Clear Sky Science · en
Biological valorization of methane and nitrogen gas-derived ammonia via methanotrophic bacteria for gut-beneficial nutrients
Turning Waste Gases into Helpful Nutrition
Methane is often viewed as a troublesome greenhouse gas leaking from mines, landfills, and farms, while nitrogen from the air is usually turned into fertilizer using energy-hungry factories. This study shows that both gases can be captured and fed to specialized bacteria that make a nutritious protein-rich powder. Even more intriguingly, this powder, when eaten, can help protect the gut and ease signs of bowel inflammation in mice. The work links climate-friendly gas recycling with animal health, suggesting a future in which wasted gases are transformed into smart feed ingredients.
From Stray Methane to Useful Feed
Across China and many other countries, large amounts of methane escape from fossil fuel extraction and waste treatment sites. Because it is scattered and costly to pipe, this gas is often simply burned, wasting its carbon content. At the same time, animal producers rely heavily on imported soybean and fishmeal to supply protein in feed. The authors argue that methanotrophic bacteria—microbes that naturally eat methane—offer a way to address both problems at once. Grown in tanks, these microbes can convert methane into “single-cell protein,” a dry biomass that can be blended into animal feed, potentially easing pressure on cropland and fisheries while making use of local gas sources.
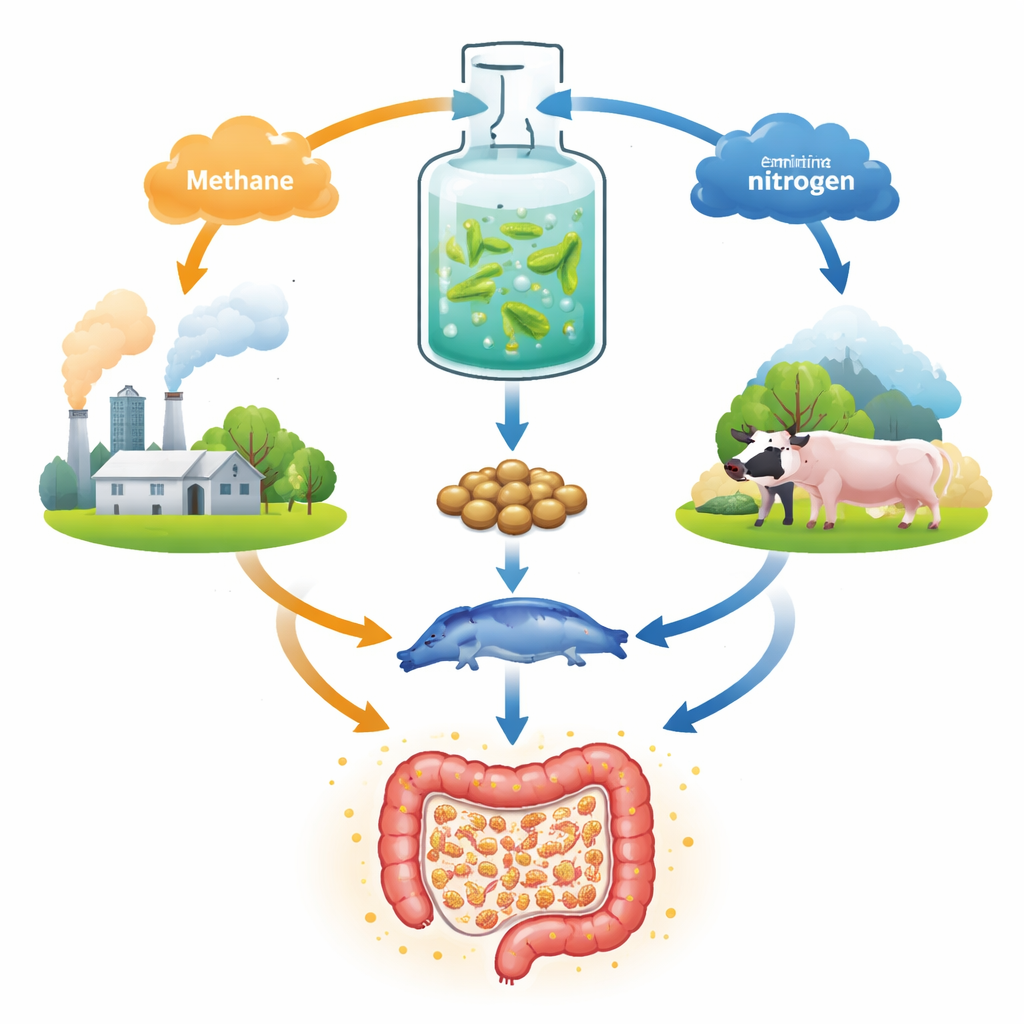
Feeding Bacteria with Air-Derived Ammonia
To grow well, these methane-eating bacteria also need nitrogen, one of the main building blocks of protein. Instead of using nitrate salts, which are more expensive, the team focused on ammonia made from nitrogen gas through an emerging clean process that uses light and electricity. Oxygen from standard air separation can simultaneously supply the bacteria’s need for breathing. Linking these chemical steps with fermentation could create a compact, local system: air is split into nitrogen and oxygen, nitrogen is turned into ammonia, and together with methane, all three gases are fed to bacteria that churn out protein close to farms that need it.
