Clear Sky Science · en
Turn-on luminescence from molecular rotor realignment in metal-organic framework thin films
Seeing Solvents Evaporate with Light
Many everyday products—from paints and coatings to fuels and medicines—quietly rely on how fast liquids evaporate. Yet, watching that evaporation at the tiny scales inside pores and channels is surprisingly hard. This study presents thin, crystalline films that suddenly glow up to 50 times brighter exactly when a solvent such as ethanol finishes evaporating inside them. This "light switch" behavior turns the films into sensitive visual indicators of solvent loss and reveals how tiny moving parts inside solids can be harnessed for sensing.
Tiny Moving Parts in Rigid Solids
We usually think of crystals as rigid and motionless, but chemists are learning how to build “molecular machines” into them. In this work, the researchers use metal–organic frameworks (MOFs), a class of highly porous materials made from metal ions and organic building blocks. The pores in these structures provide empty space where parts of the molecules can still twist and shift, even though the overall solid remains ordered. Here, the key moving parts are light-emitting units based on anthracene, a flat aromatic molecule, that behave like tiny rotors. In solution these rotors shine brightly, but when packed too closely together in a solid, they typically quench each other’s light.
Simple Films with Hidden Order
To turn these rotors into useful sensors, the team needed highly ordered, wafer-like coatings. They created thin MOF films on heated substrates simply by drop-casting an ethanolic solution of zinc ions, anthracene-based linkers (ADC), and pillar molecules (DABCO or BPy). As the solvent dries, the components self-assemble into a “pillared-layer” architecture: flat sheets of zinc–anthracene units stacked parallel to the surface and connected by vertical pillars. X-ray scattering measurements show that most of the tiny crystals line up in the same orientation, forming a well-ordered layer across different types of substrates such as silicon, quartz, and oxide-coated wafers. This simple, scalable process avoids the complex step-by-step growth usually required for oriented MOF films.
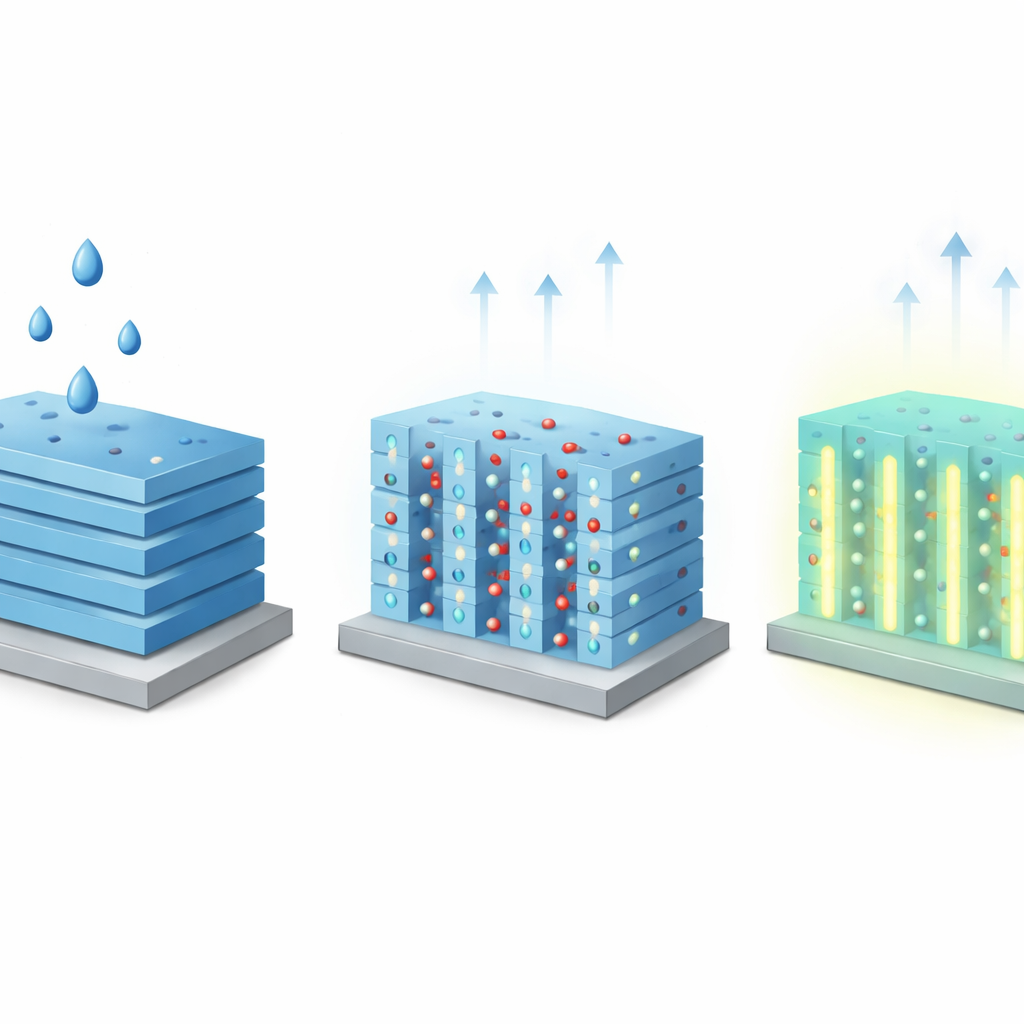
When Evaporation Makes Crystals Glow
On their own, these films emit only a faint blue light when excited by ultraviolet radiation, far dimmer than the same molecules in liquid. The surprise comes when a small droplet of ethanol is placed on the film. As the liquid initially covers and penetrates the pores, the brightness rises modestly. But in the final moments of drying—during a brief “transient” stage—the emission suddenly surges, reaching a quantum yield comparable to that of the free molecules in solution, and then fades again once the film is fully dry. This cycle can be repeated many times without damaging the film. The effect depends strongly on the solvent: ethanol and the smaller methanol both trigger the turn-on glow, while the bulkier isopropanol does not, indicating that the liquid must be able to enter and move through the narrow MOF channels.
