Clear Sky Science · en
Dynamic hydroxyl cycle of zeolite for short and ultra-short chain PFAS free potable water
Why Cleaner Tap Water Matters
Many communities are learning that their tap water contains “forever chemicals” called PFAS—industrial compounds that barely break down in nature and may harm human health even at extremely low levels. While older, long-chain PFAS are being phased out, their shorter cousins are now more common in drinking water and are harder to remove with standard filters. This study describes a simple, low-cost way to retool existing water purifiers using a common mineral called zeolite and ordinary water vapor, aiming to strip even the smallest PFAS from household drinking water.
The Hidden Threat in Everyday Water
PFAS have been used for decades in products like nonstick pans, stain-resistant fabrics, and firefighting foams, so they now contaminate water sources worldwide. Regulators in Europe have set strict limits on PFAS in drinking water because these chemicals can build up in our bodies and are linked to a range of health concerns. Long-chain PFAS have drawn the most attention so far, but short and ultra-short chain PFAS—molecules with only a few carbon atoms—are increasingly replacing them. These smaller molecules are more water-loving and slip past conventional treatment methods such as coagulation or advanced oxidation, and even many home filters struggle to catch them reliably.
Turning a Common Mineral into a Smart Filter
The researchers focused on zeolites, porous minerals already used in various purification systems. Traditional zeolite filters attract the oily tails of PFAS but do not interact well with their water-loving heads, especially for the smaller molecules. Instead of permanently attaching fragile chemical groups to the zeolite surface (which can degrade during regeneration), the team introduced a “dynamic hydroxyl cycle” that uses only water and heat. In this cycle, spent zeolite is first heated to high temperature, stripping off water-related groups