Clear Sky Science · en
Rapid synthesis of micron-thick flexible graphite films via non-equilibrium carbon flux engineering
Why Faster Graphite Matters
From smartphones that stay cool to flexible robots that can feel heat, many emerging technologies depend on materials that move heat quickly and survive extreme conditions. Graphite, a familiar form of carbon related to pencil lead, is one of the best heat conductors known and remains stable at temperatures that would melt most metals. Yet making large, thin, flexible sheets of high‑quality graphite has been slow, energy‑hungry, and expensive. This article describes a new way to grow such graphite films in minutes instead of days, potentially opening the door to lighter, safer electronics, advanced batteries, and heat‑spreading layers that can bend without breaking.
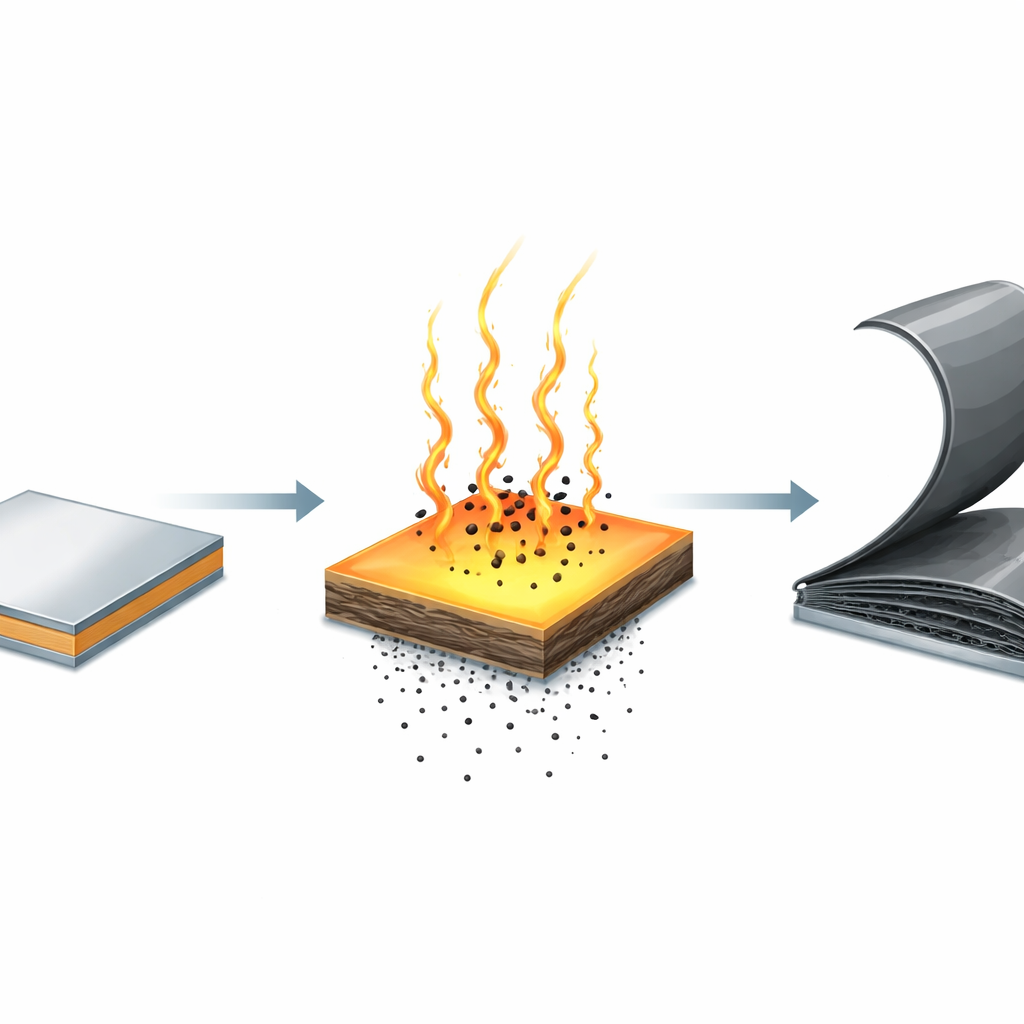
Limits of Today’s Graphite Making
Traditional methods for producing the best graphite resemble running an industrial kiln for days. Highly oriented pyrolytic graphite and Kish graphite, two benchmark forms, require extremely high temperatures, high pressures, and long treatment times to coax carbon atoms into near‑perfect order. Other approaches start from polymers or grow graphene layers on metal foils, but they still struggle to reach micron‑scale thickness quickly or to control defects. In practice, manufacturers have had to choose between crystal perfection on one side and speed and scalability on the other. This trade‑off has held back the wider use of thin, flexible graphite sheets in real‑world devices.
