Clear Sky Science · en
Conducting polymer-stabilized nanozymes alleviate sepsis-induced myocardial injury by inhibiting iron accumulation and lipid peroxidation
Why protecting the septic heart matters
Sepsis, a runaway response to infection, is one of the world’s leading killers. Beyond overwhelming the immune system, it can suddenly weaken the heart, a condition called septic cardiomyopathy. In this state, tiny chemical storms inside heart cells damage their power plants and membranes, and there are few targeted treatments. This study explores a new type of engineered nanoparticle, called a nanozyme, designed to calm these storms, mop up excess iron, and protect the heart during sepsis.
How sepsis injures the heart
During sepsis, the body releases floods of reactive molecules known as free radicals. These include reactive oxygen and nitrogen species that attack fats, proteins, and DNA. Heart muscle cells are especially vulnerable because their mitochondria work nonstop to supply energy. When overwhelmed by radicals, mitochondria malfunction, energy production drops, and cells can die. A particularly harmful form of cell death, ferroptosis, is driven by iron build-up and the breakdown of fatty components in cell membranes, further weakening the heart’s pumping function.
Designing smart antioxidant particles
To counter this damage, the researchers created a library of tiny catalyst-like particles, or nanozymes, built on a conductive polymer framework made from polypyrrole and polythiophene. They coordinated eleven different metal ions within this framework and screened the resulting particles for their ability to safely break down harmful oxidants. A ruthenium-based version stood out and was named Ruzyme. It mimics key natural defense enzymes, breaking down hydrogen peroxide into water and oxygen, converting particularly aggressive radicals into milder forms, and neutralizing nitrogen-based radicals, all while avoiding unwanted over-oxidation.
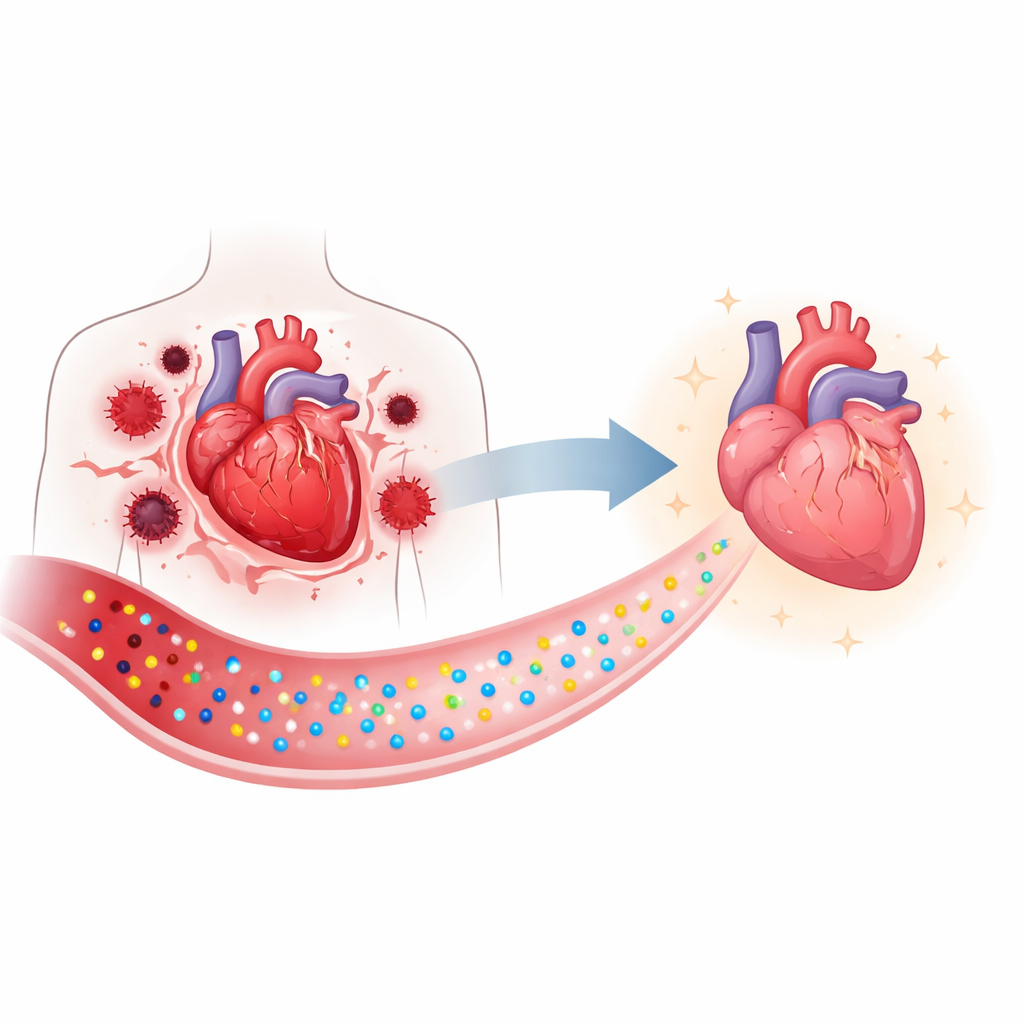
Targeting the heart’s power plants
Beyond simply working as a potent antioxidant, Ruzyme was engineered to go exactly where it is needed. The team attached a mitochondrial homing group, a positively charged triphenyl phosphonium unit, so the particles are drawn to the negatively charged interior of mitochondria. They then added a short peptide that recognizes injured heart tissue, creating a dual-targeted version called CICT-Ruzyme. These modifications also help keep the particles well dispersed in bodily fluids, improving their stability and electrical conductivity, which in turn supports faster and more efficient catalytic reactions.
