Clear Sky Science · en
Selective weakening of population-coupled synaptic activity in vivo in a mouse model of amyloid-beta pathology
Why this research matters for brain health
Alzheimer’s disease is often described as a disease of “dying brain cells,” but long before many cells are lost, their connections – the synapses – start to fail. This paper asks a precise question with big implications: in the early stages of Alzheimer-like pathology, does the brain lose function everywhere at once, or are particular synapses hit first? By following living brain circuits in a mouse model that develops amyloid-beta plaques, the authors uncover a surprisingly selective weakness in a specific class of connections that help keep brain activity in balance.
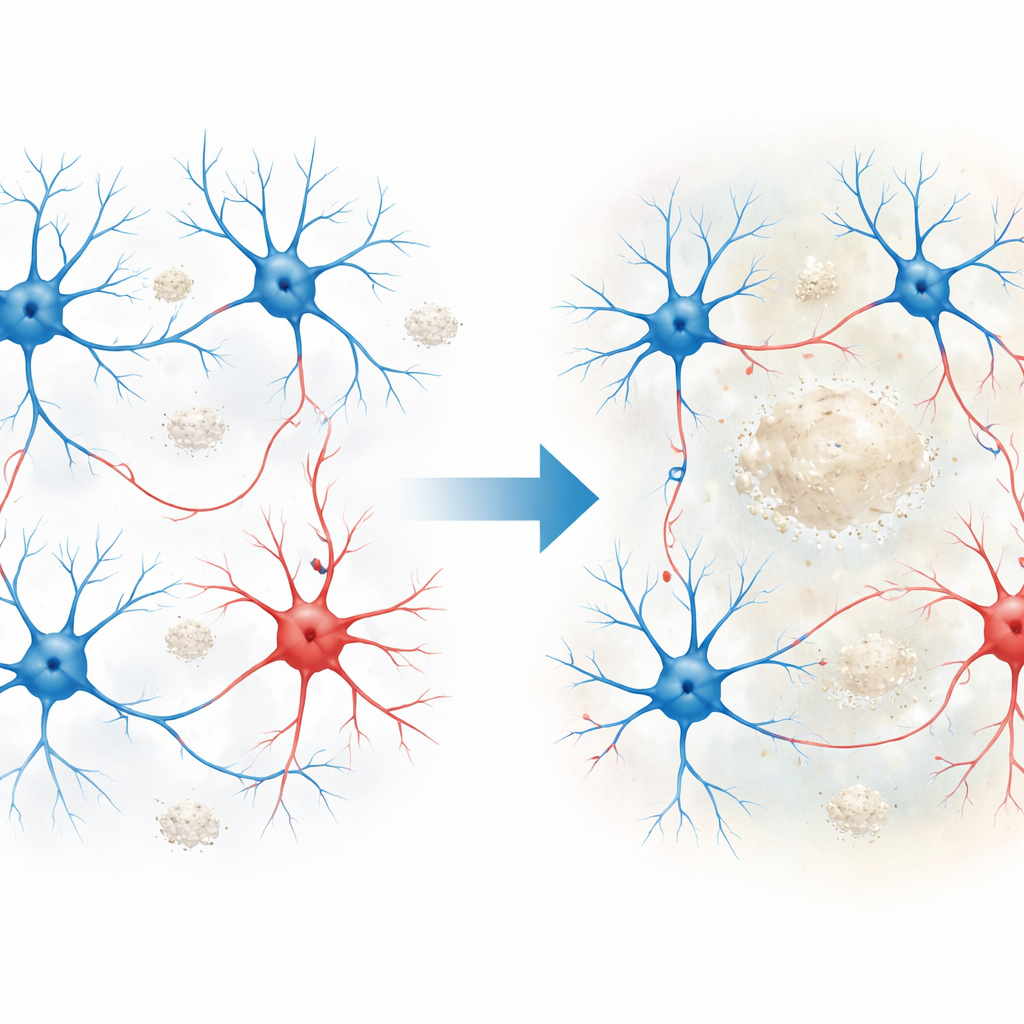
Early warning signs in brain activity
The researchers first looked at how whole brain regions behaved as amyloid plaques accumulated. Using widefield calcium imaging and EEG in mice, they tracked the activity of large groups of neurons across the cortex during quiet “resting” periods. They found that posterior brain regions, especially visual and retrosplenial cortex, became unusually active early on, while frontal areas stayed relatively stable at these stages. At the cellular level, both excitatory cells (which drive activity) and inhibitory cells (which restrain it) showed stronger calcium signals, indicating hyperactivity. Yet, despite this extra firing, the usual tight coordination between excitatory and inhibitory groups broke down: neuron pairs that normally fire together became less correlated, hinting at a circuit that is both overactive and poorly organized.
A closer look at vulnerable connections
To understand what lies beneath these circuit-level changes, the team zoomed in to the level of single synapses. They separately imaged excitatory dendritic spines and tiny swellings along inhibitory axons called boutons, which release the calming neurotransmitter GABA. Around plaques in vulnerable cortical regions, both excitatory and inhibitory synaptic markers were reduced, but a clear pattern emerged: loss of GABA-related presynaptic proteins was earlier and stronger than changes at excitatory synapses. In live imaging, inhibitory boutons showed reduced calcium activity even before their numbers fell, with the sharpest weakening in boutons closer to plaques and as the animals aged. Excitatory spines did change, but their alterations were more modest and appeared later, suggesting that inhibitory connections are the earlier and more fragile link in this disease model.