A Sudden Heat Shock Approach
The researchers introduce a very different strategy based on pulsed Joule heating, in which strong electrical currents briefly surge through a metal foil to create intense, short‑lived thermal shocks. They coat nickel or cobalt foils with a thin layer of a plastic called PMMA, which serves as a solid carbon source, then pass rapid current pulses through the stack inside a chamber filled with inert gas. The foil temperature shoots up above 1300 °C at heating rates exceeding 300 °C per second and then cools quickly. During the hot stage, carbon released from the decomposing plastic dissolves deeply into the metal. During the rapid cooling stage, the metal can suddenly hold far less carbon, forcing the extra atoms to rush outward and condense as ordered graphite layers on the surface. This non‑equilibrium carbon "traffic jam" greatly accelerates growth compared with slow, steady heating.
Growing Thick, High‑Quality Films Quickly
By carefully tuning the timing of the current pulses, the team achieves a vertical graphite growth rate of about 730 nanometers per minute on nickel—an order of magnitude faster than state‑of‑the‑art techniques. Measurements show that most of the graphite actually forms in just a few seconds during cooling, when carbon is violently expelled from the supersaturated metal. Using repeated heating–cooling cycles, the researchers push beyond the natural thickness limit of a single pulse and build up graphite films between 1 and 5 micrometers thick on both nickel and cobalt foils. The thickness increases almost linearly with the number of cycles, and a 5‑micrometer film can be produced in roughly two hours, a drastic improvement over multi‑day processes. Optical and surface measurements confirm that these films are continuous, uniform, and flexible over large areas.

Peering Inside the Metal During Growth
To understand why the process is so efficient, the authors trace how carbon moves inside the metal. A technique called time‑of‑flight secondary ion mass spectrometry lets them reconstruct three‑dimensional maps of carbon and nickel through the foil thickness at different growth times. Early on, carbon is spread fairly evenly throughout the nickel. After only about 10 to 12 seconds at high temperature, a distinct graphite layer emerges at the surface, revealing a rapid transformation. The maps also show that carbon flows especially quickly along the metal’s internal grain boundaries—tiny defects where atoms are packed less tightly—leading to thicker graphite ridges above those channels. Electron microscopy confirms a sharp, clean interface between the graphite and the metal, and atom‑scale imaging reveals the expected hexagonal lattice and stacking pattern that signify well‑ordered graphite.
Matching the Best in Structure and Performance
Beyond simply growing fast, the new films rival commercial graphite in structure and function. Diffraction measurements show that the spacing between carbon layers in these films is almost identical to that of high‑end graphite standards. Large‑area maps of crystal orientation indicate millimeter‑scale domains with highly aligned layers, interrupted mainly by wrinkles and occasional grain boundaries. Electrical measurements show high, uniform conductivity. Most strikingly, time‑domain thermoreflectance tests reveal in‑plane thermal conductivity exceeding 1300 watts per meter–kelvin, comparable to or better than many commercial graphite films and much higher than typical natural graphite. In other words, these rapidly grown sheets move heat nearly as well as the slow‑cooked materials they aim to replace.
What This Means Going Forward
In plain terms, the study shows that by briefly pushing a metal–carbon system far from its comfort zone—heating and cooling it much faster than usual—carbon atoms can be driven to organize into high‑quality graphite films at record speed. The resulting micron‑thick, flexible sheets combine strong heat‑spreading ability with large size and reasonable structural order, making them promising for cooling electronics, protecting components in extreme environments, and serving as building blocks in future carbon‑based technologies. Although the films are not yet as perfect as single crystals, the method already bridges a key gap between atomic‑level control and industrial‑scale output, and the same non‑equilibrium design principles may be extended to manufacture other layered materials efficiently.
Citation: Liu, H., Wang, Z., Wang, X. et al. Rapid synthesis of micron-thick flexible graphite films via non-equilibrium carbon flux engineering. Nat Commun 17, 3280 (2026). https://doi.org/10.1038/s41467-026-70028-8
Keywords: graphite films, thermal shock processing, carbon diffusion, heat spreaders, flexible electronics