Clear Sky Science · en
Elucidating genetic backgrounds of myasthenia gravis in Japanese by genome-wide association studies and multi-omics analyses of thymoma
Why this research matters
Myasthenia gravis is a chronic disease that causes muscle weakness, drooping eyelids, and trouble chewing, speaking, or even breathing. Many patients never achieve long-lasting remission, and doctors still struggle to predict who will get which form of the disease or how well treatments will work. This study focuses on Japanese patients and uses large-scale genetic and tissue analyses to uncover hidden risk factors, with the goal of improving diagnosis, prognosis, and eventually treatment.
Looking for clues in DNA
The researchers began by scanning the genomes of 1,434 Japanese people with myasthenia gravis and more than 42,000 unaffected individuals. Using a technique called a genome-wide association study, they searched millions of genetic markers for subtle differences between patients and controls. One stretch of DNA near a gene called TERT clearly stood out. People carrying certain TERT variants were more likely to develop myasthenia gravis, especially the generalized form that affects many muscles and the type linked to tumors of the thymus gland, called thymomas. The study also confirmed that genes in the major immune region of the genome, known as HLA, play an important role, but in different ways depending on the age at which the disease begins.
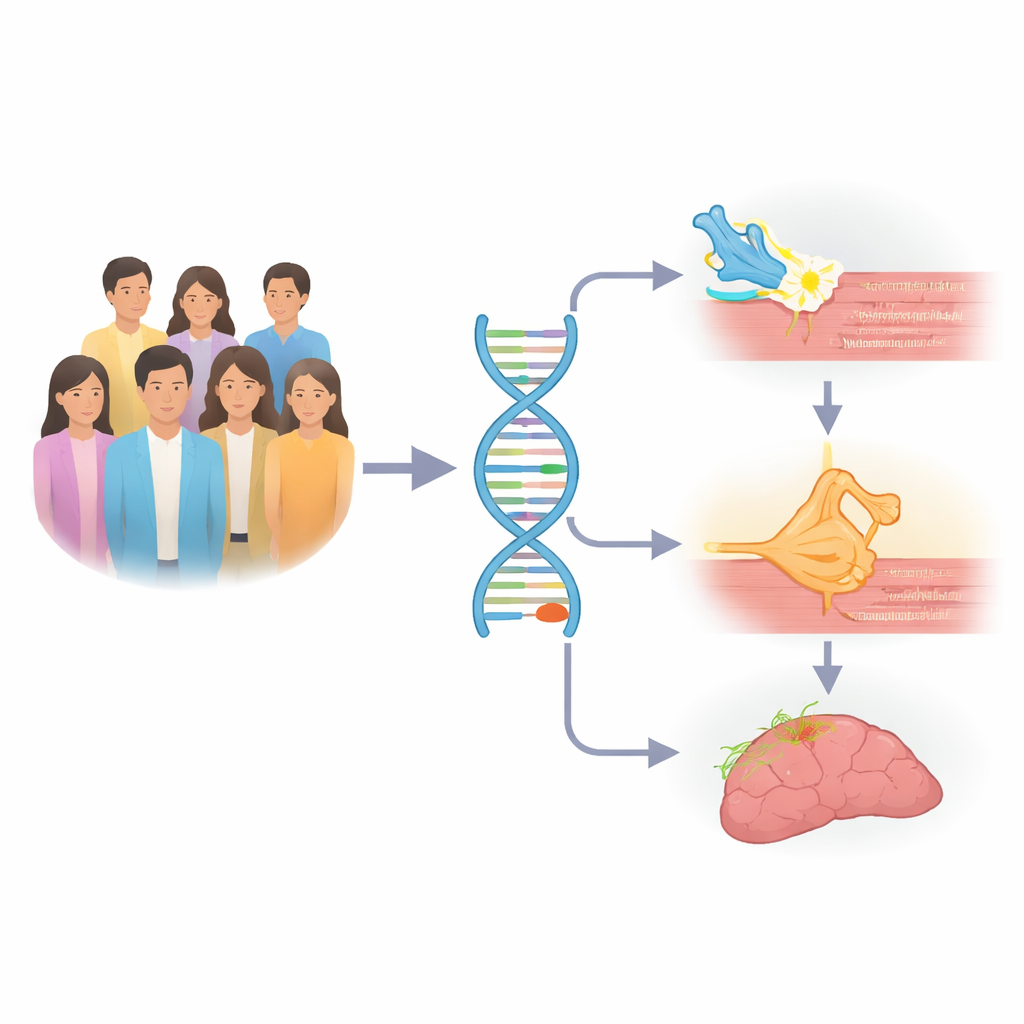
Different gene patterns for different patients
Myasthenia gravis is not a single uniform illness. Some patients have weakness only in the eye muscles, some develop widespread symptoms, some carry specific antibodies, and some have a thymoma. When the team re-analyzed the genetic data by these subtypes, the TERT signal became even stronger in patients with generalized disease, those with anti–acetylcholine receptor antibodies, and those with thymoma. Meanwhile, early-onset and late-onset cases without thymoma showed distinct HLA patterns: one HLA gene variant was more important in younger patients, another in older ones. These findings suggest that what looks like one disease in the clinic actually arises from partly different genetic paths.