and breaking down trapped PFAS. Then, exposing the hot, clean zeolite to very humid air allows water molecules to diffuse into its tiny pores and self-assemble into confined clusters, temporarily creating new hydrophilic sites. This reversible transformation equips the zeolite to grab both ends of short PFAS molecules more effectively.
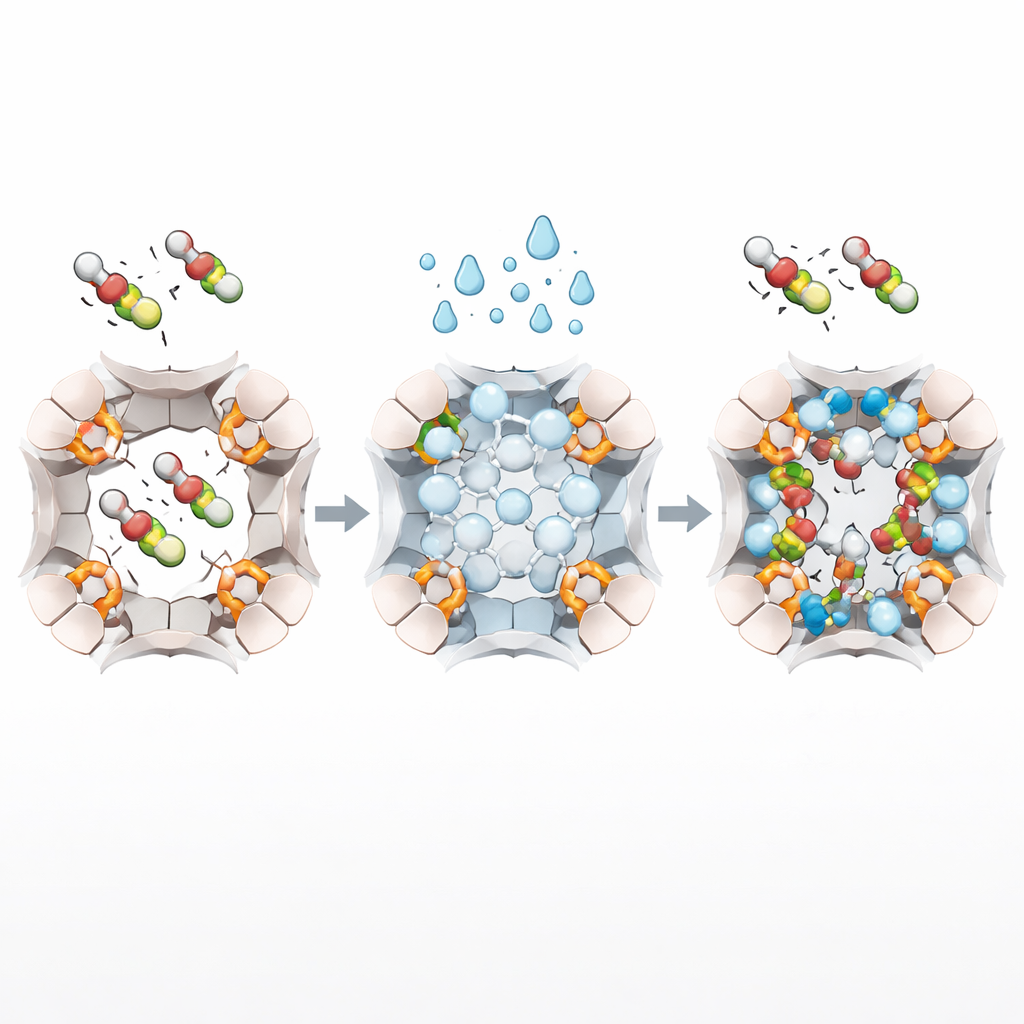
How Confined Water Does the Heavy Lifting
Detailed experiments and computer simulations reveal what happens inside the zeolite’s nanometer-scale channels during this cycle. When water vapor is introduced, some of it becomes locked inside the pores as structured clusters and chains, while also regenerating hydroxyl (–OH) groups on the mineral framework. These confined water clusters act like tiny sticky hubs that strongly interact with the charged, water-loving heads of PFAS. At the same time, the fluorinated tails of the molecules nestle against the hydrophobic walls of the zeolite channels. This “dual-binding” pattern—water clusters gripping one end and the mineral backbone holding the other—smooths the energy barrier for PFAS to move from bulk water into the pores, leading to much higher loading of even the shortest PFAS compared with untreated zeolite or many advanced adsorbents.
A Reusable Filter That Breaks the PFAS Cycle
Because the confined water is not chemically bonded, the filter can be regenerated simply by heating, which removes the water clusters and destroys the accumulated PFAS without damaging the zeolite structure. The team showed that a specific form of zeolite, known as β200, can be cycled through this construct–use–remove sequence multiple times with little loss of performance. Across a range of short and ultra-short PFAS with different structures, the modified zeolite achieved some of the highest adsorption capacities reported—hundreds of milligrams of PFAS per gram of solid—while maintaining stability in water that also contained salts and natural organic matter similar to real environmental conditions.
