Clear Sky Science · en
Low-intensity focused ultrasound-activated piezoelectric gel bandage for diabetic wound repair and neuropathic pain relief
Healing stubborn sores and easing nerve pain
For many people with diabetes, small foot wounds can quietly turn into deep, stubborn ulcers that refuse to heal and often hurt or burn. These sores raise the risk of infection and even amputation, and current treatments rarely tackle all the problems at once. This study describes a soft, smart bandage that works together with gentle focused ultrasound to both speed wound repair and calm nerve pain in diabetic animals, hinting at a more comfortable and effective way to care for hard-to-heal skin.
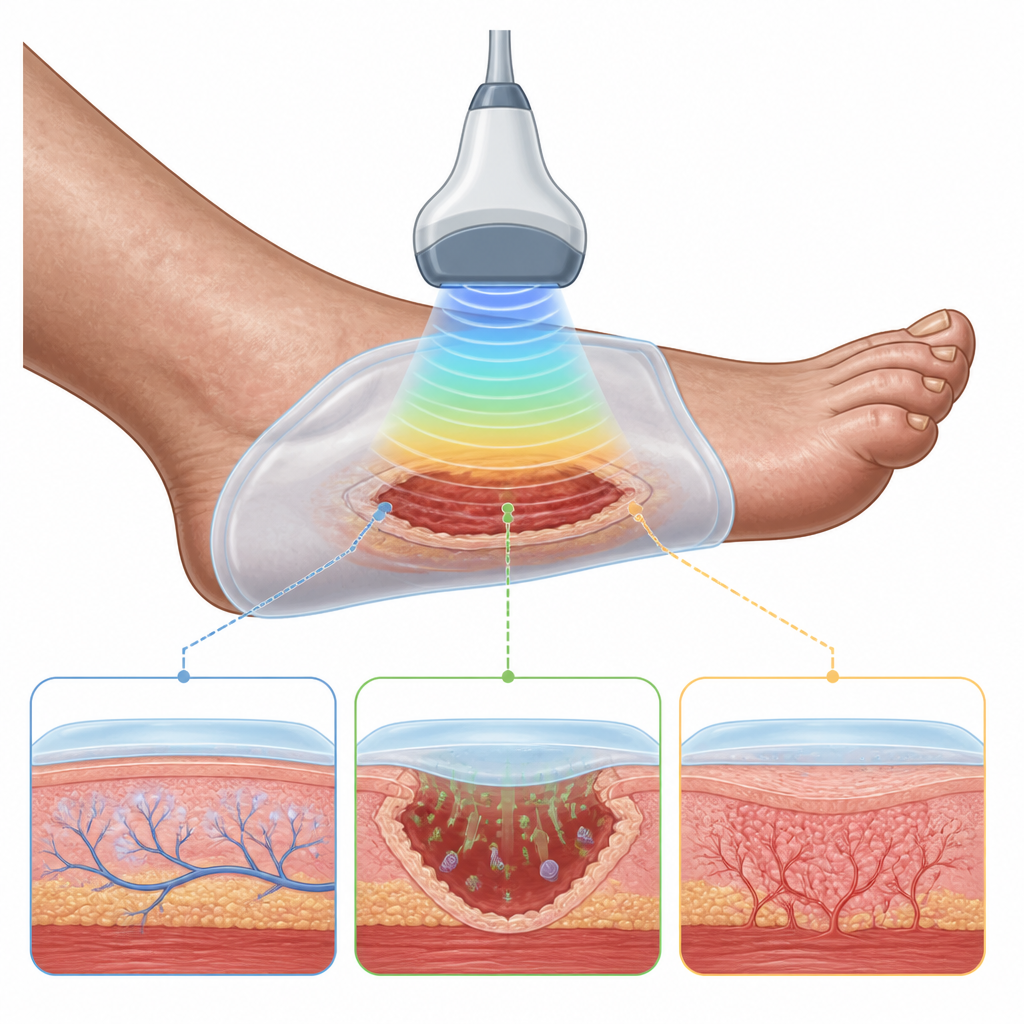
Why diabetic foot wounds are so hard to fix
Diabetic foot ulcers do not linger just because the skin is broken. Long-term high blood sugar disrupts blood vessels, weakens local immune defenses, and damages nerves in the feet. The result is a chronic, inflamed wound environment that heals poorly and is easily invaded by bacteria. At the same time, injured nerves can send distorted pain signals, leaving patients with a mix of numbness and hypersensitivity. Standard care focuses on cleaning the wound, fighting infection, and controlling blood sugar, but often does not restore normal immune balance or address nerve pain in a coordinated way.
A soft bandage that turns sound into gentle energy
The researchers created a flexible gel bandage loaded with special ceramic nanoparticles and a chemical donor that can release nitric oxide, a natural signaling gas in the body. When the bandage is exposed to low-intensity focused ultrasound delivered from outside the body, the sound waves make the ceramic particles slightly deform and generate tiny electrical currents, along with controlled bursts of reactive oxygen species and nitric oxide. The gel itself clings tightly to moist tissue, so it stays in place on irregular wounds, absorbs fluid, stops bleeding, and transmits the ultrasound and electrical effects into deeper layers of skin.
Helping cells fight germs and rebuild tissue
In lab dishes, the ultrasound-activated bandage encouraged the growth and movement of human blood vessel cells, skin cells, and fibroblasts, all of which are essential for closing a wound. The controlled nitric oxide release and small electrical currents nudged immune cells called macrophages away from a pro-inflammatory “attack” mode toward a more soothing, pro-healing state. At the same time, the reactive oxygen species generated by the ceramic particles damaged bacterial membranes and reduced the survival of both common Gram-positive and Gram-negative germs, while remaining within safe limits for healthy human cells. This combination gave the bandage a rare mix of pro-healing and antimicrobial powers.