Genes that shape both disease and treatment success
The scientists then asked whether the TERT variant that raises disease risk also influences how well patients respond to therapy. Using long-term follow-up data, they found that carriers of the risk version of the TERT marker were more likely to respond poorly to treatment, particularly among patients with early-onset generalized disease and those with the common antibody-positive subtype. In other words, the same piece of DNA that nudges someone toward myasthenia gravis may also make their illness harder to control. By analyzing data from large Japanese biobanks, the team further showed that this variant is tied to several other conditions, including increased risk of lung cancer, changes in blood cell counts, and shorter telomeres—the protective caps on chromosome ends that TERT helps maintain.
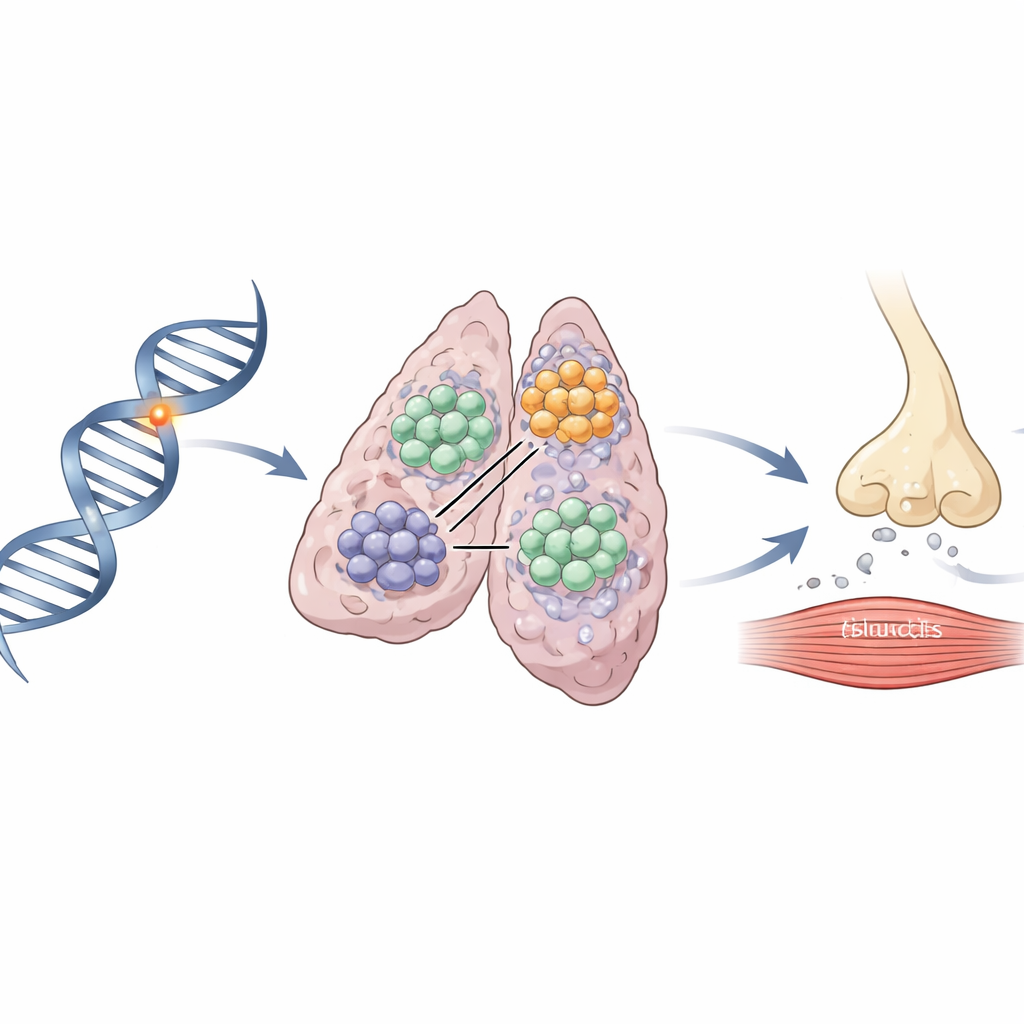
Zooming into the thymus
Because TERT is active mainly in a few tissues, including the thymus where immune cells mature, the researchers examined actual thymoma samples from patients. Single-cell RNA analysis and microscopic staining revealed that TERT is switched on specifically in immature lymphocytes inside these tumors, but not in the tumor’s epithelial cells. Using a specialized full-length RNA sequencing method, they measured how the risk variant affects TERT production in these cells. The risk version of the variant reduced TERT expression in an allele-specific manner, likely by weakening the binding of regulatory proteins that normally boost the gene’s activity. This altered TERT activity in key immune cells may in turn disturb how the immune system learns to distinguish self from non-self, setting the stage for an attack on the neuromuscular junction.
What this means for patients
Taken together, the work shows that specific genetic variants in TERT and HLA help shape who develops myasthenia gravis in Japan, which subtype they get, and how well they respond to treatment. It also links a single TERT variant to a wide range of traits, from blood measures to cancer risk, underscoring how one genetic change can ripple through the body. In the long run, such insights could enable more personalized care—using genetic information to identify high-risk patients, tailor monitoring for complications like thymoma or lung cancer, and choose therapies more likely to bring meaningful, lasting relief.
Citation: Ueda, H., Kubota, T., Goto, R. et al. Elucidating genetic backgrounds of myasthenia gravis in Japanese by genome-wide association studies and multi-omics analyses of thymoma. Nat Commun 17, 3830 (2026). https://doi.org/10.1038/s41467-026-70376-5
Keywords: myasthenia gravis, genetic risk, TERT, thymoma, HLA