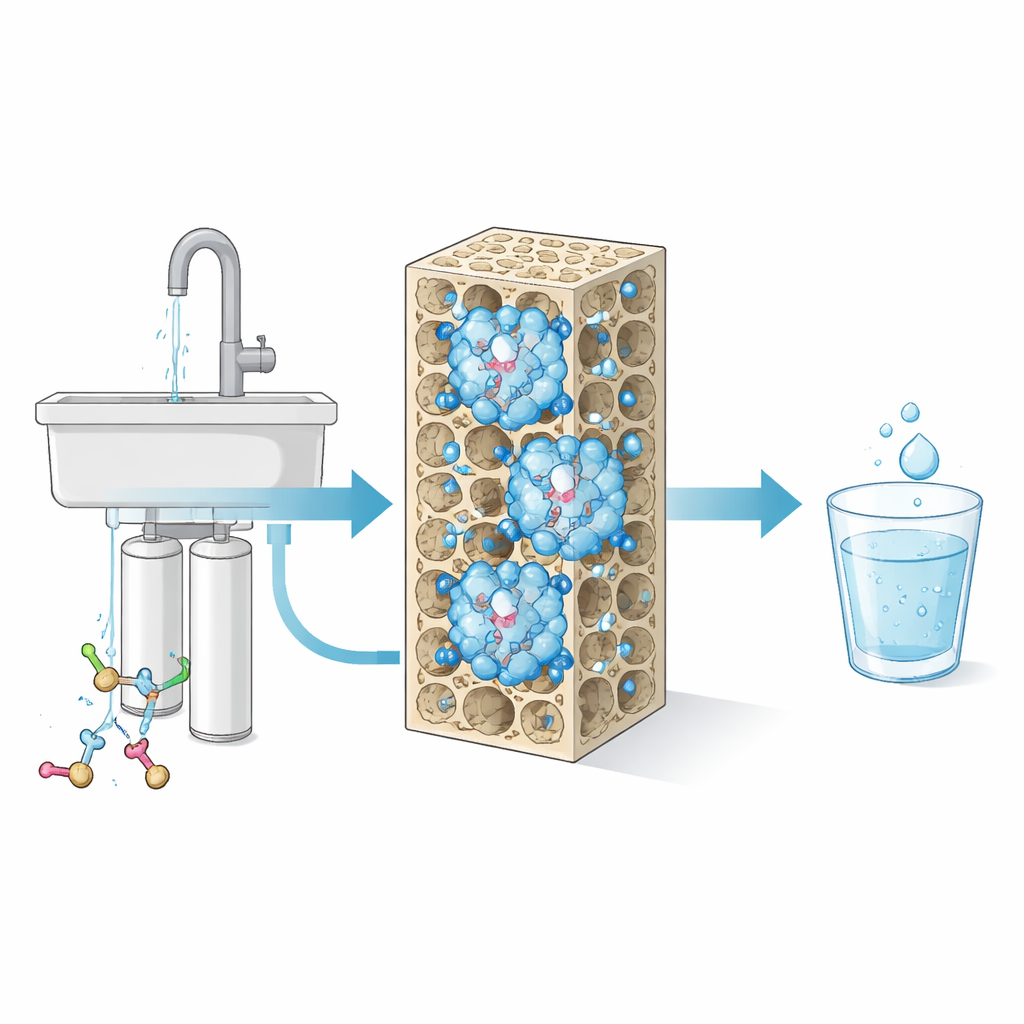
From Lab Concept to Everyday Kitchen
To test whether this approach could work at real-world scale, the researchers replaced the final activated carbon stage in a commercial three-step household purifier with a cartridge packed with the modified zeolite. In a six‑month simulation of family water use, this dynamic hydroxyl purifier removed 73–95% of short and ultra-short PFAS, outperforming the original commercial unit and approaching the levels achieved by far more expensive reverse osmosis systems. Importantly, the new filter did not compromise removal of other common contaminants such as organic matter and heavy metals. Because the only extra “reagent” needed is water, and regeneration uses heat similar to existing industrial practices, the method offers a practical way to upgrade current devices.
What This Means for Safer, Affordable Water
In simple terms, the study shows how to turn a widely available mineral into a kind of smart sponge that can latch onto the tiniest, most elusive PFAS molecules using just water and heat. By cycling the zeolite through dry and humid conditions, it repeatedly builds and erases a network of microscopic water clusters that help pull PFAS from drinking water and then let them be burned away during regeneration. This balance of strong capture and easy reuse could lower the cost of providing PFAS-safe tap water, particularly in regions where advanced technologies like reverse osmosis remain out of reach.
Citation: Shi, Y., Yang, M., Mu, H. et al. Dynamic hydroxyl cycle of zeolite for short and ultra-short chain PFAS free potable water. Nat Commun 17, 3749 (2026). https://doi.org/10.1038/s41467-026-70507-y
Keywords: PFAS, drinking water, zeolite filters, water purification, adsorption