Clear Sky Science · en
Structural basis for prostaglandin and drug transport via SLCO2A1
How a Molecular Gatekeeper Shapes Drug and Hormone Effects
Many everyday medicines and hormone-like molecules must pass through tiny gateways in our cells before they can do their jobs. This paper explores one such gateway, a protein called SLCO2A1, which helps move powerful signalling fats called prostaglandins and several common drugs into cells. By revealing this protein’s 3D structure and how it grips its cargo, the researchers uncover why some drugs are carried through, while others simply block the gate. These insights could guide safer drug design and new treatments for conditions linked to inflammation, pain, gut disease, and poor wound healing.
The Body’s Traffic Controller for Pain and Inflammation Signals
Prostaglandins are short-lived, hormone-like fats that help control inflammation, pain, fever, blood flow, and organ development. They act near where they are made and must be rapidly cleared once their job is done. SLCO2A1 sits in the cell membrane and helps pull prostaglandins, and related molecules like thromboxanes, out of the surrounding fluid and into cells, where they can be broken down. When this transport fails, prostaglandin levels can stay abnormally high. Inherited faults in SLCO2A1 have been linked to rare bone and skin overgrowth disorders, chronic bowel disease, and poor healing of ulcers. Because prostaglandins and many widely used drugs share this route into cells, understanding how SLCO2A1 works is key to predicting drug effects and side effects across the body.
Seeing the Gate in Atomic Detail
To work out how SLCO2A1 recognizes and moves its cargo, the team used cryogenic electron microscopy, a technique that images flash-frozen proteins at near-atomic detail. They studied a closely related rat version of the transporter that behaves very similarly to the human one. They captured structures of the protein bound to two natural prostaglandins and to four medications used to treat asthma, high blood pressure, inflammation, and Parkinson’s disease. In all these images, SLCO2A1 appears as a bundle of twelve membrane-spanning helices forming a central cavity that is open to the outside of the cell. The prostaglandins sit deep in this cavity, with their ring-shaped core nestled near a cluster of key amino acids and their fatty tails threading into a largely oily groove that matches their greasy nature.
The Key Grip That Selects What Gets Through
By comparing structures and running long computer simulations, the researchers identified the features that let SLCO2A1 distinguish true cargo from mere look-alikes. A positively charged amino acid, arginine 561, and a nearby tryptophan and phenylalanine form a key “grip” for the negatively charged end of prostaglandins. When the team mutated these residues, transport largely failed, showing how critical this contact is. The surrounding cavity is mostly hydrophobic, favouring oily, membrane-loving molecules, and includes a “band” that acts as a pivot during the protein’s shape change. Two drugs, zafirlukast and losartan, are actually transported because they mimic the way prostaglandins present a negative group to this arginine while engaging the same hydrophobic surfaces. In contrast, the anti-inflammatory fentiazac and the Parkinson’s drug tolcapone bind less snugly and fail to stabilize the right pose, so they block the site instead of being carried through.
