Clear Sky Science · en
Synthetic urine oversimplification results in misleading membrane fouling mechanisms in bipolar membrane electrodialysis
Why the Details in Our Waste Matter
Flushing the toilet might seem like the end of the story, but for engineers trying to recycle water and recover valuable nutrients, it is only the beginning. This study shows that when researchers use overly simple “fake urine” in the lab, they can be badly misled about how treatment systems will behave in the real world. By comparing simple and realistic urine recipes in a promising electric separation technology, the authors reveal that missing ingredients can flip our understanding of how and why treatment membranes clog, and how costly these systems will be to run.
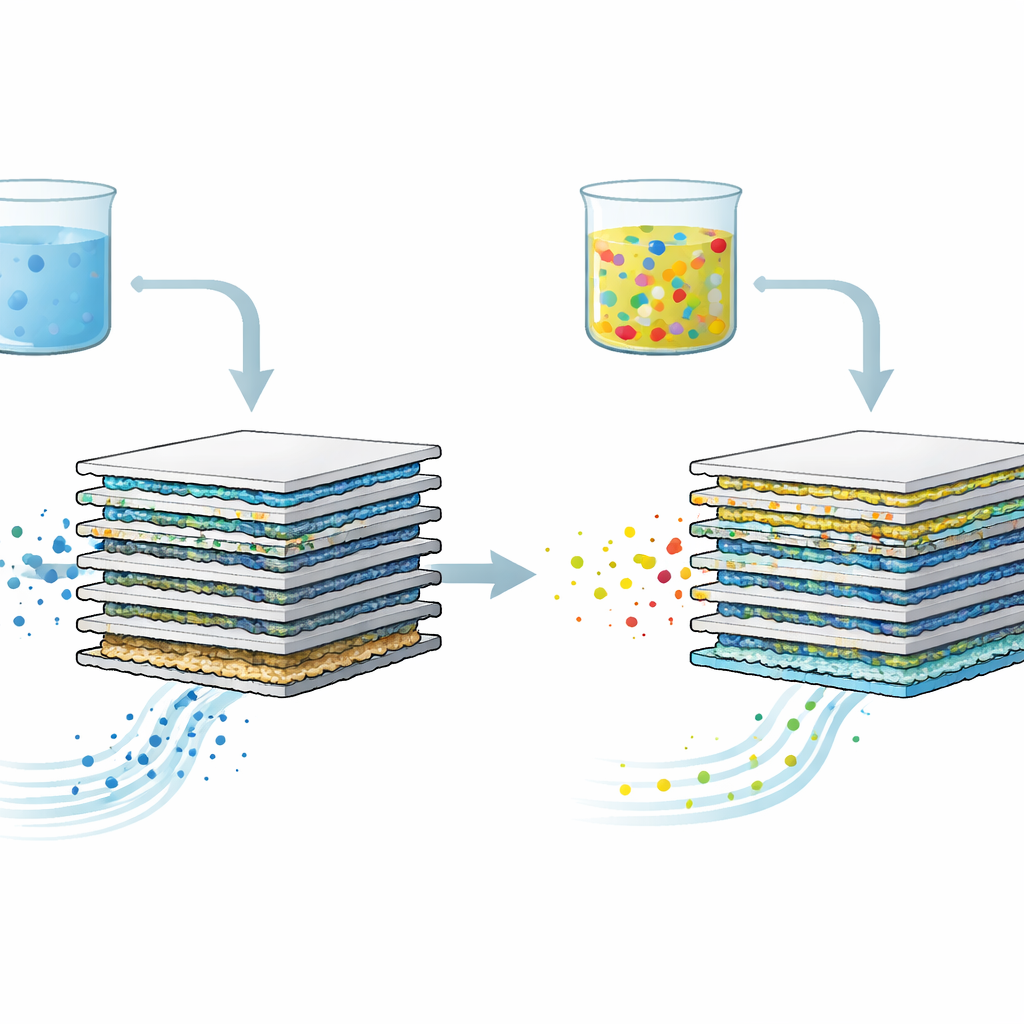
Turning Urine into Useful Resources
The work focuses on bipolar membrane electrodialysis, a technology that uses electricity and layered membranes to pull useful nutrients out of urine. In these systems, dissolved salts and nitrogen compounds move through special membranes to form acids, bases, and fertilizer products, while unwanted substances ideally stay behind. Because collecting large volumes of real urine is inconvenient and messy, many lab studies use simplified mixtures that contain only a few main ingredients, such as urea and common salts. The authors questioned whether this shortcut might hide important behavior seen in real urine, which is a rich cocktail of small molecules, proteins, and minerals.