Making Ammonia Safe for the Microbes
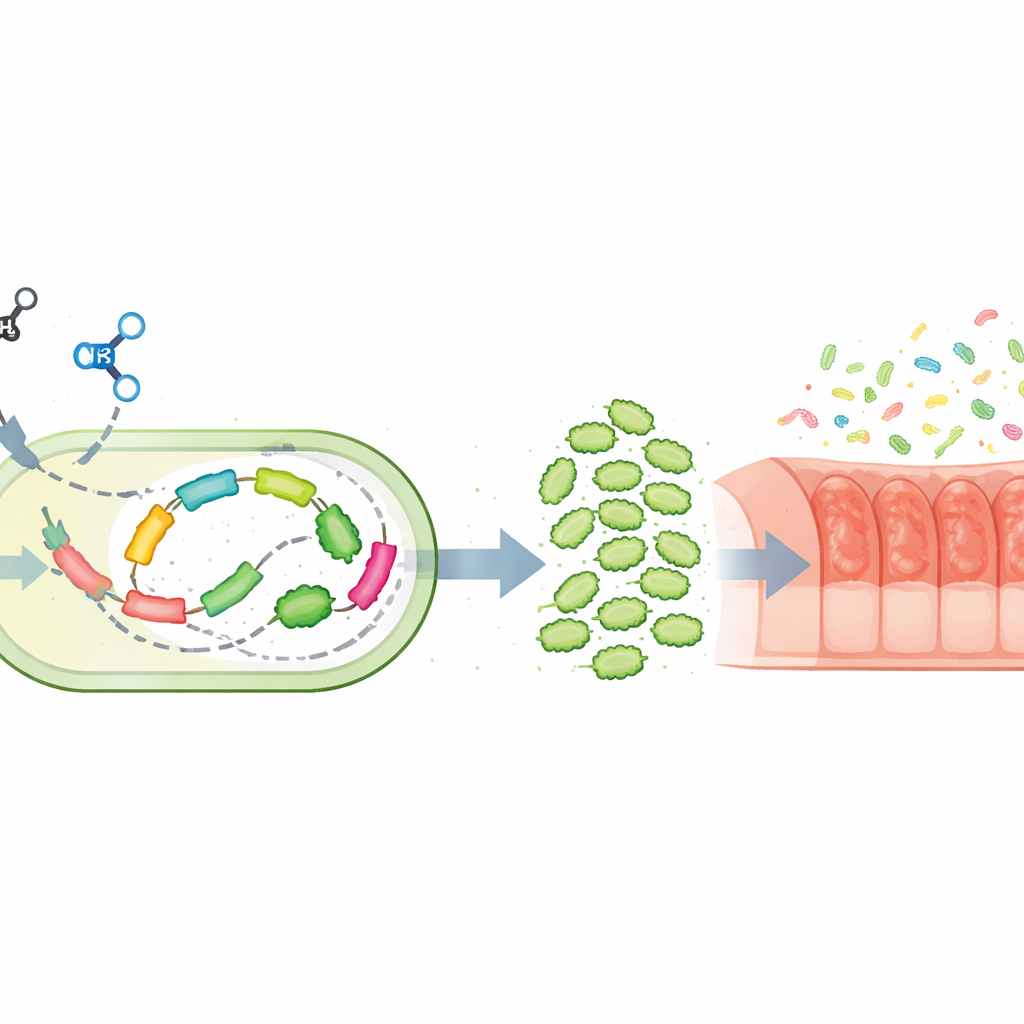
A key obstacle is that ammonia, while a favorite nitrogen source for most microbes, is partly mis-processed by methane-eating bacteria. Their methane-oxidizing enzyme also acts on ammonia and creates hydroxylamine, a toxic intermediate that slows growth. Using whole-genome transcript analysis, the researchers mapped how the bacterium Methylotuvimicrobium sanxanigenens responds when grown on ammonia instead of nitrate. They found strong activation of genes involved in handling hydroxylamine and confirmed that both this compound and its downstream product, nitrite, build up under ammonia feeding. Guided by these insights, they engineered the cells to overproduce an enzyme called hydroxylamine reductase, which directly converts the toxic intermediate back into harmless ammonia inside the cell, reducing its accumulation and easing stress.
Boosting Production of Nutrient-Rich Cell Protein
Genetic tuning alone was not enough; the amount and timing of ammonia supply also mattered. The authors developed a fed-batch strategy in which ammonia is added gradually and kept below a toxicity threshold. In a 3‑liter fermenter, this engineered strain reached much higher cell densities and an 18-fold increase in protein productivity compared with earlier ammonia-based cultures. Careful measurements showed that the resulting “methanotrophic cell protein” contains high levels of essential amino acids and substantial polysaccharides, with a nutritional profile comparable to fishmeal, egg, and milk. A fraction of small peptides with potential anti-inflammatory activity was also detected, and the biomass passed safety checks for endotoxins, supporting its use as a feed ingredient.