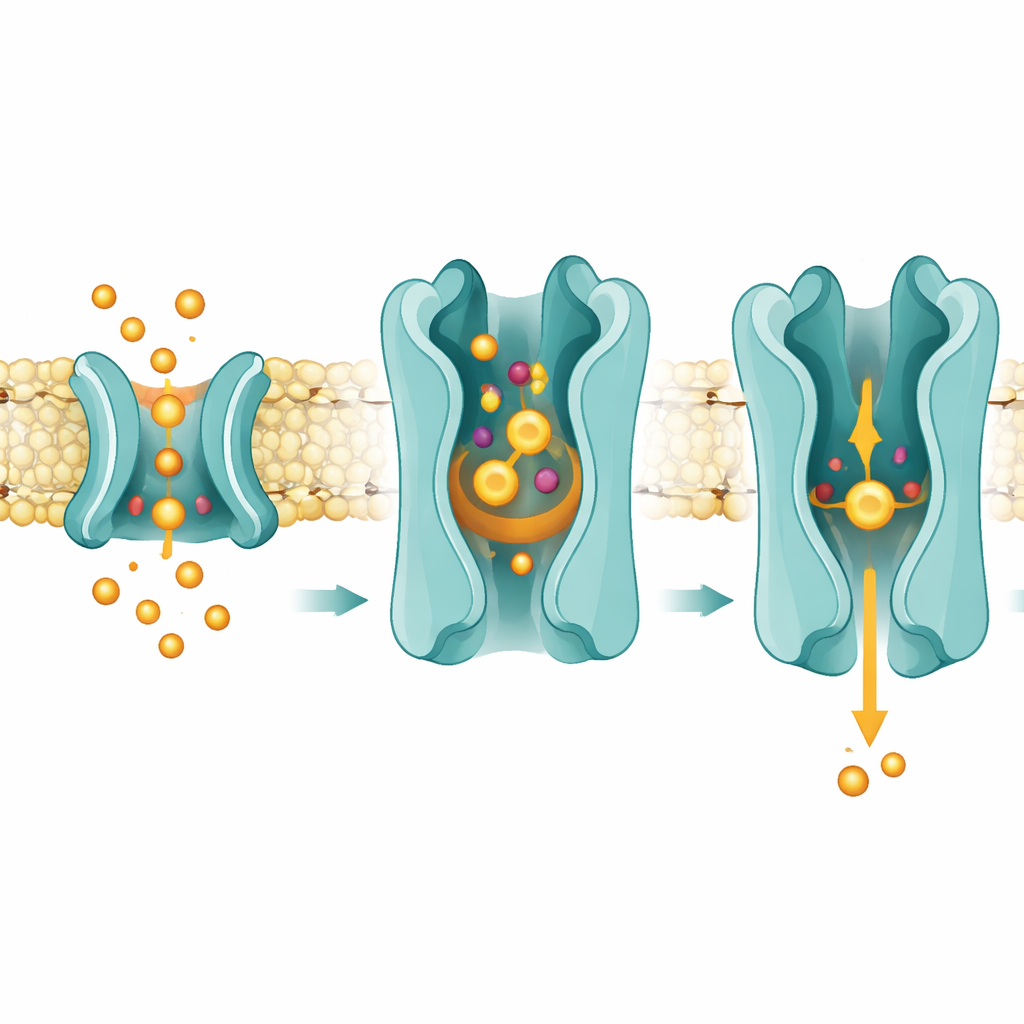
A Side Door Through the Membrane
The structures also hint that prostaglandins reach SLCO2A1 not from open water, but by sliding sideways through the fatty core of the membrane. The researchers observed densities consistent with lipid-like molecules nestled in a lateral opening between two helices, just above where prostaglandins bind. Disease-causing mutations and functional tests pinpointed this region as crucial for transport. The proposed mechanism is that prostaglandins first dissolve in the outer layer of the membrane, then slip into the transporter through this opening. Once bound in the central pocket, they disrupt a conserved salt bridge between arginine 561 and a glutamate residue, which in turn helps trigger the transporter to swing from an outward-open to an inward-open shape, releasing the prostaglandin toward the inner membrane and cell interior.
Why This Matters for Medicines and Disease
To a layperson, the main message is that SLCO2A1 acts as a finely tuned gate that governs how strong chemical signals of pain, fever, and inflammation are switched off, and how certain drugs enter cells. This study shows, in molecular detail, how the gate recognizes prostaglandins and why only some drugs can hitch a ride while others jam the door. By mapping the crucial contact points and the side entry route through the membrane, the work provides a blueprint for designing new drugs that either ride this transport system efficiently or deliberately block it in a controlled way. Ultimately, such knowledge may improve therapies for inflammatory diseases, gut disorders linked to prostaglandin imbalance, and drug regimens where transporter interactions currently cause unpredictable responses.
Citation: Joshi, C., Deme, J.C., Nakamura, Y. et al. Structural basis for prostaglandin and drug transport via SLCO2A1. Nat Commun 17, 2285 (2026). https://doi.org/10.1038/s41467-026-70227-3
Keywords: prostaglandin transport, drug transporters, SLCO2A1, membrane proteins, cryo-EM structure