Clear Sky Science · en
Significant stability enhancement in photocatalytic CO2 reduction via flow-driven strategies
Turning Greenhouse Gas into Useful Fuel
Imagine if the carbon dioxide that warms our planet could instead be turned into useful fuels, the way plants do during photosynthesis. Scientists have been trying to copy that trick with light-driven devices, but most of these artificial “leaves” burn out within hours. This paper explores a surprisingly simple idea—keeping carbon dioxide and water flowing past the catalyst—that allows these systems to keep working not just for hours, but for days and even weeks.
Why Artificial Photosynthesis Burns Out
Many research groups are racing to develop photocatalysts, materials that use light to convert carbon dioxide and water into energy-rich molecules like carbon monoxide and methane. In principle, such systems could both reduce greenhouse gases and provide renewable fuels. In practice, though, most catalysts lose more than 80% of their activity within a few hours. The surfaces of these materials get clogged with reaction leftovers, or the material itself starts to corrode and rearrange, much like a metal tool rusting over time. Earlier efforts focused mainly on redesigning the materials themselves, which often required complex syntheses and still did not achieve the long lifetimes needed for practical use.
Keeping Things Moving to Keep Them Working
Instead of inventing ever more complicated materials, the authors look at the problem from the point of view of the reactor—the “box” that holds the catalyst, gas, and water. They compare a traditional, closed system, where carbon dioxide and water just sit over the catalyst, to a new three-phase setup where gas and liquid are constantly flowing across a thin catalyst layer. In the stagnant case, products and reaction intermediates build up near the surface, driving the reaction toward a standstill and encouraging unwanted side reactions. In the flowing case, fresh carbon dioxide and water are continually supplied while products are swept away, much like a stream that stays clear because the water never stands still. Measurements show that this design boosts the rate at which carbon dioxide reaches the surface by about 15 times compared with a closed batch reactor. 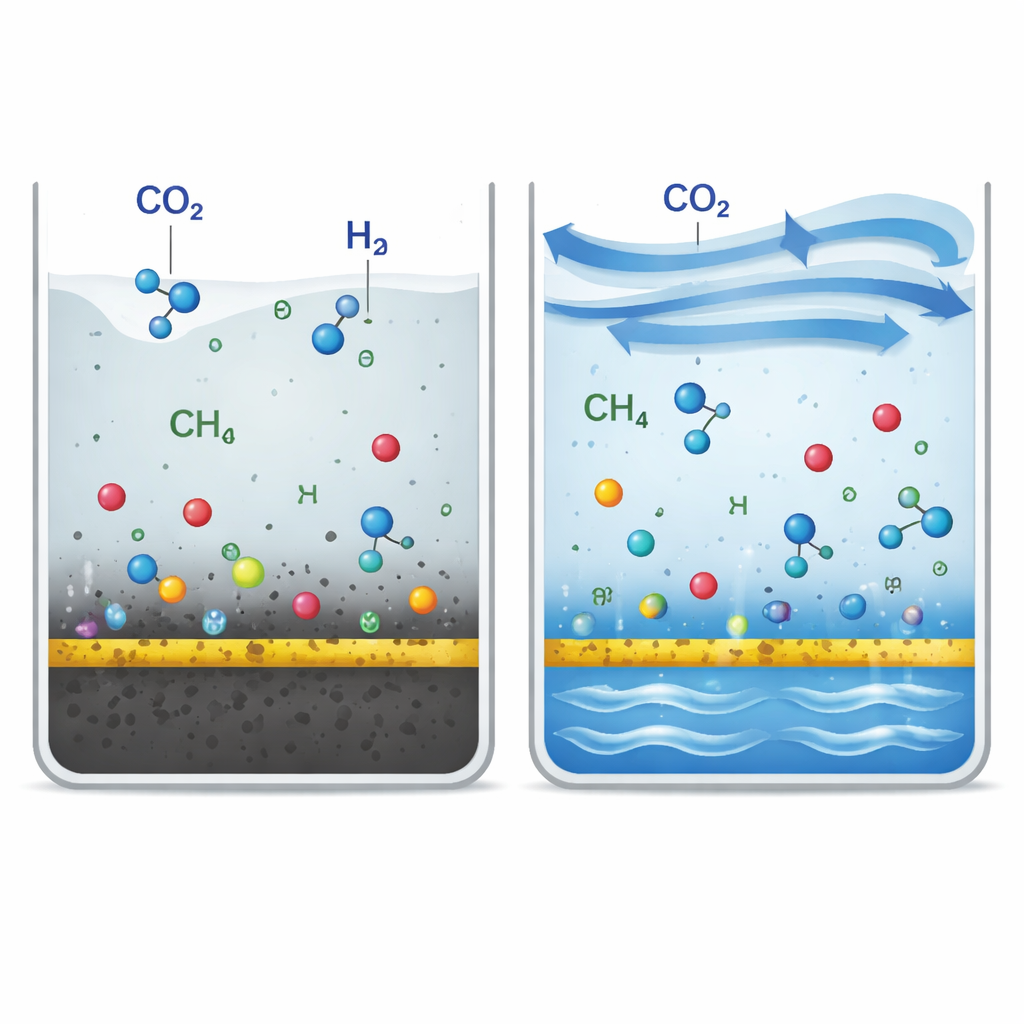