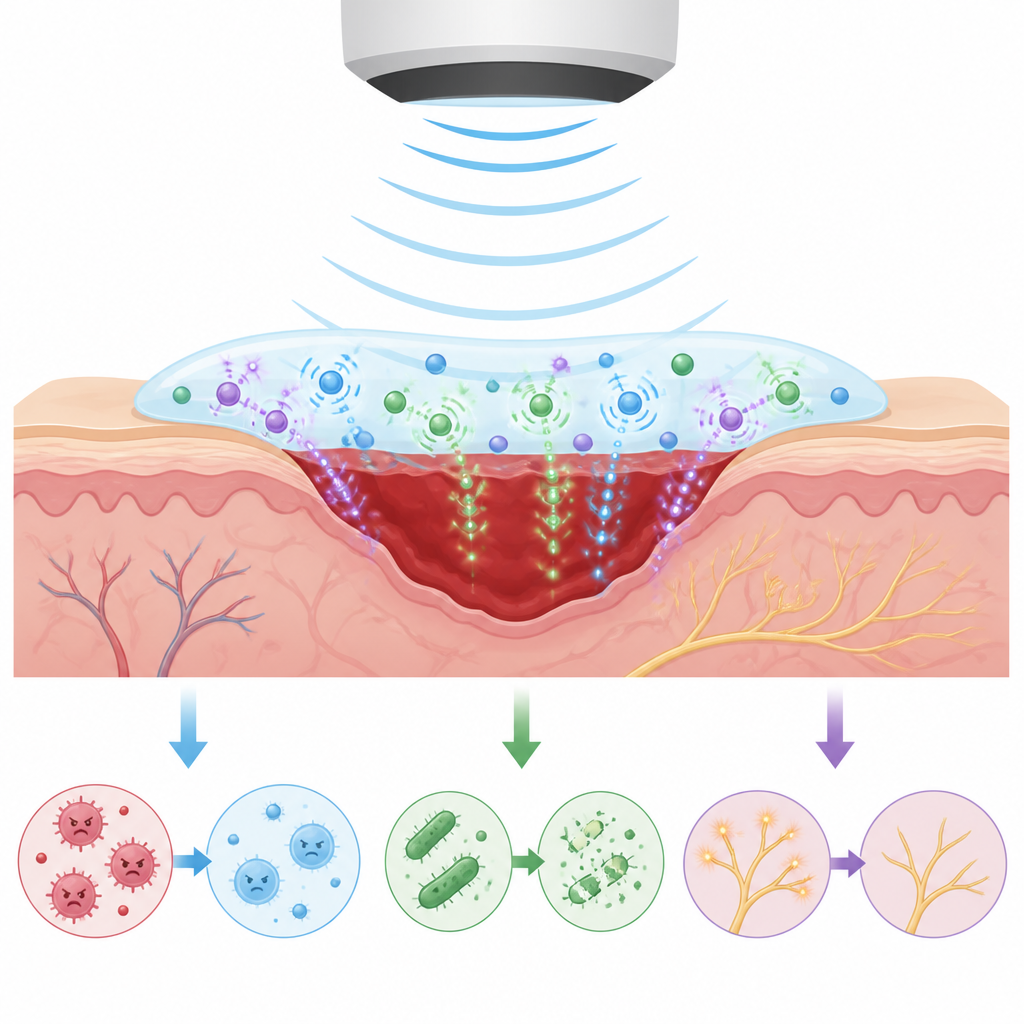
Speeding closure of clean and infected diabetic wounds
When tested on diabetic rats with large full-thickness skin defects, the ultrasound-triggered bandage led to faster wound closure than control treatments or even a commonly used growth-factor dressing. The treated wounds showed thicker new skin, denser and better organized collagen fibers, and many more new and mature blood vessels. In both clean and deliberately infected diabetic wounds, the bandage reduced bacterial counts, increased helpful anti-inflammatory macrophages, and lowered levels of inflammatory molecules while raising growth factors that support skin repair. Genetic analyses of wound tissue revealed that many genes tied to inflammation, tissue structure, and pain responses shifted from a chronic-injury pattern toward a healthier profile resembling normal skin.
Quieting misfiring nerves and tuning immune crosstalk
The team also asked whether the same bandage could ease neuropathic pain in diabetic rats. Animals with diabetic nerve damage usually react strongly to light touch and heat. With repeated ultrasound-bandage treatments, their pain thresholds rose and stayed higher, indicating lasting relief. Studies of sensory neurons showed that the tiny electrical currents help desensitize a key pain-sensing channel called TRPV1, reducing calcium influx and dampening nerve excitability, without relying on opioid or adrenergic pathways. At the same time, treated nerves released more of a signaling peptide known as CGRP, which in turn encouraged nearby macrophages to adopt a protective, anti-inflammatory state, forming a feedback loop between nerves and immune cells that supports both healing and pain relief.
What this work could mean for future care
To a non-specialist, the message is that this smart bandage uses gentle sound to switch on its own internal pharmacy and electrical stimulator right where it is needed. In diabetic animals, it helped close stubborn wounds, limited infection, calmed overactive pain nerves, and nudged the local immune system toward repair instead of chronic irritation. While much work remains before it reaches the clinic, this approach suggests that future dressings might not just cover a sore, but actively listen and respond to the body’s signals to guide both healing and comfort.
Citation: Li, X., Lin, L., Zhu, M. et al. Low-intensity focused ultrasound-activated piezoelectric gel bandage for diabetic wound repair and neuropathic pain relief. Nat Commun 17, 4243 (2026). https://doi.org/10.1038/s41467-026-70771-y
Keywords: diabetic foot ulcers, smart bandage, ultrasound therapy, neuropathic pain, wound healing