Clear Sky Science · en
Permeable nanoreactor eye drop for enzymatic cascade-mediated treatment for acute retinal injury model mimicking geographic atrophy
Why Protecting Aging Eyes Matters
As people live longer, more of us face vision loss from age-related macular degeneration, a common eye disease that slowly erodes the sharp central vision needed for reading, driving, and recognizing faces. Its most widespread form, called geographic atrophy, has no truly effective treatment yet. This study explores a new kind of eye drop built from tiny protective cages carrying natural antioxidant enzymes, aiming to shield fragile retinal cells from damage and offer a needle-free option for preserving sight.
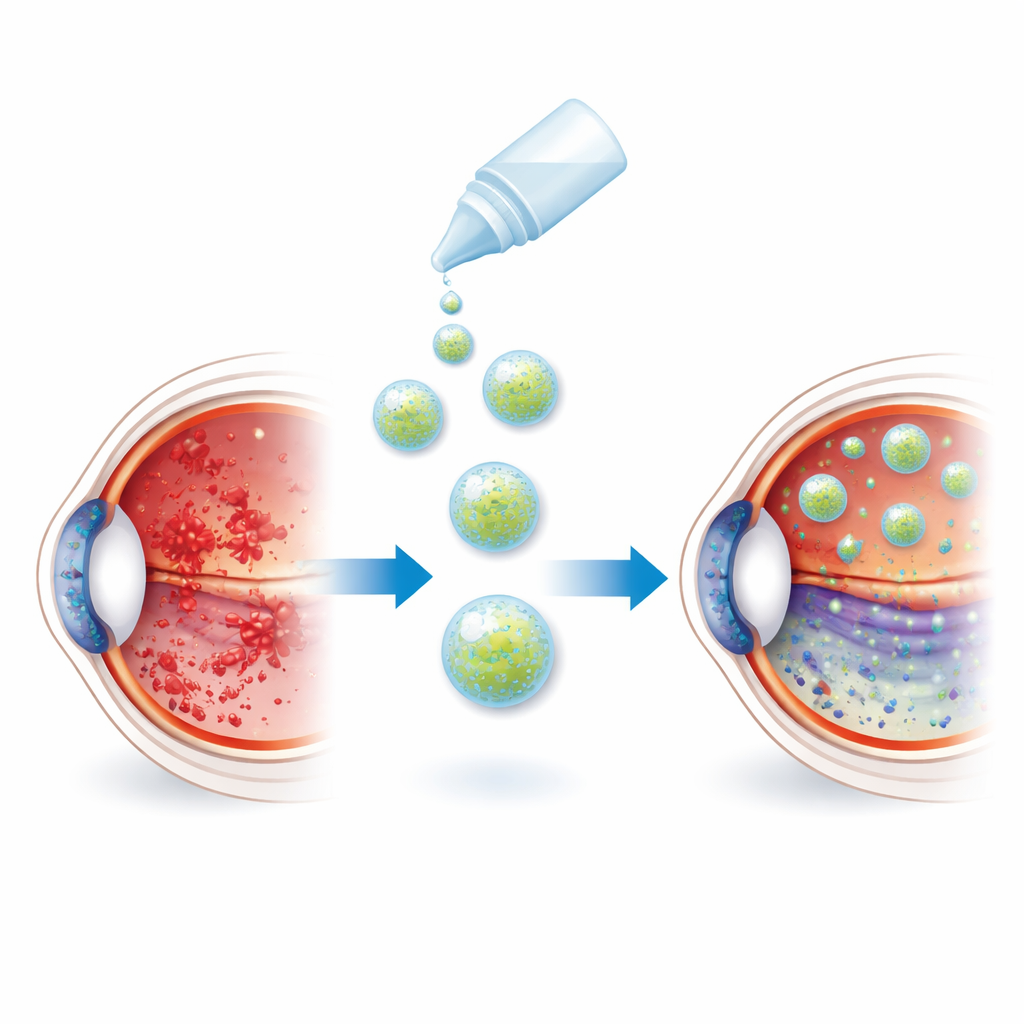
When the Retina Suffocates in Chemical Stress
The light-sensing layer at the back of the eye, the retina, is constantly bombarded by both light and oxygen. Over time this environment can generate high levels of reactive oxygen species, chemically aggressive molecules that damage fats, proteins, and DNA. In geographic atrophy, cells in the retinal pigment epithelium and the photoreceptors they support gradually die, leaving dead patches in the central retina and causing permanent blind spots. Existing drugs that block abnormal blood vessel growth help only in the “wet” form of the disease, not in this slowly thinning “dry” form. Today, patients are mostly limited to antioxidant supplements like lutein, which offer only modest slowing of vision loss.
Building a Fast Model of a Slow Disease
To probe what drives this degeneration and how to stop it, the researchers used a mouse model of acute retinal injury created by injecting sodium iodate, a chemical that selectively injures retinal pigment cells. Within two weeks, the animals developed many hallmarks of human geographic atrophy: drusen-like deposits under the retina, thinning of key cell layers, and a sharp drop in electrical responses that reflect visual function. The team measured a surge of reactive oxygen species in the retina, along with a marked fall in two key protective enzymes, superoxide dismutase and catalase, which normally work together to neutralize harmful molecules. This loss of antioxidant defenses coincided with increased cell death and activation of inflammatory pathways.
