Clear Sky Science · en
Water-generated dangling linkers in a metal-organic framework
Why water can secretly reshape “super-stable” materials
Metal–organic frameworks, or MOFs, are sponge-like crystals that can soak up enormous amounts of water and other molecules, making them promising for clean water production, cooling systems, and drug delivery. UiO-66, one of the most studied MOFs, is widely regarded as almost indestructible in water. This paper shows that water is not always a harmless guest: even in this “water-stable” material, it can quietly rearrange the building blocks at the molecular level—yet in a way that turns out to be reversible.
Designer sponges for water and more
MOFs are built from metal clusters connected by organic linkers, forming a rigid, highly porous network. Because they can selectively capture and release water vapor, they are being explored for harvesting drinking water from desert air, humidity control in buildings, and water-based heat pumps. UiO-66, based on zirconium metal clusters and simple organic linkers, has become a workhorse because standard tests such as X-ray diffraction show that its long-range crystal structure survives repeated contact with water. Until now, this apparent robustness led many to assume that water barely alters its internal connections.
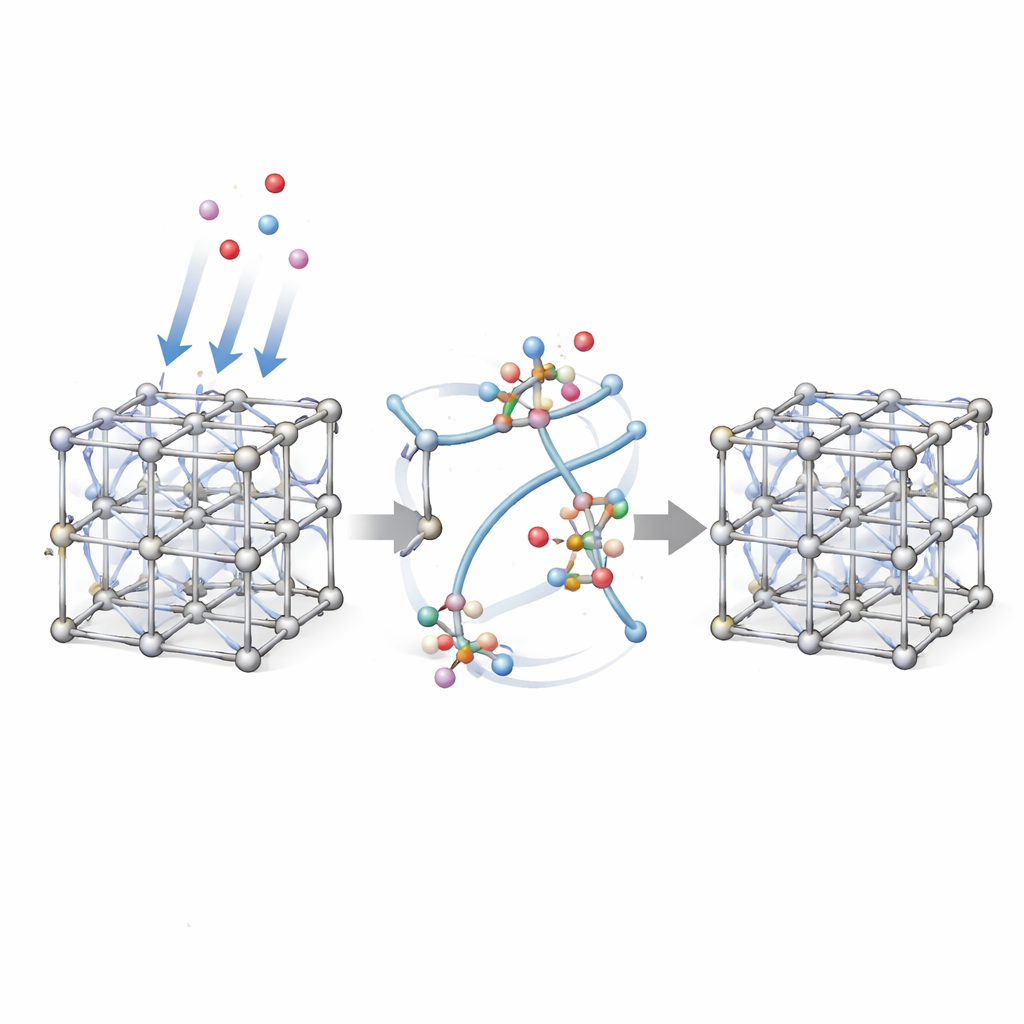
Water that nudges bonds without breaking the framework
The authors revisited this assumption by examining UiO-66 loaded with carefully controlled amounts of water. Using advanced solid-state nuclear magnetic resonance (NMR), they tracked how both the water molecules and the framework atoms behave. While X-ray patterns confirmed that the overall crystal lattice remained ordered, the NMR fingerprints told a subtler story: as more water was adsorbed, new signals appeared and existing ones broadened. These changes revealed that some of the organic linkers were no longer bound in the usual way to all of their metal neighbors, even though the overall framework did not collapse.