A Simple Design that Works Across Many Materials
The team tests their flow-based approach on several widely used photocatalysts, including titanium dioxide (TiO2), zinc oxide (ZnO), cadmium sulfide (CdS), and a nitrogen-rich carbon material called C3N4. Under usual, non-flowing conditions, all of these materials lose most of their activity within 1 to 10 hours. Under continuous flow, however, the picture changes dramatically. Titanium dioxide, for example, keeps more than 80% of its initial performance for over 15 days of continuous operation, producing carbon monoxide steadily over that period. Other materials also last far longer, though those that are intrinsically more fragile, like CdS and C3N4, still eventually degrade because of their own chemical weaknesses. This shows that good reactor design cannot fix every problem, but it can greatly extend the usable lifetime of even fairly simple photocatalysts.
How Flow Protects the Catalyst Surface
To understand why the flowing system is so much more durable, the researchers closely examine the catalyst surfaces after long operation. In the non-flowing reactors, the catalyst becomes visibly discolored, and detailed surface analyses reveal a heavy buildup of carbon-based deposits—tangled chains and rings formed from reaction byproducts. These deposits behave like grime on a cooking pan, blocking the spots where the reaction should take place. In the flow-enabled reactors, by contrast, the surface chemistry remains close to that of the fresh material, with only minor carbon accumulation. The authors also show that if they deliberately let carbon build up and then clean it away with a mild acid wash, the catalyst’s activity can be almost fully restored, underscoring that surface fouling, not permanent damage, is the main culprit. 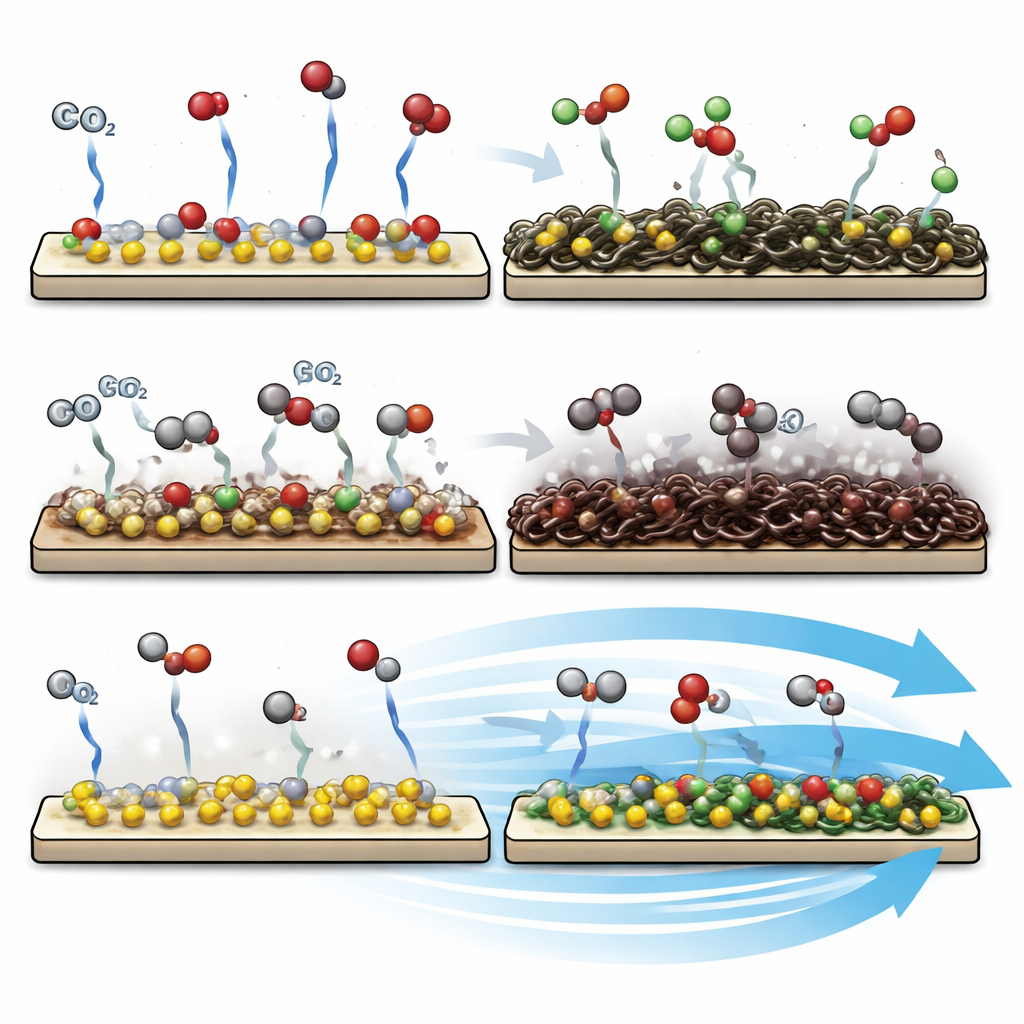