Turning Enzymes into Tiny Protective Reactors
Simply injecting these enzymes into the eye helped, but not enough. The proteins became trapped in the gel-like vitreous, struggled to cross inner retinal barriers, and were quickly broken down. To overcome these hurdles, the team wrapped the enzymes in “zwitterionic” nanocages—ultra-thin polymer shells that carry balanced positive and negative charges. These shells keep the enzymes’ structure intact, prevent sticky interactions in the vitreous, and encourage cells to take them up through specific endocytosis routes. The optimized nanocages, loaded with both enzymes, formed nearly neutral particles about 200 nanometers wide, with high loading efficiency and strongly improved stability against digestive enzymes and warm storage conditions.
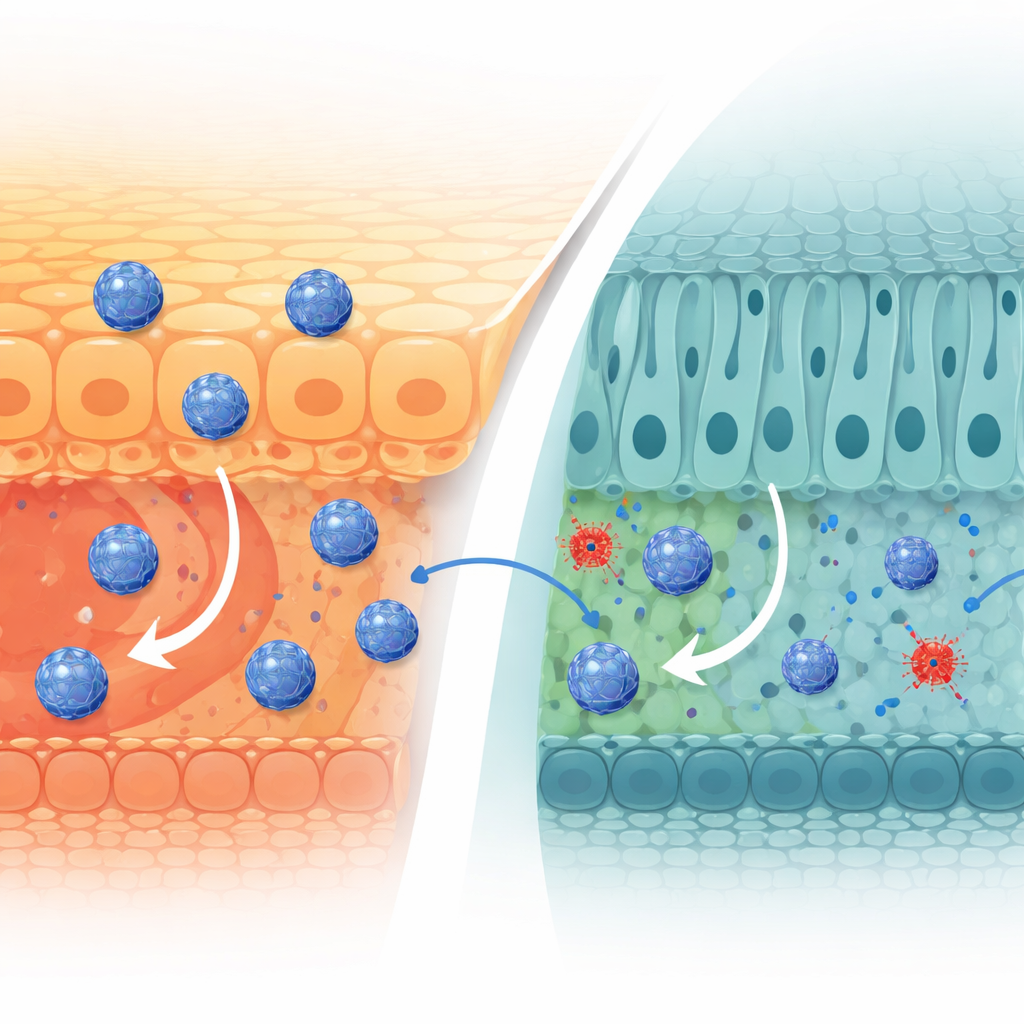
From Needle Injections to Potent Eye Drops
Injected directly into the vitreous, nanocage-packaged enzymes spread throughout the retina far more effectively than free enzymes. Mice treated with the dual-enzyme nanocages showed almost complete preservation of retinal thickness and structure, near-normal electrical responses, and greatly reduced cell death. Inflammatory markers and harmful immune cell types were tamped down, while more “healing” immune cells appeared. The researchers then reformulated the nanocages as simple eye drops. After topical application, these droplets achieved an eye penetration rate of about 9%, vastly higher than unprotected enzymes, reaching the retina through both tissue and blood routes. In the same injury model, a week of nanocage eye drops protected the retina about as well as a single injection into the eye and clearly outperformed both free enzymes and standard oral lutein.
What This Could Mean for Future Eye Care
For patients with or at risk of geographic atrophy, this work suggests that failure of the retina’s own antioxidant system may be a central driver of disease, and that restoring a powerful enzyme duo directly to the eye can meaningfully protect vision. By transforming fragile enzymes into stable, permeable nanoreactors, the study points to a noninvasive eye drop that can cross the eye’s natural barriers, quench damaging chemical stress, calm inflammation, and preserve retinal cells. While this approach still needs to be tested in long-term, human-like disease models and eventually in clinical trials, it offers a hopeful path toward simple, at-home treatments that could delay or prevent blindness in millions of older adults.
Citation: Shen, J., Zhao, H., Fang, Y. et al. Permeable nanoreactor eye drop for enzymatic cascade-mediated treatment for acute retinal injury model mimicking geographic atrophy. Nat Commun 17, 4092 (2026). https://doi.org/10.1038/s41467-026-70761-0
Keywords: age-related macular degeneration, geographic atrophy, nanoparticle eye drops, oxidative stress, retinal protection