Clear Sky Science · en
Homoharringtonine exhibits senotherapeutic activity that mitigates diet- and age-associated obesity and insulin resistance and extends lifespan in mice
Why an Obesity Drug Story Matters for Aging
Many people struggle with weight gain, high blood sugar, and the health problems that follow as we grow older. This study explores whether an existing cancer drug, homoharringtonine (HHT), can be repurposed to target worn-out “zombie” cells in body fat. By clearing these damaged cells, the researchers show in mice that it may be possible to ease obesity-related diabetes and even extend lifespan, offering a glimpse of future medicines that tackle aging itself rather than just its symptoms.
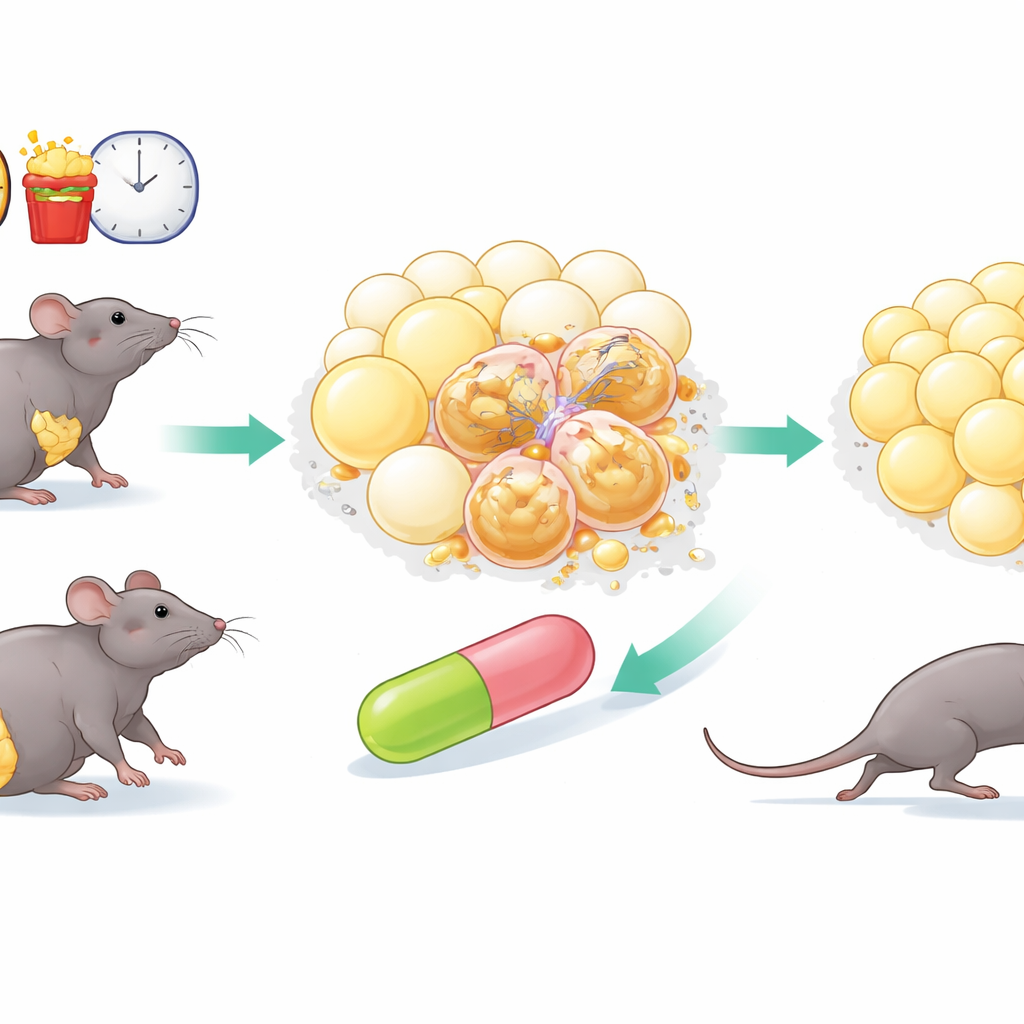
How Aging Fat Turns Against the Body
Fat tissue is more than a passive storage depot; it constantly adjusts to changes in diet and temperature, releases hormones, and helps regulate whole-body metabolism. With age and overnutrition, however, this flexibility erodes. Certain cells in fat enter a state called senescence: they stop dividing, swell in size, and begin secreting a cocktail of inflammatory molecules and tissue-remodeling enzymes. These senescent cells accumulate in white fat, stiffen the tissue with scar-like fibers, attract immune cells, and push neighboring cells toward dysfunction. The result is larger, less healthy fat cells, chronic inflammation, and a higher risk of problems like fatty liver and type 2 diabetes.
