Clear Sky Science · en
Self-regulation of Lewis acid sites on FeOCl toward piezo-self-Fenton reaction for continuous hydroxyl radicals generation
Turning Everyday Vibrations into Clean Water
From the hum of traffic to the buzz of industrial machinery, our world is full of wasted mechanical energy. This study shows how such motion can be harnessed to clean water polluted with hard‑to‑remove drugs and chemicals. By designing a special material that responds to vibrations, the researchers demonstrate a way to continuously generate powerful cleaning agents inside water itself, without adding extra chemicals. The work points to compact, low‑waste treatment systems that could help tackle pharmaceutical pollution and other persistent contaminants.
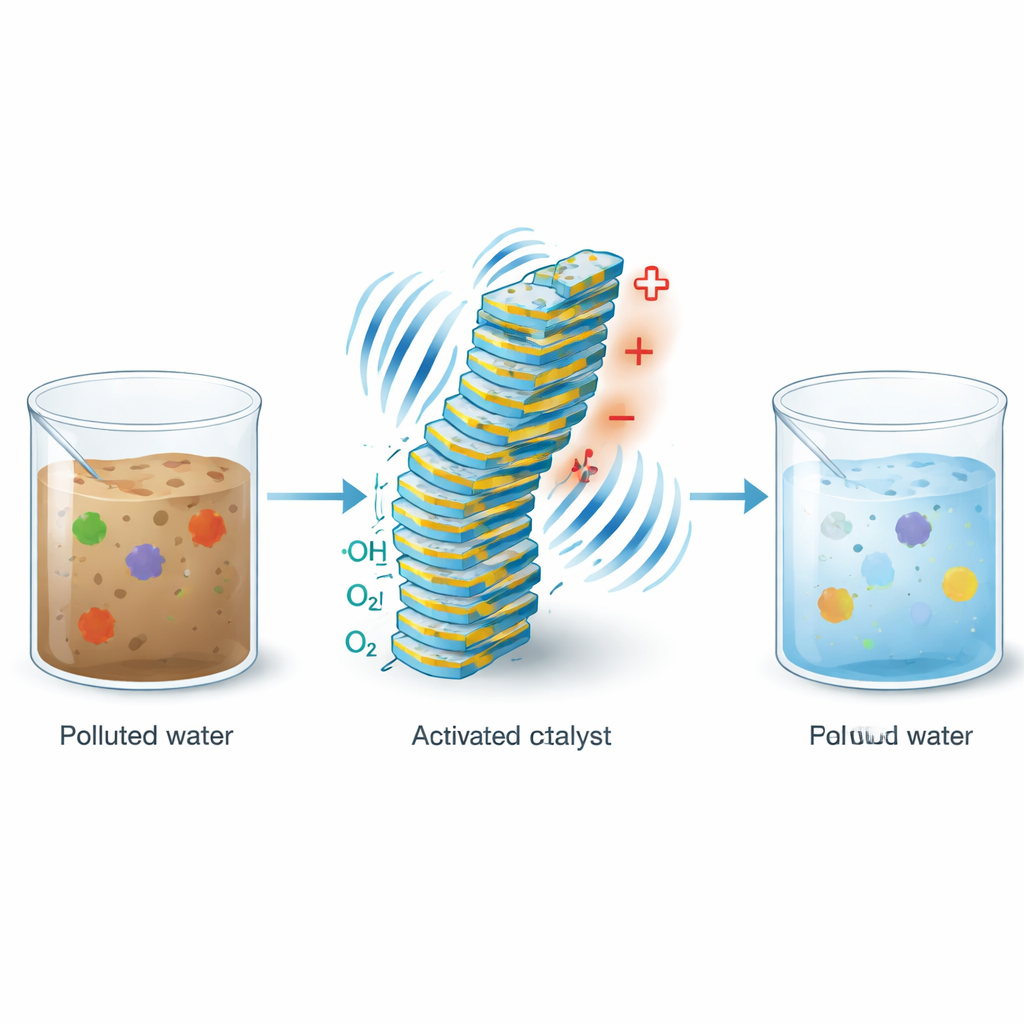
A Material That Wakes Up When It Is Shaken
The heart of the study is a layered compound called iron oxychloride, or FeOCl. It belongs to a class of materials that generate small electrical potentials when they are bent, squeezed, or vibrated – an effect known as piezoelectricity. When FeOCl particles in water are exposed to ultrasound, they flex and develop tiny surface charges. These charges help move electrons and holes around the surface, which is already known to boost a classic water‑cleaning chemistry called the Fenton reaction. In that reaction, iron helps convert hydrogen peroxide into highly reactive hydroxyl radicals that can tear apart organic pollutants. Until now, however, this process usually required adding hydrogen peroxide from outside.