Stopping iron-driven cell death
In cell experiments, these nanozymes reduced radical levels, prevented iron from accumulating, and lowered markers of lipid peroxidation, all hallmarks of suppressed ferroptosis. The particles chelated, or grabbed onto, free iron with high efficiency, depriving damaging reactions of their metal fuel. They also preserved the activity of GPX4, a natural enzyme that protects membranes from oxidative breakdown, and dampened signals from genes linked to ferroptosis. When another chemical trigger of ferroptosis was used, nanozyme treatment kept most heart cells alive and reduced toxic by-products of lipid damage.
Benefits in septic mice
In mice given bacterial toxins to mimic sepsis, the targeted nanozymes homed to diseased heart tissue and accumulated in mitochondria. Treated animals showed better heart pumping measures on echocardiography, lower blood levels of enzymes that signal heart injury, and healthier heart tissue under the microscope. Levels of reactive oxygen and nitrogen species, iron deposits, and lipid peroxidation markers all fell. Importantly, the particles showed good safety: they remained stable, released negligible metal into surrounding fluids, did not harm major organs, and were well tolerated at therapeutic doses.
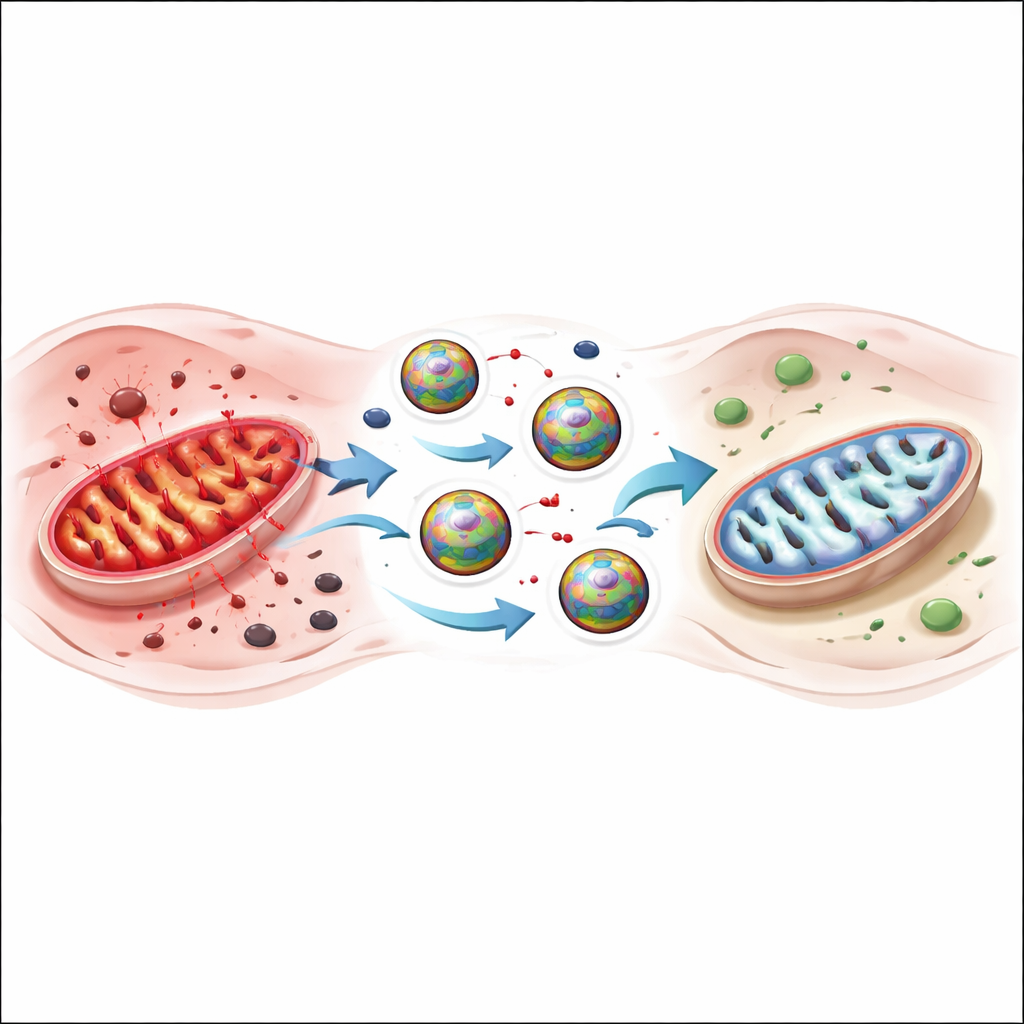
What this could mean for future care
This work suggests that carefully designed nanozymes can act like compact, programmable antioxidant systems that go straight to the heart’s power plants during sepsis. By combining radical scavenging with iron removal, and by stabilizing their own catalytic behavior through a conductive polymer shell, these particles interrupt a vicious cycle of oxidative stress and ferroptosis. While much testing remains before use in people, the approach points toward a new generation of targeted treatments for severe heart complications in sepsis and possibly other heart diseases driven by oxidative and iron-related damage.
Citation: Wu, T., Liu, Y., Wang, W. et al. Conducting polymer-stabilized nanozymes alleviate sepsis-induced myocardial injury by inhibiting iron accumulation and lipid peroxidation. Nat Commun 17, 3874 (2026). https://doi.org/10.1038/s41467-026-70385-4
Keywords: septic cardiomyopathy, nanozymes, oxidative stress, ferroptosis, mitochondrial protection