Simple Recipes, Wrong Answers
To test this, the researchers created several synthetic urines with increasing complexity. One contained mainly salts and urea; another added typical small organic molecules found in real urine, such as creatinine and uric acid; a third also included a model protein similar to those excreted by the human body. They ran each mixture through the same electrodialysis setup for multiple batches and tracked how efficiently salts were removed and nutrients recovered. Surprisingly, the simplest mixture fouled the membranes the most and lost performance the fastest, with efficiency dropping by more than half after several runs. In contrast, the more complete mixtures kept working better for longer and recovered more urea, phosphorus, and potassium.
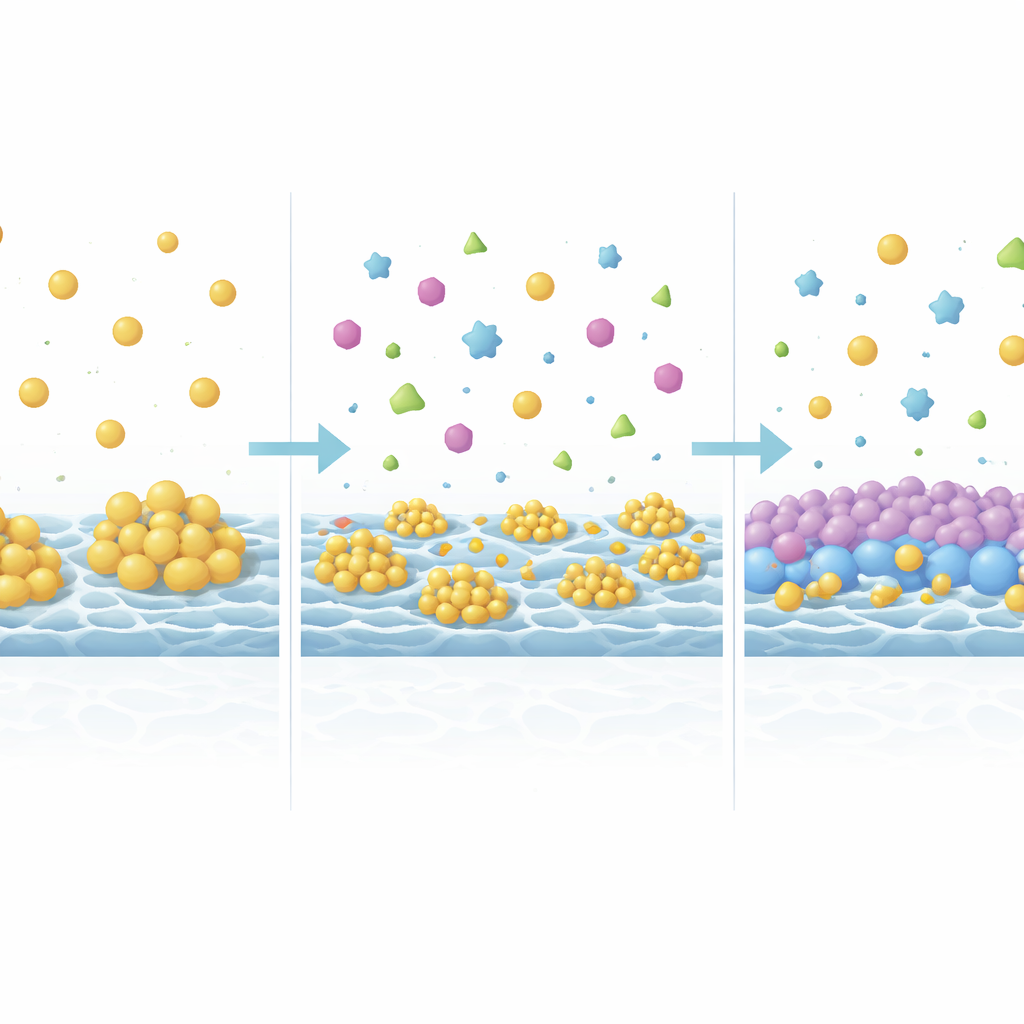
How Hidden Helpers Tame Membrane Clogging
Using high‑resolution imaging, surface chemistry measurements, and computer simulations, the team uncovered why complexity helps. In the simple mixture, urea molecules clumped together into large aggregates right on the membrane surfaces, held in place by networks of hydrogen bonds and other weak forces. These bulky deposits blocked ion pathways and degraded performance. When the missing organic molecules were added back in, they competed with urea for contact points on the membrane and on each other, breaking up or preventing these clumps. The added protein formed a thin, more water‑loving coating over the membrane, which physically kept urea away and allowed ions to move more easily. In short, natural urine components that had been left out of many experiments actually act as quiet stabilizers that slow membrane fouling.
Minerals, Deposits, and Shifting Trouble Spots
The study also examined how dissolved minerals like calcium and magnesium cooperate or misbehave with organics. In the more realistic mixtures, these ions tended to bind with urea and other organics near certain membranes, forming composite deposits that, while not ideal, stayed localized and predictable. But in an extreme case with only inorganic salts and no organics at all, calcium ions traveled farther into the device and crystallized directly on a key bipolar membrane, causing severe scaling in an entirely different location. This showed that stripping out organic matter does not simply reduce complexity: it can redirect where and how damaging deposits form, leading researchers to diagnose the wrong part of the system as the main risk.
