Clear Sky Science · en
Catalytic asymmetric functionalization of bicyclo[1.1.0]butane boronic esters enabled by 1,2-oxygen migration
Why tiny square rings matter
Chemists love to build new medicines and materials by snapping small molecular pieces together, much like using Lego bricks. Four‑membered carbon rings called cyclobutanes are especially prized because they pack a lot of energy and shape into a small space, which can change how a drug behaves in the body. This article describes a new way to make these rings in a very controlled, “one‑handed” (chiral) form, using a clever reaction that forces an oxygen‑containing group to hop from one atom to the next. The work opens doors to designing more complex and precise molecules for pharmaceuticals and advanced materials.
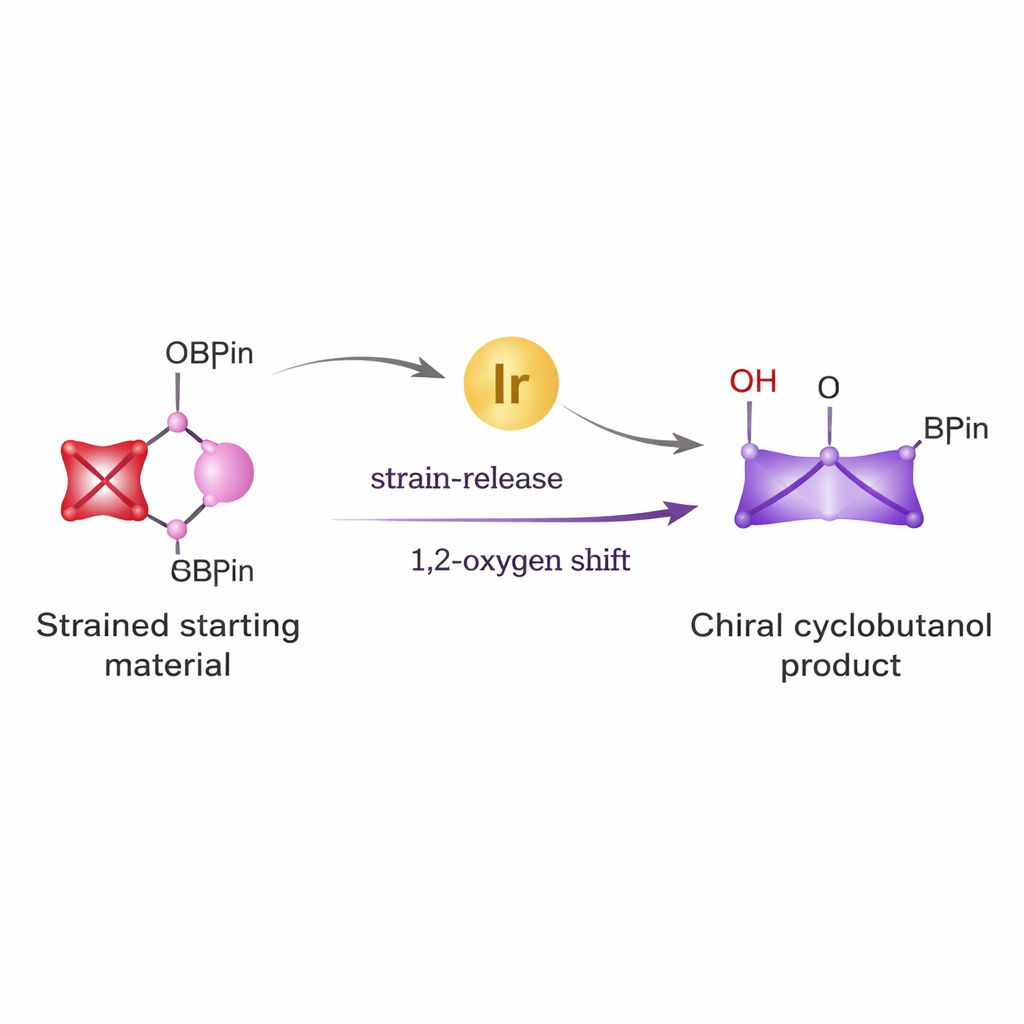
From simple building blocks to powerful rings
Cyclobutanes appear in a variety of antiviral agents, anticancer candidates, and other bioactive molecules. Yet building them with the exact three‑dimensional arrangement needed for medicine has been difficult. Traditional routes often require harsh reagents and give mixtures of mirror‑image products that are hard to separate. The authors set out to solve this problem by combining two powerful ideas: the unusual reactivity of a tiny, highly strained ring called bicyclo[1.1.0]butane, and the versatility of boronic esters, a well‑known family of reagents widely used in cross‑coupling chemistry for forming carbon–carbon bonds.
Turning a drawback into a new reaction path
In standard metal‑catalyzed coupling reactions, boronic esters form an intermediate complex with an alkoxide (an oxygen‑based base). This complex almost always follows a well‑worn path called transmetallation, which then leads directly to new carbon–carbon or carbon–halogen bonds. That “default” route has made it hard to explore other possibilities, such as shifting the oxygen group itself. The team realized that if they started from a ring‑strained bicyclo[1.1.0]butane boronic ester, the built‑in tension of the small ring could be used to override the usual pathway. By pairing this strained system with an iridium catalyst and a carefully chosen chiral ligand, they hoped to coax the oxygen to migrate in a controlled fashion, while the ring opened and re‑closed into a cyclobutane.
