Clear Sky Science · en
Carbon monoxide metabolism in freshwater anaerobic methanotrophic archaea
Why buried microbes matter for our climate
Deep in waterlogged mud, tiny microbes quietly destroy much of the methane that would otherwise leak into the air and warm our planet. This study reveals that some of these methane-eating microbes actually prefer another gas—carbon monoxide—and that this unexpected appetite can change how carbon moves through muddy lake and river bottoms. Understanding this hidden chemistry helps scientists refine climate models and rethink how wetlands and sediments act as natural filters for greenhouse gases. 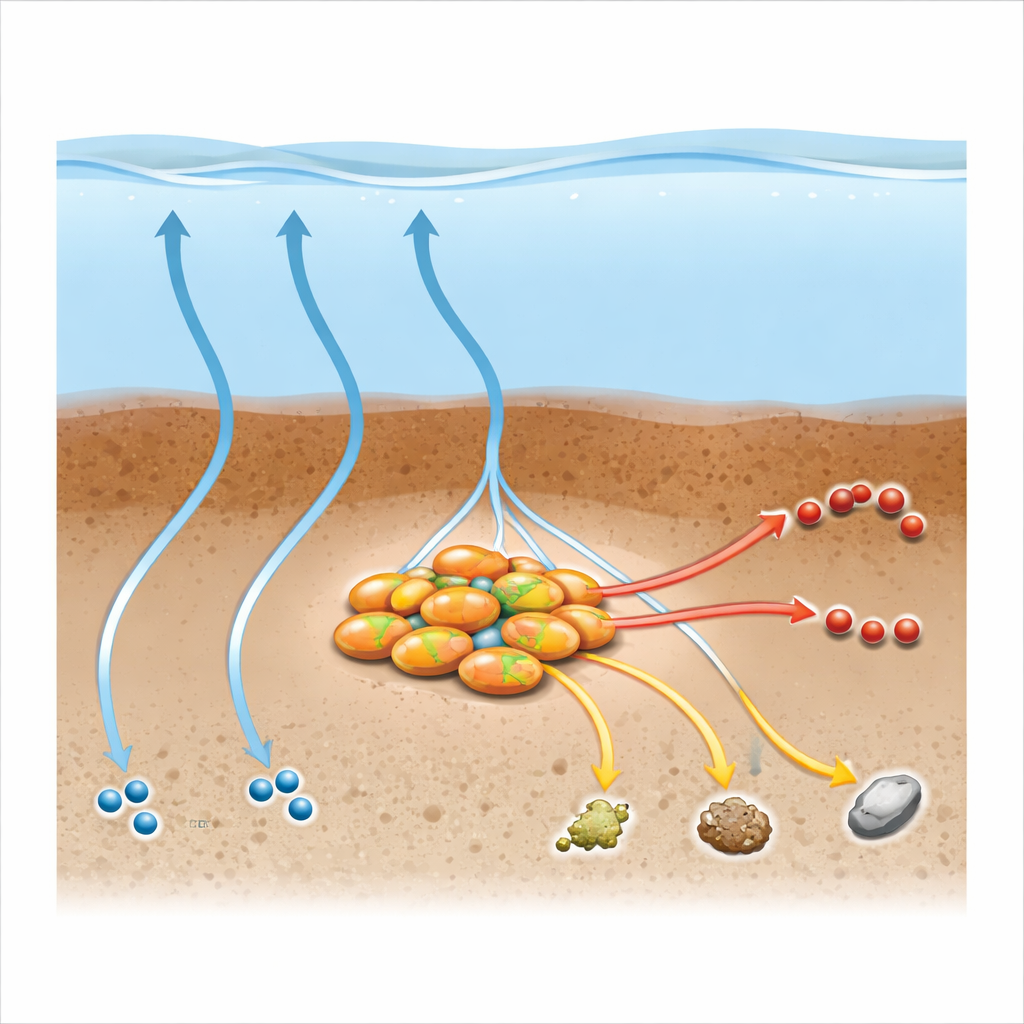
Methane’s secret gatekeepers
Methane is a powerful greenhouse gas, responsible for roughly one fifth of global warming despite being present in the atmosphere at only trace levels. In oxygen-poor mud at the bottom of lakes, rivers, and wetlands, specialized archaea—microbes distinct from bacteria—consume methane before it reaches the air. These anaerobic methanotrophic archaea form a major biological “methane filter.” One group, called Methanoperedenaceae, is especially versatile: it can link methane oxidation to several types of dissolved chemicals that serve as electron acceptors, such as nitrate and metals. Yet scientists have known surprisingly little about whether these microbes can also use fuels other than methane, or how such alternatives might strengthen—or weaken—the methane filter.
