords: osteoporosis, bone formation, gene regulation, enhancers, osteoblasts