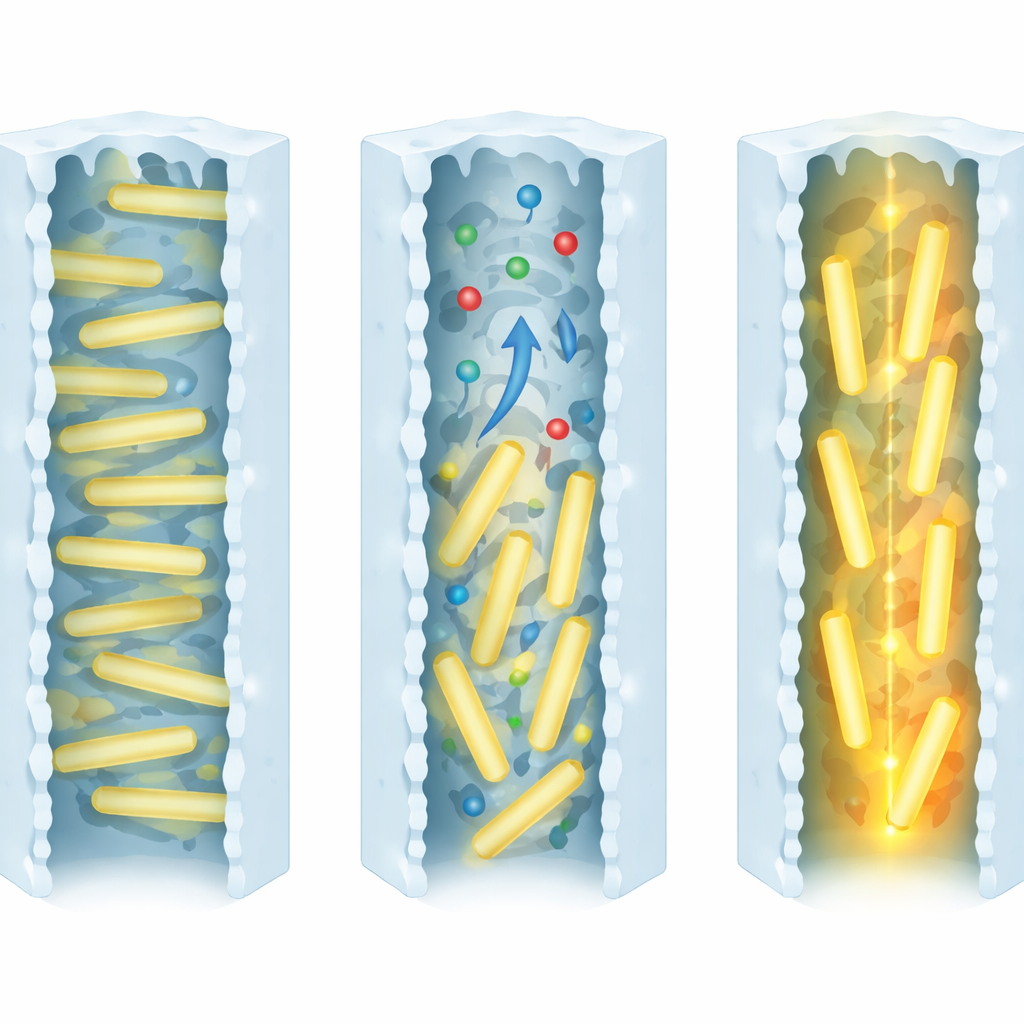
Stress, Rotation, and Bright Light
To understand this behavior, the researchers monitored both light emission and mechanical changes in the film as solvent came and went. A quartz crystal microbalance revealed that liquid ethanol causes a gentle, reversible distortion of the MOF, while time-resolved optical measurements showed that the strong brightening coincides with the very last stage of evaporation. Reflection experiments at ultraviolet wavelengths suggested that the light-absorbing anthracene units reorient collectively during this moment. Detailed computer simulations support a picture in which departing solvent molecules drag on the anthracene rotors, tilting them from a more flat configuration toward a more upright one inside the pores. This partial realignment reduces how strongly neighboring units quench each other and makes it easier for electronic excitations to move along aligned pathways, both of which boost the emitted light.
Turning Glow into a Practical Signal
To showcase a simple application, the team coated commercial molecular sieve beads with the MOF and placed them on a methanol droplet. Under ultraviolet light, a bright ring of emission appeared where the moving front of absorbed and evaporating liquid passed through the bead, then slowly migrated and shrank to a glowing spot at the top before eventually disappearing. This eye-visible signal directly maps where evaporation is happening inside the porous solid. The findings demonstrate that carefully designed molecular motion inside a crystal can serve as a built-in reporter of mechanical stress and flow at the nanoscale. In practical terms, such films and coatings could act as sensitive, reversible indicators for solvent drying or fluid transport in tiny channels, with potential uses in chemical processing, safety monitoring, and advanced microdevices.
Citation: Fischer, J.C., Zhou, T., Sievers, P. et al. Turn-on luminescence from molecular rotor realignment in metal-organic framework thin films. Nat Commun 17, 3969 (2026). https://doi.org/10.1038/s41467-026-70551-8
Keywords: metal-organic frameworks, luminescent sensors, solvent evaporation, molecular rotors, thin films