Costs, Lifetimes, and Real‑World Decisions
Beyond the physics and chemistry, the authors translated these differences into money and time. They built a basic economic model using their lab data to estimate cleaning frequency, energy use, and membrane replacement for each urine recipe. Designs based on the oversimplified mixture predicted more frequent cleanings, shorter membrane lifetimes, and higher overall costs than systems treating more realistic urine. In numbers, simplification led to an overestimate of cleaning costs by roughly one‑sixth and an underestimate of membrane life by about one‑eighth. If such skewed estimates were carried into large‑scale projects, they could discourage investment in technologies that are actually more robust under real conditions.
What This Means for Future Water Recycling
For non‑specialists, the message is clear: when it comes to designing next‑generation wastewater recycling systems, cutting corners on realism can backfire. By carefully reconstructing the missing pieces of real urine, this study shows that natural mixtures contain built‑in checks and balances that can reduce membrane clogging and stabilize operation. Ignoring those interactions does not just give slightly off results; it can reverse our conclusions about what causes fouling, where it happens, and how expensive it will be to manage. The authors argue that future lab work on urine and other complex waste streams must preserve key ingredients and their interactions if we want trustworthy predictions for full‑scale, resource‑recovering treatment plants.
Citation: Yang, HR., Hu, SJ., Zhang, MY. et al. Synthetic urine oversimplification results in misleading membrane fouling mechanisms in bipolar membrane electrodialysis. Nat Commun 17, 3395 (2026). https://doi.org/10.1038/s41467-026-70034-w
Keywords: urine resource recovery, bipolar membrane electrodialysis, membrane fouling, wastewater treatment, synthetic urine formulations