Surface Sites That Reorganize Themselves
The team discovered that mechanical shaking does more than just shuffle electrons. It also reconfigures the atomic neighborhoods on the surface of FeOCl. Certain iron atoms act as so‑called "Lewis acid" sites – places that strongly attract electron‑rich molecules. Using probe molecules and spectroscopy, the researchers showed that when ultrasound is applied, these sites become both more numerous and more strongly attractive, yet the overall crystal structure and iron oxidation state barely change. In other words, the material does not break down or permanently transform; instead, the applied stress subtly adjusts its electronic structure, temporarily sharpening its surface reactivity. Similar behavior was found in other piezoelectric materials, suggesting a general strategy for tuning catalysts in real time.
Making Strong Oxidants from Ordinary Oxygen
Because the activated surface sites are better at grabbing electron‑rich species, they can now latch onto dissolved oxygen and step it through a chain of reactions. Under vibration, FeOCl converts oxygen first into hydrogen peroxide and then into hydroxyl radicals, all within the same material. Careful experiments and computer simulations traced this pathway: oxygen sticks more strongly to the stressed surface, accepts electrons more easily, and passes through several intermediate forms before finally releasing aggressive radicals right at the solid–liquid boundary. These radicals are extremely short‑lived, so making them at the catalyst surface increases the odds that they immediately attack nearby pollutants instead of fading away harmlessly in bulk water.
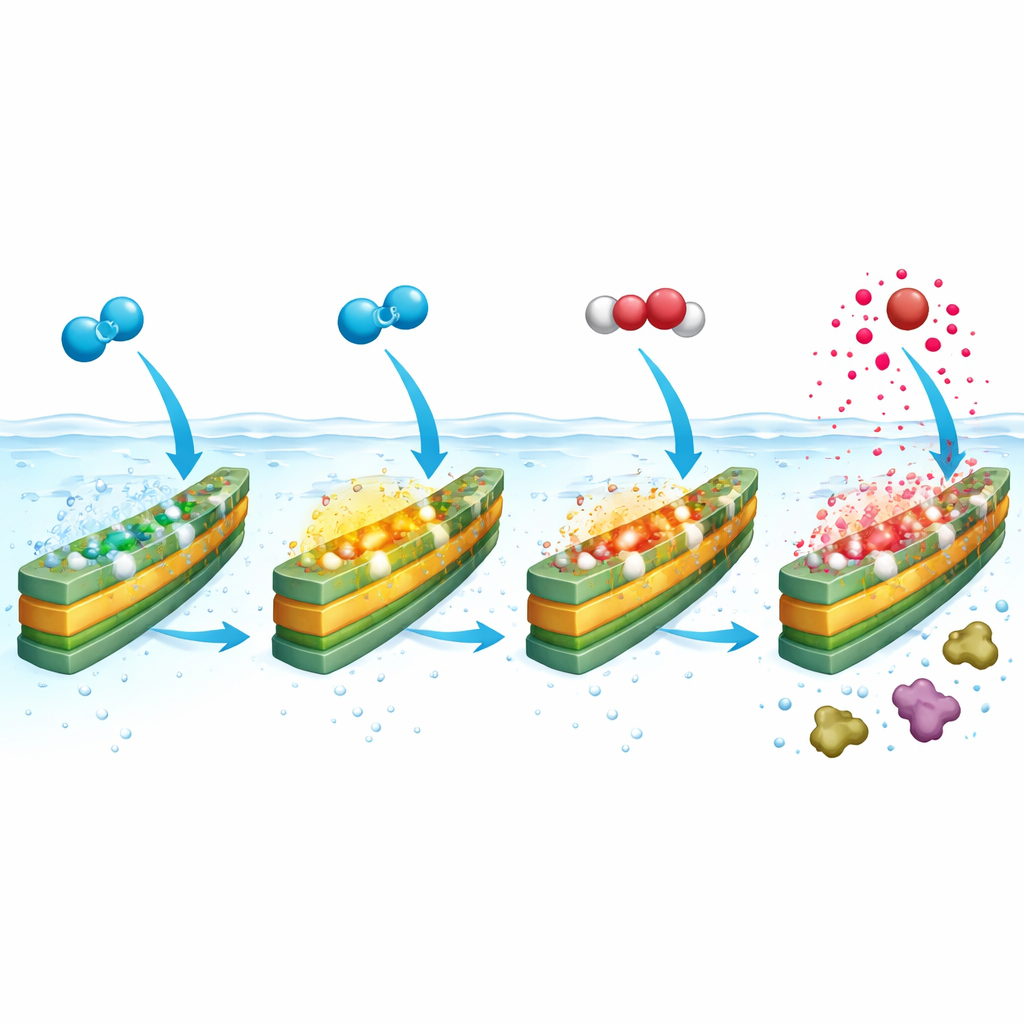
A Self‑Driving Cleaning Loop for Tough Pollutants
By merging oxygen activation and radical formation in one vibrating material, the authors create what they call a "piezo‑self‑Fenton" system. It no longer needs bottled hydrogen peroxide or added iron ions; oxygen from air and mechanical energy from ultrasound are enough. In tests with the antibiotic sulfamethazine, the new system removed about 99% of the pollutant in an hour, matching