Clear Sky Science · en
Eliminating the negative pore synergy in hierarchical porous metal-organic frameworks for isomer separation
Why this matters for everyday chemicals
Many products we use daily—from plastics and paints to gasoline and solvents—depend on mixtures of very similar molecules that are difficult and energy-intensive to separate. This study tackles a hidden problem inside advanced porous materials used for such separations: when their tiny channels are connected in the wrong way, molecules wander chaotically instead of flowing efficiently. By redesigning how these channels talk to each other, the authors show a new way to make chemical separations faster, cleaner, and more precise.
Tiny tunnels with different jobs
Porous materials called metal–organic frameworks, or MOFs, are built from metal clusters and organic linkers to form regular networks of nanoscale tunnels. Some of these tunnels are extremely narrow (micropores), which are excellent for gripping small molecules and telling similar ones apart. Others are wider (mesopores), which act like express lanes that let molecules move quickly. In theory, combining both types of pores in one material should give the best of both worlds: strong recognition of target molecules and rapid transport. The authors focus on a zirconium-based MOF named PCN-608, which naturally contains narrow triangular channels and wider hexagonal channels woven together, making it an ideal testbed for studying how these mixed tunnels behave.
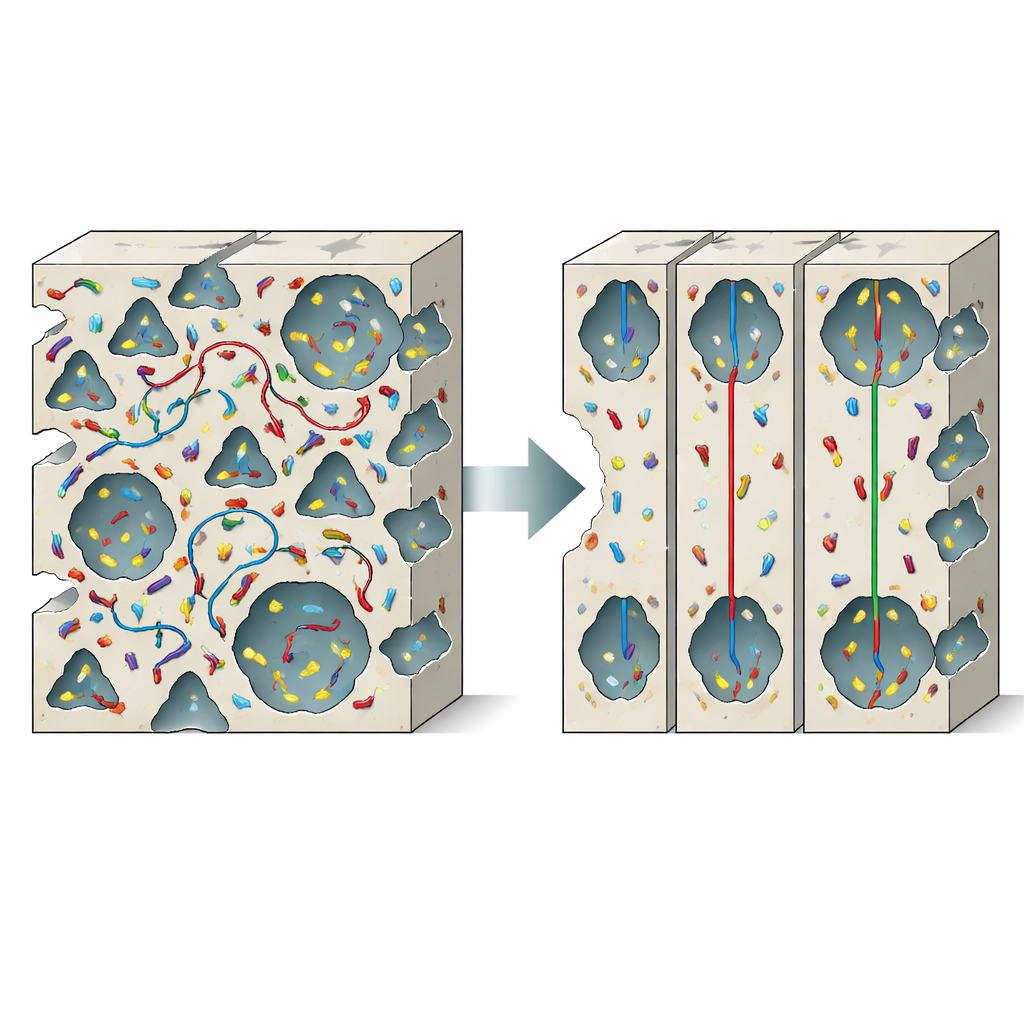
When more connections make things worse
Instead of helping, the connections between the small and large channels in PCN-608 actually create a problem the authors call negative pore synergy. Using computer simulations, they show that xylene molecules—a family of industrially important aromatics with three nearly identical isomers—do not move smoothly through the framework. Instead, they keep hopping back and forth between the two types of channels through side windows lined with highly attractive metal sites. This multitunnel wandering stretches out their paths, slows overall movement, and prevents the narrow pores from fully “reading” the subtle shape differences between isomers. In effect, the very feature meant to improve performance—the interlinked pore network—scrambles the motion of molecules and blurs separation.