A stepwise dance guided by strain
Experiments confirmed that this strategy works. Using lithium alkoxides made from a wide variety of alcohols—including sugars, terpenes, steroids, and simple aliphatic alcohols—the authors converted bicyclo[1.1.0]butane boronic esters into cis‑cyclobutanol products with both high yield and excellent control over chirality. The iridium catalyst first forms a reactive allyl complex from an allylic carbonate partner. This complex then attacks the strained bicyclo[1.1.0]butane system, forming a new carbon–carbon bond as the tiny ring opens. Crucially, instead of immediately following the usual transmetallation route, the resulting boron‑containing intermediate undergoes a subtle rotation of the carbon–boron bond, positioning the attached oxygen group so that it can shift cleanly to a neighboring carbon atom. This 1,2‑oxygen migration locks in the desired cis‑arrangement on the new cyclobutane ring.
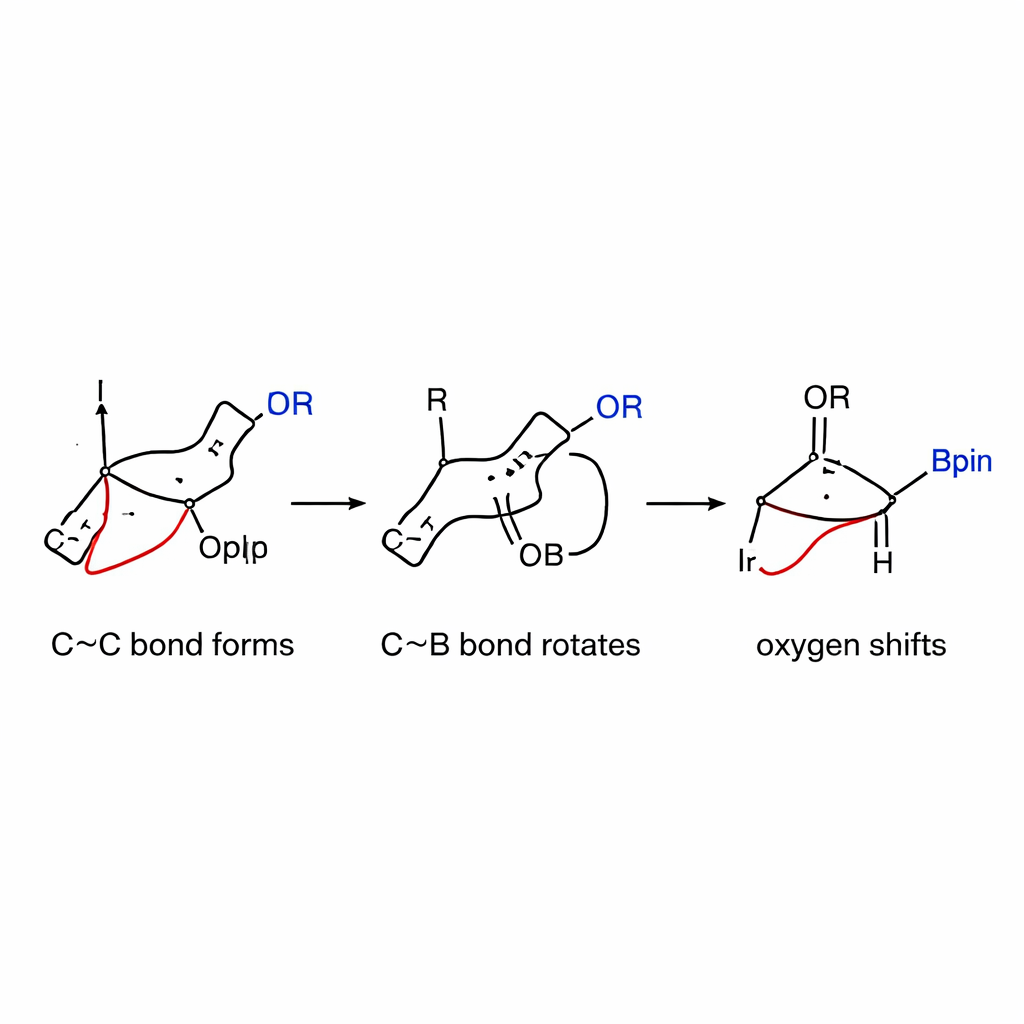
Peeking under the hood of the mechanism
To understand why the reaction is so selective, the authors combined kinetic measurements with detailed computer simulations. They found that the slow, rate‑controlling step is the initial carbon–carbon bond formation between the iridium–allyl species and the strained boronate complex, not the earlier activation of the allylic carbonate. Electronic tests showed that allyl partners that stabilize positive charge react faster, supporting a mechanism in which a partially charged intermediate is formed. Calculations further revealed that rotation around the carbon–boron bond—previously thought to be difficult—has a surprisingly low energy barrier in this system, thanks to the stored ring strain. Once rotation has aligned the oxygen group, the 1,2‑shift proceeds smoothly and preferentially to give the cis product rather than the more common trans form.
New tools for shaping future medicines
Beyond proving a mechanistic point, the reaction delivers versatile products. The cyclobutanols retain a boronic ester unit, which can be transformed into many other functional groups using established chemistry. The authors showcased numerous “late‑stage” modifications, such as hydrogenation, oxidation, olefin metathesis, and further couplings, all while preserving the precise three‑dimensional shape of the ring. In plain terms, this work turns a once‑problematic side pathway—oxygen migration in boronate complexes—into a powerful synthetic tool. By harnessing ring strain and a finely tuned iridium catalyst, the researchers provide chemists with a reliable way to build complex, chiral cyclobutanes that could play key roles in next‑generation drugs and advanced molecular materials.
Citation: Zhu, XY., Ji, CL., Dong, TG. et al. Catalytic asymmetric functionalization of bicyclo[1.1.0]butane boronic esters enabled by 1,2-oxygen migration. Nat Commun 17, 1941 (2026). https://doi.org/10.1038/s41467-026-69860-9
Keywords: cyclobutane, organoboron chemistry, asymmetric catalysis, bicyclobutane, oxygen migration