or surpassing many conventional Fenton setups. It also broke down a wide range of other drug molecules and phenols, kept working over repeated cycles, tolerated common salts, and performed well over a broad pH range, including near‑neutral water. A life‑cycle analysis suggested that, for treating the same amount of pollutant, this approach could have lower impacts on resources, toxicity, and greenhouse emissions than standard chemical Fenton treatments.
From Lab Beakers to Flowing Wastewater
To move beyond small‑scale tests, the researchers loaded FeOCl onto granular activated carbon and packed it into a fixed‑bed column, through which they pumped real pharmaceutical wastewater while applying ultrasound. Over many hours of operation, color and total organic carbon both dropped substantially, and fluorescence measurements showed that complex organic residues were being removed. These proof‑of‑concept trials highlight how mechanically driven, self‑regulating catalysts could be built into compact flow systems powered by inexpensive vibration sources such as pumps, flowing water, or industrial equipment.
What This Means for Future Water Treatment
In everyday terms, the study shows how a smart material can "turn itself up" when shaken, using that motion to both reorganize its reactive sites and drive a chain of reactions that turn ordinary oxygen into powerful cleaners. Because the process avoids continuous chemical dosing and exploits mechanical energy that is often wasted, it offers a route to more sustainable water‑treatment technologies. With further engineering to tap gentle, naturally available motions instead of laboratory ultrasound, similar systems could help keep pharmaceuticals and other stubborn pollutants out of rivers, lakes, and drinking water supplies.
Citation: Dong, H., Zhou, Y., Li, Z. et al. Self-regulation of Lewis acid sites on FeOCl toward piezo-self-Fenton reaction for continuous hydroxyl radicals generation. Nat Commun 17, 3775 (2026). https://doi.org/10.1038/s41467-026-70327-0
Keywords: piezocatalysis, advanced oxidation, water purification, pharmaceutical wastewater, hydroxyl radicals