Finding a New Use for an Old Drug
To search for drugs that might selectively disarm or remove senescent cells, the team screened 2,150 medicines already in clinical use or trials. They first tested these compounds on human cells driven into senescence in the lab and looked for those that either killed senescent cells outright or dampened their harmful secretions, while sparing healthy cells. From an initial set of 110 hits, they narrowed the list to 15 promising “senotherapeutic” candidates. In obese mice fed a high-fat diet, one drug clearly stood out: homoharringtonine. Originally developed from a plant alkaloid and approved for certain leukemias, HHT reduced weight gain and improved how the animals handled a surge of glucose, without changing how much they ate.
Cleansing Fat Tissue and Restoring Its Flexibility
Digging deeper, the researchers found that HHT-treated obese mice had smaller fat depots, lower fasting blood sugar, and better insulin sensitivity. Microscopic and molecular analyses showed that fat tissue from treated animals contained far fewer senescent cells and less inflammatory scarring. Structures where dead or dying fat cells are ringed by immune cells were reduced, and the balance shifted from oversized fat cells toward many smaller ones, a pattern linked to healthier metabolism. Single-nucleus RNA sequencing revealed that HHT increased the number of mature fat cells with a “healthy” hormone profile and reduced gene signatures associated with aging in adipocytes. It also improved the behavior of fat precursor cells, restoring their ability to form new fat cells and cold-responsive “beige” cells, which help burn energy. In clamp studies that precisely measure sugar handling, treated mice showed greater glucose uptake into fat and less fat buildup in the liver.
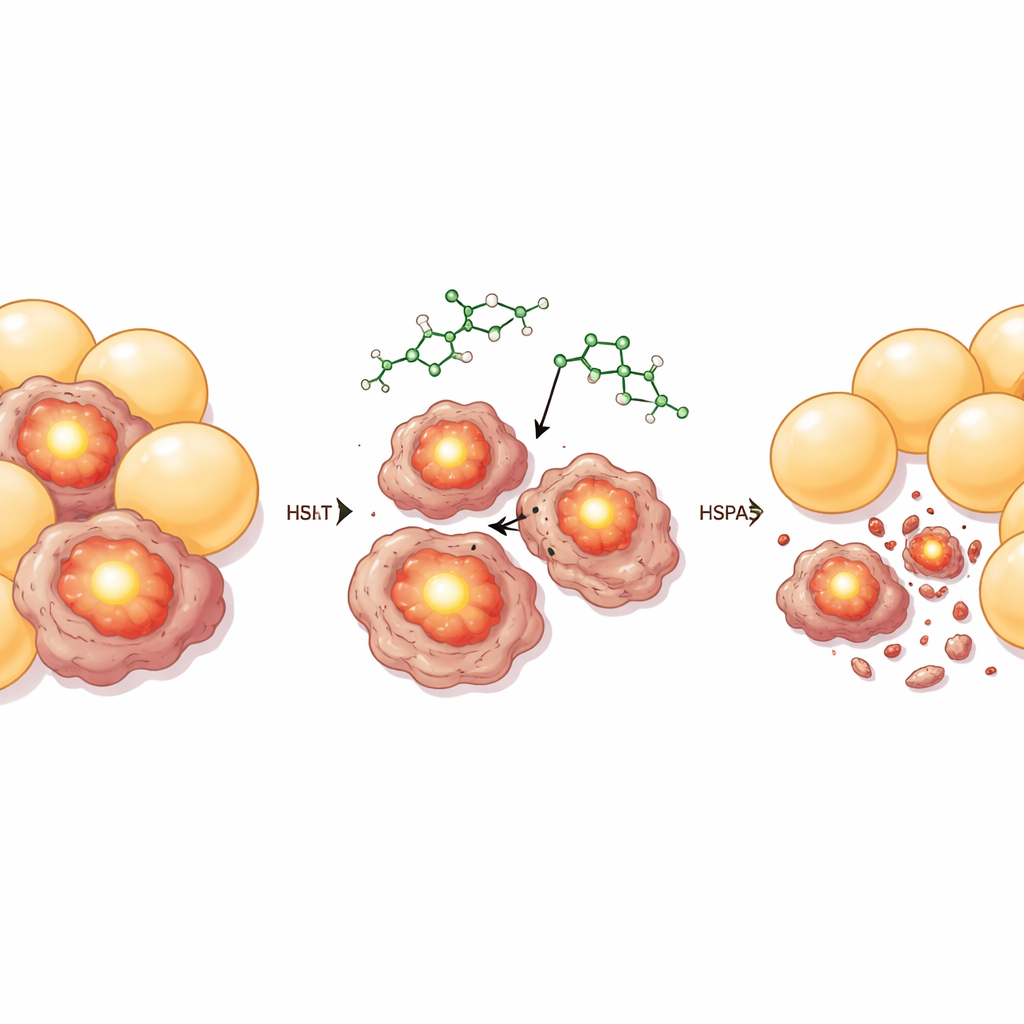
Uncovering the Drug’s Target and Testing Longevity