Protecting the Gut and Calming Inflammation
To test whether this microbial protein offers benefits beyond basic nutrition, the team fed it to mice with chemically induced colitis, a widely used model of inflammatory bowel disease. Mice receiving the protein lost less weight, had lower disease scores, and showed longer, healthier colons on examination. Tissue slices revealed fewer ulcers and less structural damage, while a key barrier-forming protein in the intestinal lining, Occludin, rebounded. Levels of inflammatory signals in the colon fell, and anti-inflammatory signals rose. At the same time, the mix of gut microbes shifted away from harmful species toward communities that produce short-chain fatty acids and other beneficial compounds. Certain metabolites linked to reduced inflammation and better energy metabolism became more abundant, and even healthy mice showed strengthened gut barriers and more favorable microbiota when given the protein.
A Gas-to-Gut Pipeline for a Greener Future
In essence, this work creates a bridge between climate and health: methane and nitrogen from the air are turned into a functional feed ingredient that nourishes animals and helps maintain a resilient gut. By solving the toxicity hurdle that once limited ammonia use, the authors show that methane-fed bacteria can be grown efficiently and economically on air-derived ammonia. If scaled, this approach could reduce waste methane emissions, cut reliance on conventional protein feeds, and lessen the need for antibiotics and other drugs in animal production by supporting gut health from the inside out.
Citation: Gao, Z., Liu, Y., Jiao, S. et al. Biological valorization of methane and nitrogen gas-derived ammonia via methanotrophic bacteria for gut-beneficial nutrients. Nat Commun 17, 3803 (2026). https://doi.org/10.1038/s41467-026-70448-6
Keywords: methanotrophic bacteria, single-cell protein, methane upcycling, gut microbiome, functional feed additive