A microbe that favors a different fuel
The researchers focused on a freshwater archaeon known as “Candidatus Methanoperedens BLZ2,” now proposed to be renamed “Ca. Methanoperedens carboxydivorans” to reflect its taste for carbon monoxide. In carefully controlled bottle experiments, they supplied this microbe-rich sludge with either methane, carbon monoxide, or a mixture of both, and tracked gas consumption and product formation. Under conditions where nitrate was plentiful, the culture oxidized carbon monoxide much faster than methane, and when both gases were present, carbon monoxide almost completely suppressed methane use. When nitrate was removed, the microbes still rapidly consumed carbon monoxide, but now converted it mainly into methane, acetate, and formate—compounds that stay in the sediment rather than escaping directly to the air. 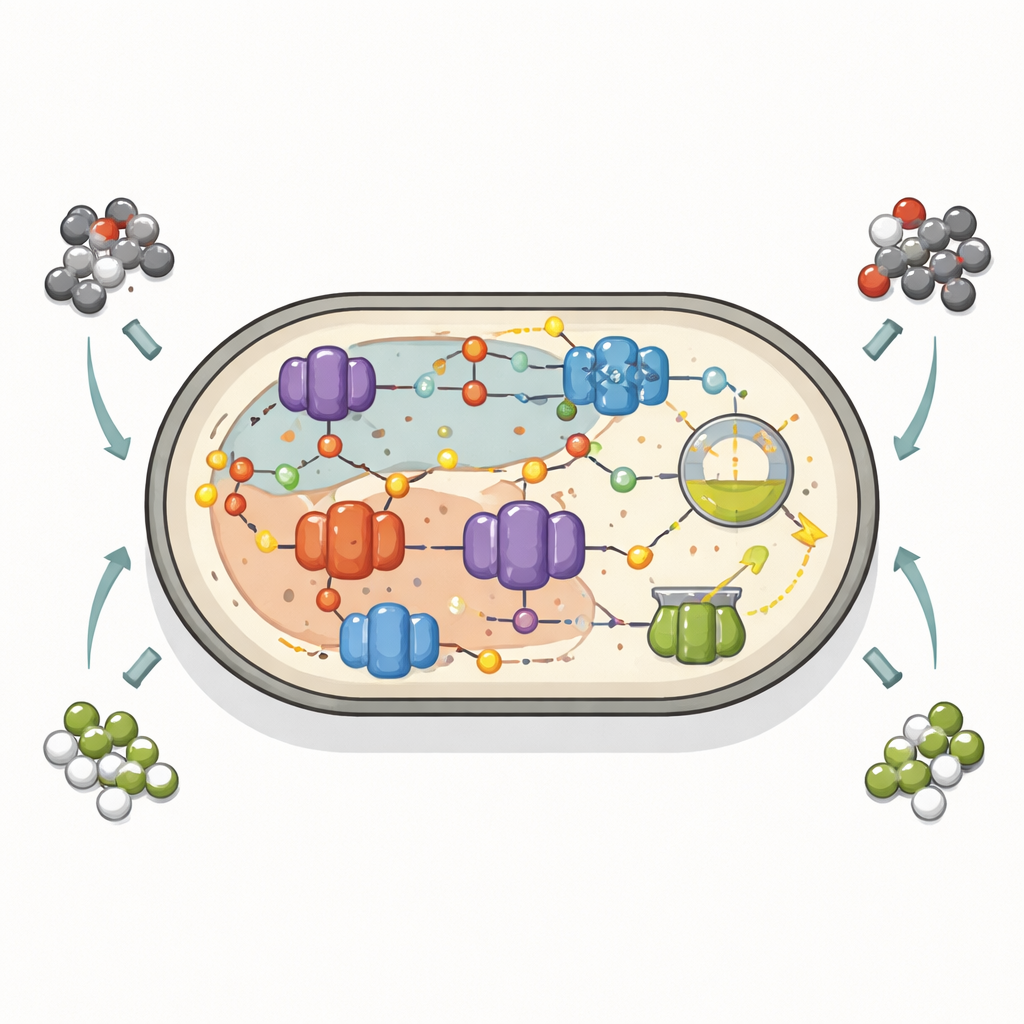
Hidden genetic tools for gas conversion
To understand how a single organism could handle such different tasks, the team reconstructed and fully circularized the genome of Ca. Methanoperedens carboxydivorans. They found an unusually rich toolkit for handling carbon monoxide: six genes for nickel-based carbon monoxide dehydrogenase enzymes in the main chromosome, plus two more on a separate circular piece of DNA known as a mobile genetic element. These enzymes sit at the heart of an ancient carbon-processing route that can run either toward building cell material or toward making products like acetate. The mobile element also carries extra modules for nitrate and nitrite reduction and for handling formate and other redox reactions, suggesting that chunks of metabolism can move between related microbes, boosting their ability to cope with shifting conditions in the mud.
How redox “traffic control” shapes the outcome
By examining which genes were switched on under different gas mixtures, the researchers pieced together a map of how the microbe reroutes its internal electron traffic. With nitrate available, the cell channels electrons from carbon monoxide toward respiration, powering energy-conserving pathways and leaving little in the way of side products. Without nitrate, those electrons must go somewhere else. The microbe then leans on fermentative processes: it uses carbon monoxide to generate reduced carriers and relieves this pressure by making acetate, methane, and formate. This behavior shows that a microbe normally known for destroying methane can also act like classic methane producers and acetate formers, depending on the surrounding chemical landscape.
Rethinking methane filters in nature
The study concludes that freshwater methane-oxidizing archaea are not strict methane specialists. Instead, they are flexible carbon converters that can shift between consuming methane and burning carbon monoxide, sometimes even producing methane themselves. Because genes for carbon monoxide conversion are widespread among related microbes, carbon monoxide—often overlooked in environmental surveys—may frequently nudge these communities away from methane oxidation. That means the effectiveness of the natural methane filter in sediments could rise or fall depending on subtle changes in carbon monoxide levels, reshaping how we think about greenhouse gas control in Earth’s hidden, oxygen-free corners.
Citation: Egas, R.A., Lin, H., Leu, A.O. et al. Carbon monoxide metabolism in freshwater anaerobic methanotrophic archaea. Nat Commun 17, 3460 (2026). https://doi.org/10.1038/s41467-026-70080-4
Keywords: methane cycle, carbon monoxide, wetland sediments, archaea, greenhouse gases