At the molecular level, HHT’s senescent-cell selectivity traced back to a chaperone protein called HSPA5, which is abundant in stressed and aging fat cells. Using a protein-stability assay, mass spectrometry, and biophysical measurements, the team showed that HHT binds directly to HSPA5 and inhibits its activity. Senescent cells with high HSPA5 levels were especially prone to HHT-induced cell death, whereas knocking down HSPA5 blunted this effect and overexpressing it made cells more sensitive. In obese and aged mice, HHT treatment lowered HSPA5 levels in fat tissue, consistent with the removal of HSPA5-rich senescent cells. The researchers then asked whether this cellular cleanup translated into broader health benefits. In both a rapid-aging mouse model and naturally aged mice, HHT improved grip strength, organ function, and markers of tissue aging, reduced fibrosis in several organs, and significantly extended median lifespan, all without clear signs of liver, kidney, or blood toxicity at the doses used.
What This Could Mean for Healthy Aging
To a non-specialist, the core message is that a drug already used in cancer clinics can, in mice, act like a targeted cleanup crew for damaged fat cells that accumulate with age and overfeeding. By homing in on a stress protein that is overactive in these “zombie” cells, homoharringtonine helps restore healthier fat tissue, improves blood sugar control, and even lengthens life in multiple mouse models. While many questions remain—especially about safety, dosing, and effects in humans—this work strengthens the idea that removing senescent cells could become a powerful strategy to combat obesity-related diseases and promote healthier aging.
Citation: Kim, EC., Jung, HB., Park, Yk. et al. Homoharringtonine exhibits senotherapeutic activity that mitigates diet- and age-associated obesity and insulin resistance and extends lifespan in mice. Nat Commun 17, 2700 (2026). https://doi.org/10.1038/s41467-026-70475-3
Keywords: cellular senescence, adipose tissue, insulin resistance, drug repurposing, healthy aging