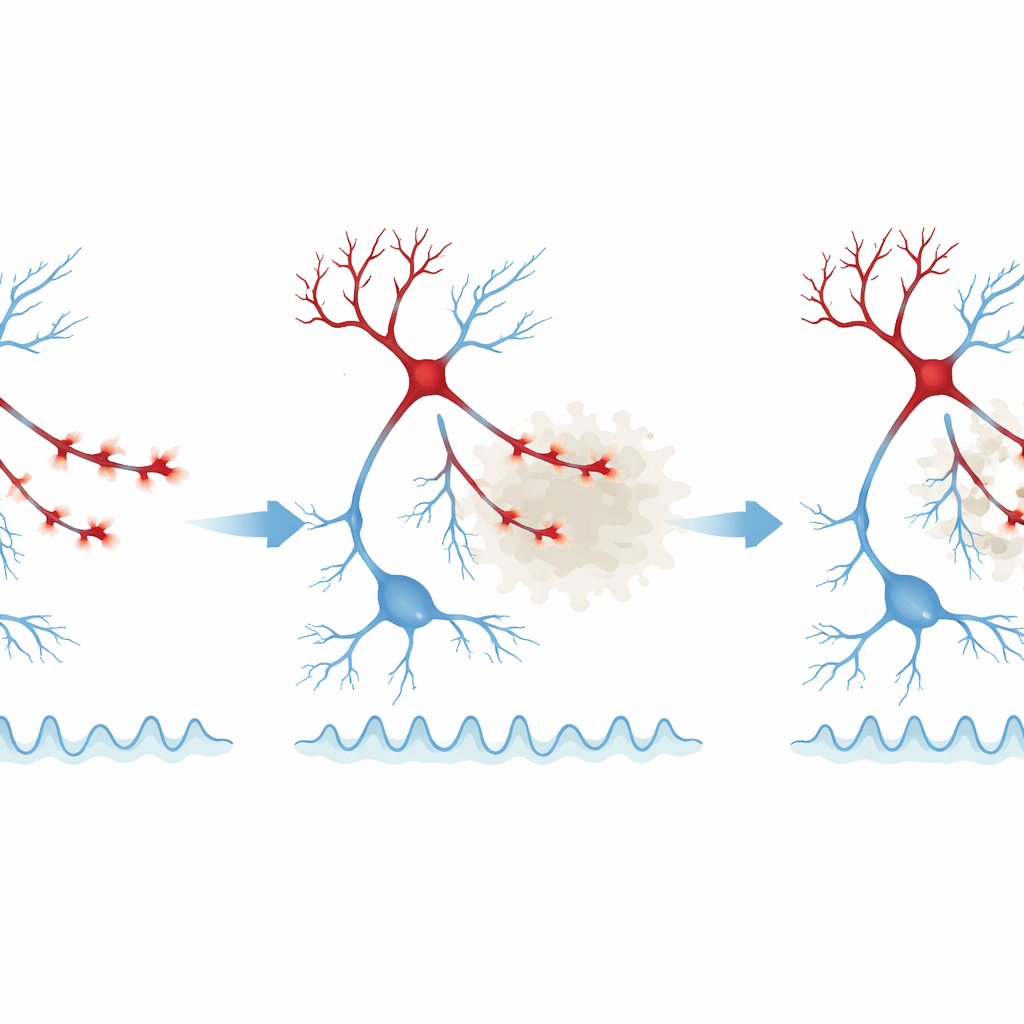
When “hub” synapses fail
A key insight from this work is that not all synapses are equally affected. The authors measured how strongly each bouton or spine was “coupled” to the overall activity of its local network – essentially, how much its behavior tracked the surrounding population. The most strongly population-coupled inhibitory boutons, especially those near plaques, showed the greatest drop in activity, while weakly coupled boutons in the same environment often looked normal. A similar but milder pattern was seen in excitatory spines. This suggests that synapses that act as hubs for coordinating network rhythms are selectively weakened, which could explain why resting-state activity becomes both excessive and disorganized, and why brain oscillations in specific frequency bands are disrupted in the presence of amyloid.
Cell types carrying the molecular burden
To link these functional changes to underlying biology, the authors used spatial transcriptomics, a technique that maps gene activity across thousands of individual cells in intact tissue. They focused on inhibitory neuron subtypes and found that parvalbumin-positive (PV) interneurons – a class known to tightly control network timing – showed the earliest and strongest shifts in gene expression. Genes related to GABA release, receptors, and the synaptic vesicle cycle were notably altered, pointing to disturbed inhibitory communication at the molecular level. As pathology progressed, other inhibitory subtypes, including somatostatin-positive (SST) cells, also showed broad changes tied to synaptic transmission and plasticity, particularly in areas with higher plaque loads. These molecular signatures align with the imaging data that highlighted early and pronounced weakening at GABAergic synapses.
What this means for understanding Alzheimer’s disease
Overall, this study argues that early synaptic problems in an Alzheimer-like brain are not a uniform “turning down” of all connections. Instead, the most vulnerable elements are specific inhibitory presynaptic boutons and related excitatory spines that are strongly wired into local network activity and sit within the amyloid-rich microenvironment. Their weakening likely undermines the brain’s ability to keep activity in check, contributing to hyperactivity, abnormal rhythms, and, eventually, cognitive decline. For a lay audience, the takeaway is that Alzheimer’s may begin with targeted failures in key circuit “brakes” rather than a slow, even fade of all synapses – a distinction that could guide more focused treatments aimed at preserving or restoring these critical inhibitory connections.
Citation: Melgosa-Ecenarro, L., Radulescu, C.I., Doostdar, N. et al. Selective weakening of population-coupled synaptic activity in vivo in a mouse model of amyloid-beta pathology. Nat Commun 17, 3646 (2026). https://doi.org/10.1038/s41467-026-69866-3
Keywords: Alzheimer’s disease, synaptic dysfunction, GABAergic inhibition, amyloid-beta, cortical circuits