Stability All the Way Down to the Atoms
The team goes further by probing what happens inside the catalyst particles using powerful X-ray techniques at a synchrotron facility. For titanium dioxide, they see that the basic atomic structure stays largely intact in both flowing and stagnant systems, though the stagnant case shows small local distortions that match the idea of stress from accumulated surface species. To test a more sensitive material, they repeat the measurements with copper oxide, which more readily changes its internal structure under reaction conditions. Under continuous flow, the copper oxide preserves its bonding patterns over hours of operation, while in the non-flowing setup those patterns weaken and become disordered as performance drops. These atomic-scale observations align with the performance data: when products and intermediates are constantly removed, the catalyst remains both chemically and structurally healthier.
Learning from Nature’s Use of Flow
In closing, the authors argue that nature has long since solved this stability problem by keeping water and carbon dioxide moving in plants—through transpiration, wind, and the intricate plumbing of leaves. By mimicking this constant motion in artificial systems, we can turn relatively simple photocatalysts into long-lived “artificial leaves” that convert carbon dioxide to fuels for days or weeks. The main message for non-specialists is that durability in artificial photosynthesis does not just depend on designing exotic materials; it also depends critically on keeping the reaction environment in motion so that the catalyst surface stays clean and active.
Citation: Jung, H., Jeon, H.S., Kim, M.G. et al. Significant stability enhancement in photocatalytic CO2 reduction via flow-driven strategies. Nat Commun 17, 4139 (2026). https://doi.org/10.1038/s41467-026-70542-9
Keywords: photocatalytic CO2 reduction, artificial photosynthesis, continuous flow reactors, solar fuel production, catalyst stability