Dangling linkers held in place by water
Detailed two-dimensional NMR experiments showed that certain carboxylate groups—the parts of the linker that anchor to the zirconium cluster—had shifted into a distinct chemical environment. The data pointed to a scenario in which one end of a linker lets go of a metal site and swings slightly away, becoming a “dangling” group inside the pore. Rather than drifting freely, this loose end is stabilized by nearby water molecules and a neighboring hydroxyl group (an OH bound to the metal cluster) through a network of hydrogen bonds. Crucially, when the water is removed by gentle heating, the NMR spectra return to their original form, demonstrating that the linkers can reattach and the local disorder is reversible.
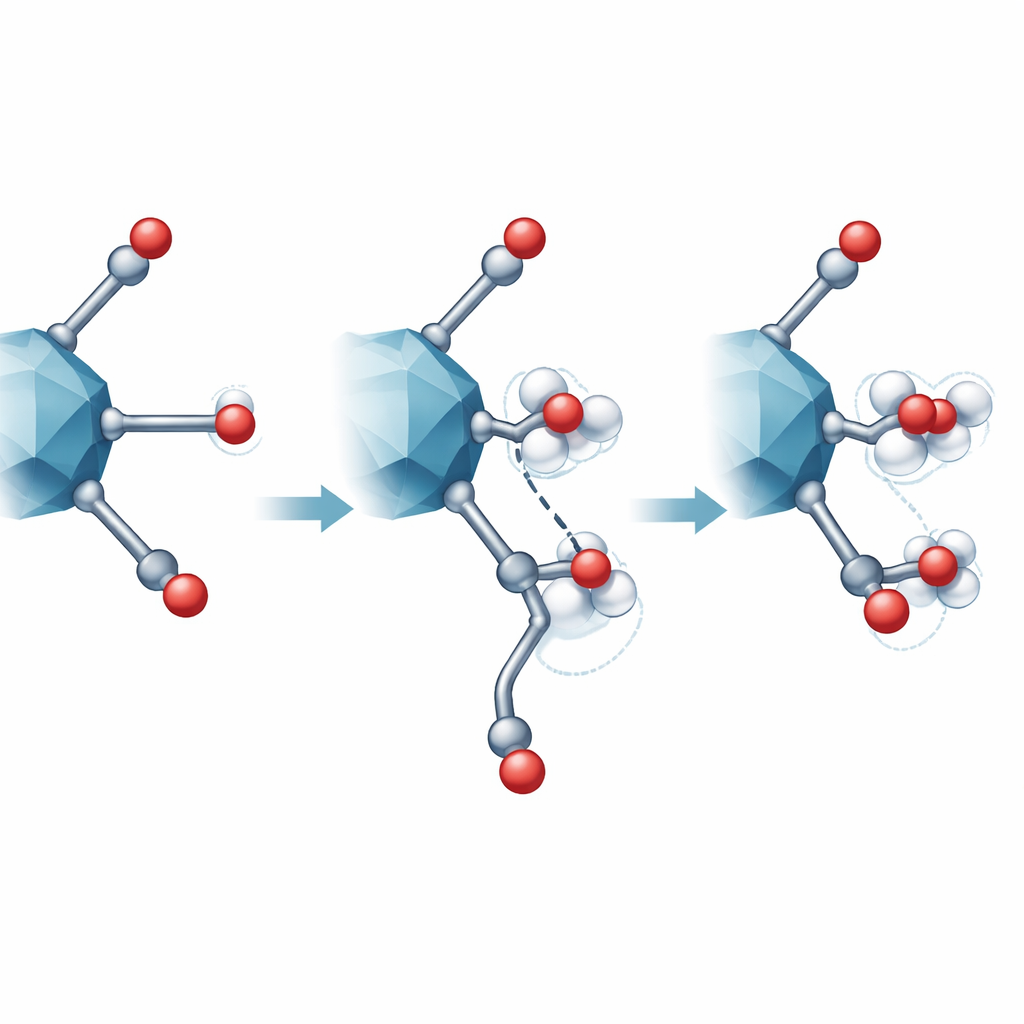
Computers reveal the most likely water–linker dance
To test which microscopic arrangement best matched the experiments, the team used quantum mechanical calculations to compare many possible ways a linker could detach and interact with one or two water molecules. They computed both the energy cost of each configuration and the expected NMR signatures. Only one arrangement fit all the observations: a carboxylate group detached on one side, pointing toward a nearby OH on the metal cluster, while two water molecules bridge between the open metal site and the dangling group. This configuration is energetically favored and reproduces the distinctive NMR shifts seen for both carbon and hydrogen atoms, including an OH group whose signal is pulled to unusually high frequency by strong hydrogen bonding.
What this means for future water-harvesting materials
This work reveals that water can temporarily reshape even supposedly water-stable MOFs at the molecular level, creating dynamic “dangling linkers” that are held in place by water itself. Because this process is reversible, UiO-66 can still withstand long-term use in wet conditions, but its internal bonding pattern is more flexible than previously believed. For designers of next-generation materials, this insight is crucial: understanding and controlling such subtle, water-driven rearrangements could help fine-tune how MOFs take up, release, and transport water and other molecules, leading to more efficient devices for water harvesting, cooling, and chemical delivery.
Citation: Fu, Y., Yao, Y., Paul, S. et al. Water-generated dangling linkers in a metal-organic framework. Nat Commun 17, 3805 (2026). https://doi.org/10.1038/s41467-026-70247-z
Keywords: metal-organic frameworks, UiO-66, water adsorption, solid-state NMR, hydrolytic stability