Clear Sky Science · en
Precise regulation of missing linkers in MOF pervaporation membranes for desalination of hypersaline waters
Turning Salty Waste into Fresh Water
As freshwater supplies shrink and industrial activities generate ever-saltier waste streams, finding ways to turn extremely salty water into drinking water becomes crucial. This study explores a new kind of ultra-thin filter made from a crystalline material called a metal–organic framework (MOF). By deliberately creating tiny “missing pieces” inside this material, the researchers show how to move water molecules quickly through the membrane while almost completely blocking salt, even in very harsh conditions. 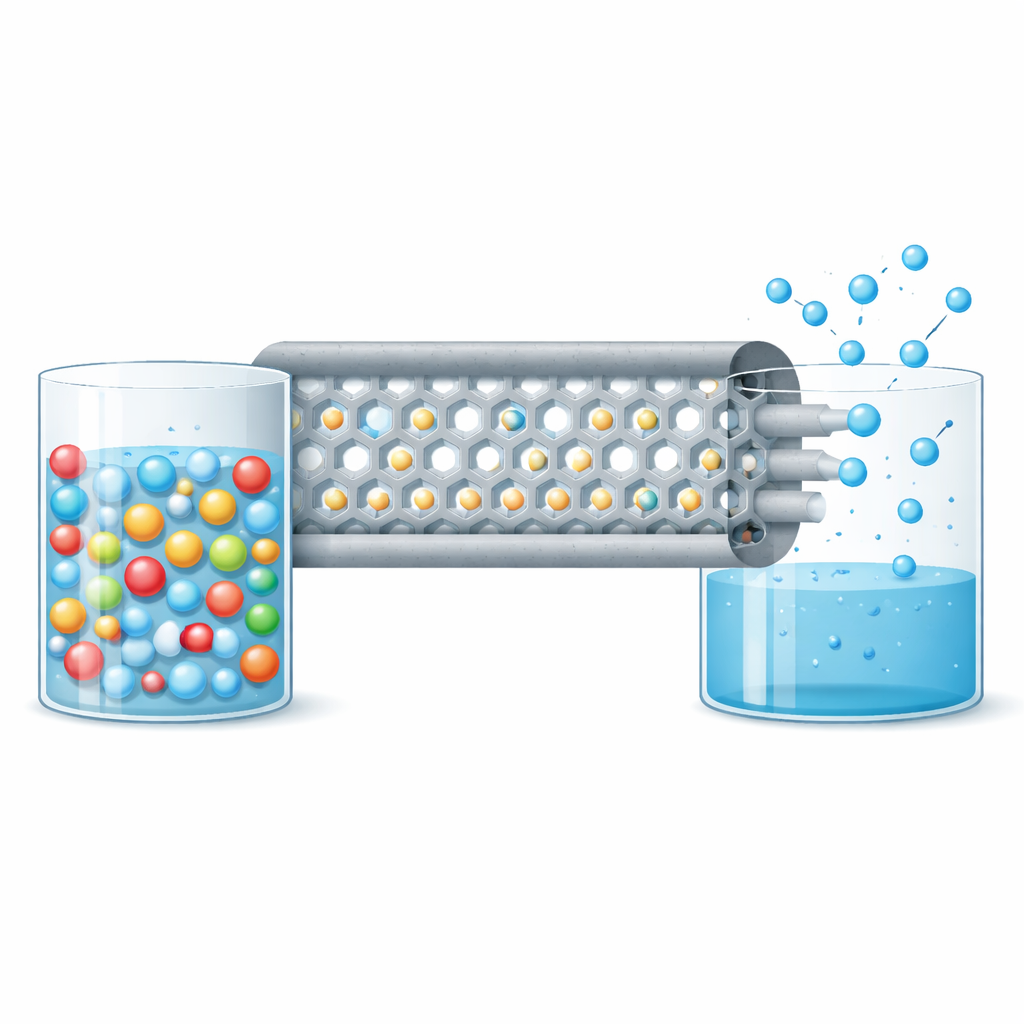
Why Very Salty Water Is So Hard to Clean
Conventional desalination plants mostly rely on reverse osmosis, which uses high pressure to push seawater through dense polymer films. This works well for seawater, but not for hypersaline waters such as concentrated brines or industrial waste streams: the salt content is so high that the required pressure becomes impractical. Heat-driven methods like distillation can handle higher salinities but often suffer from problems like wetting and scaling of the membranes. Pervaporation offers a different approach: warm salty water contacts one side of a membrane, a vacuum is applied on the other, and only water molecules evaporate through, leaving dissolved salts behind because they do not readily vaporize under operating conditions.
Building a Better Nanochannel Filter
The team focused on MOF-801, a zirconium-based framework with well-defined nano-sized channels that are inherently well matched to the size difference between water molecules and hydrated salt ions. They grew MOF-801 as a thin, continuous layer on robust ceramic hollow fibers, which pack a very large membrane area into a small volume. To encourage the formation of a smooth, crack-free layer, they first added a thin titanium dioxide coating to the ceramic to provide extra nucleation sites, and then used a “nanoseeding” step to deposit tiny MOF-801 crystals. A gentle surfactant treatment slowed solvent evaporation during drying, which prevented the formation of cracks that would otherwise provide easy, unwanted pathways for salt to slip through.
Using Missing Pieces to Speed Up Water
The key innovation is the precise control of “missing linkers”—deliberate vacancies in the molecular struts that connect the metal clusters in MOF-801. By adjusting the ratio of two simple acids, fumaric acid and formic acid, during synthesis, the authors could tune how many linkers were absent inside the crystal. Detailed measurements showed that as more linkers were removed, the pores within the framework became slightly larger, the internal surface area increased, and the material could hold more water. Computer simulations and experiments together revealed why this helps: missing linkers expose extra water-loving metal-oxide sites, making the structure more hydrophilic, and they widen both the entrances and internal cages of the channels. As a result, the energy barrier for water molecules to hop through the membrane drops, allowing them to move faster while salt ions remain too bulky to follow. 
Performance Under Tough Real-World Conditions
When tested in pervaporation, the optimized MOF-801 membranes achieved near-perfect salt rejection of about 99.9% while delivering high water fluxes that surpass many state-of-the-art silica, zeolite, and other MOF membranes. Remarkably, this performance held across a wide range of feed salinities—from seawater levels up to very concentrated brines—and even at close to room temperature, where many thermal processes become inefficient. The membranes also showed impressive durability: they withstood long-term operation at elevated temperatures, in acidic and oxidizing environments, and when exposed to real industrial wastewater containing multiple ions, oils, and surfactants. Even after extended exposure to chlorine, which rapidly damages common polymer desalination membranes, the MOF-801 films retained their structure and separation performance.
What This Means for Future Water Treatment
In simple terms, this work shows that carefully “editing” the internal structure of a crystalline filter—by deliberately removing a controlled number of tiny building blocks—can dramatically improve how quickly water passes through without sacrificing purity. The resulting MOF-801 membranes are not only highly selective and fast but also rugged enough for harsh industrial settings. This strategy of tuning missing linkers offers a blueprint for designing next-generation nanochannel membranes that can tackle some of the most challenging water purification problems, from hypersaline brines to complex industrial waste streams.
Citation: Dong, Y., De Finnda, C., Fu, M. et al. Precise regulation of missing linkers in MOF pervaporation membranes for desalination of hypersaline waters. Nat Commun 17, 3206 (2026). https://doi.org/10.1038/s41467-026-69745-x
Keywords: hypersaline desalination, metal-organic frameworks, pervaporation membranes, water purification, nanochannel transport