Sealing side doors to straighten the path
To fix this, the team develops a channel-isolation strategy. They install short organic pieces, called barrier ligands, precisely at the side windows that connect the triangular and hexagonal channels. These ligands, based on benzene dicarboxylic acids with or without an amine group, bridge metal sites and physically narrow or close the openings between pore systems. Careful structural tests confirm that the overall framework stays intact: the crystal structure, particle shape, and basic dual-pore architecture are preserved, while the pores become slightly smaller due to the added barriers. Computer simulations show that after this modification, xylene molecules no longer accumulate at the windows or shuttle between channels; instead, they follow well-defined, more direct paths within individual channels.
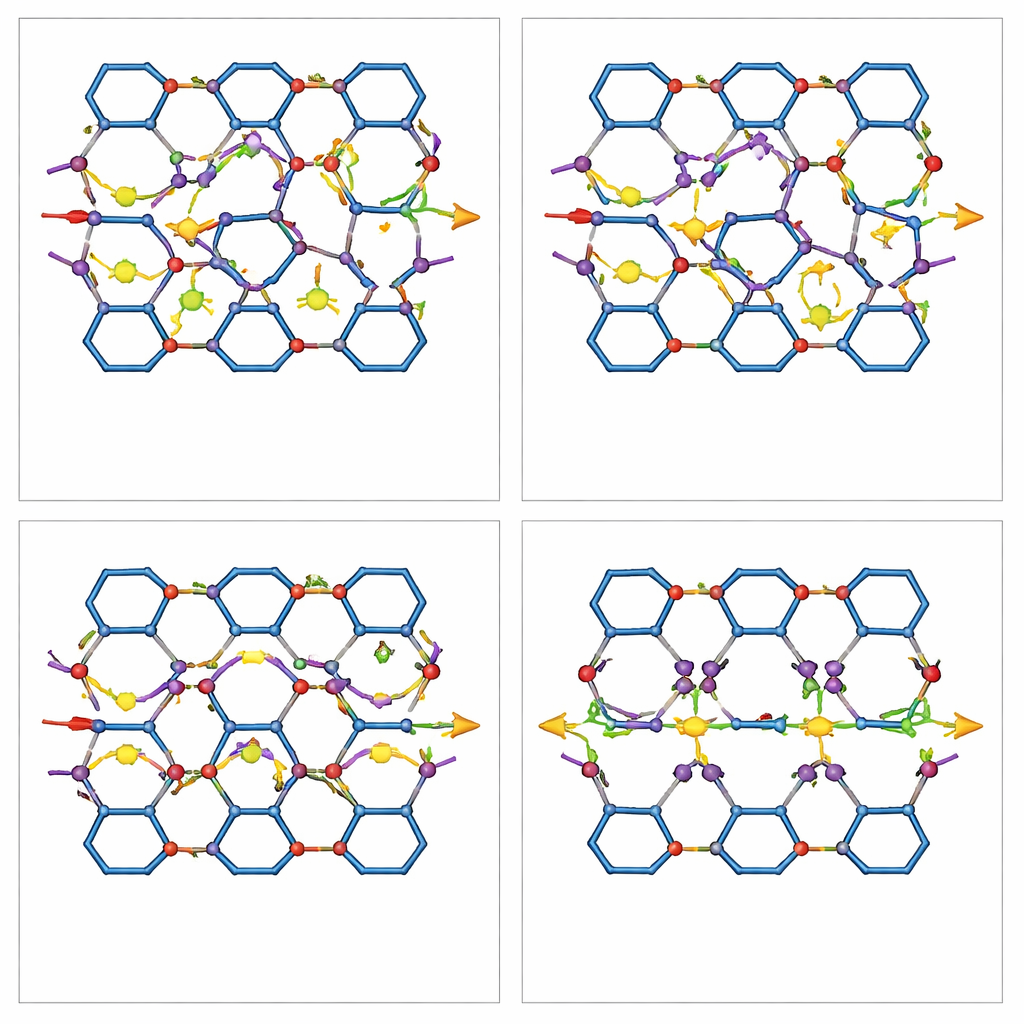
Faster motion and cleaner separations
With the channels isolated, measurements using inverse gas chromatography and vapor adsorption reveal that xylene molecules diffuse much more quickly—up to 8–13 times faster in the modified PCN-608-BDC material than in the original PCN-608. Interestingly, the barriers weaken some of the strongest binding spots in the framework, so the overall attraction to the molecules becomes slightly lower. Yet this does not harm performance; it actually helps by reducing nonspecific sticking and allowing the material to discriminate isomers more by how they move than by how strongly they bind. In gas chromatography tests, columns coated with the modified MOFs separate xylene isomers into sharp, well-resolved peaks, while the unmodified material largely fails. Breakthrough experiments, which mimic continuous industrial operation, further show that the isolated-pore version pulls apart isomer mixtures more effectively and much faster than the original.
A general blueprint for better porous materials
To test whether this idea is specific to one framework, the authors apply the same barrier-ligand approach to another well-known MOF, NU-1000, which shares a similar channel layout. Again, blocking the interchannel windows transforms muddled separations into clean ones, confirming that negative pore synergy is a widespread issue and that channel isolation is a broadly useful solution. Overall, the work shows that it is not enough to design materials with many pores and large internal surface areas; how those pores are connected can make or break performance. By deliberately sealing unnecessary side doors between channels, the authors offer a practical design rule for future separation materials that aim to combine high selectivity, rapid transport, and long-term stability.
Citation: Liu, JJ., Xu, M., Meng, SS. et al. Eliminating the negative pore synergy in hierarchical porous metal-organic frameworks for isomer separation. Nat Commun 17, 3193 (2026). https://doi.org/10.1038/s41467-026-69971-3
Keywords: metal-organic frameworks, pore connectivity, xylene isomer separation, molecular diffusion, gas chromatography