Clear Sky Science · en
Cross-domain metabolic interactions link Methanobrevibacter smithii to colorectal cancer microbial ecosystems
Why tiny gut neighbors matter for colon health
The human gut is home to trillions of microbes that help digest food, train the immune system, and shape overall health. Most research has focused on bacteria, but another lesser‑known group, the archaea, has quietly lived alongside us for millennia. This study shines a spotlight on one common archaeal species, Methanobrevibacter smithii, and uncovers how its close partnership with certain bacteria may influence the risk and behavior of colorectal cancer, one of the most common cancers worldwide.
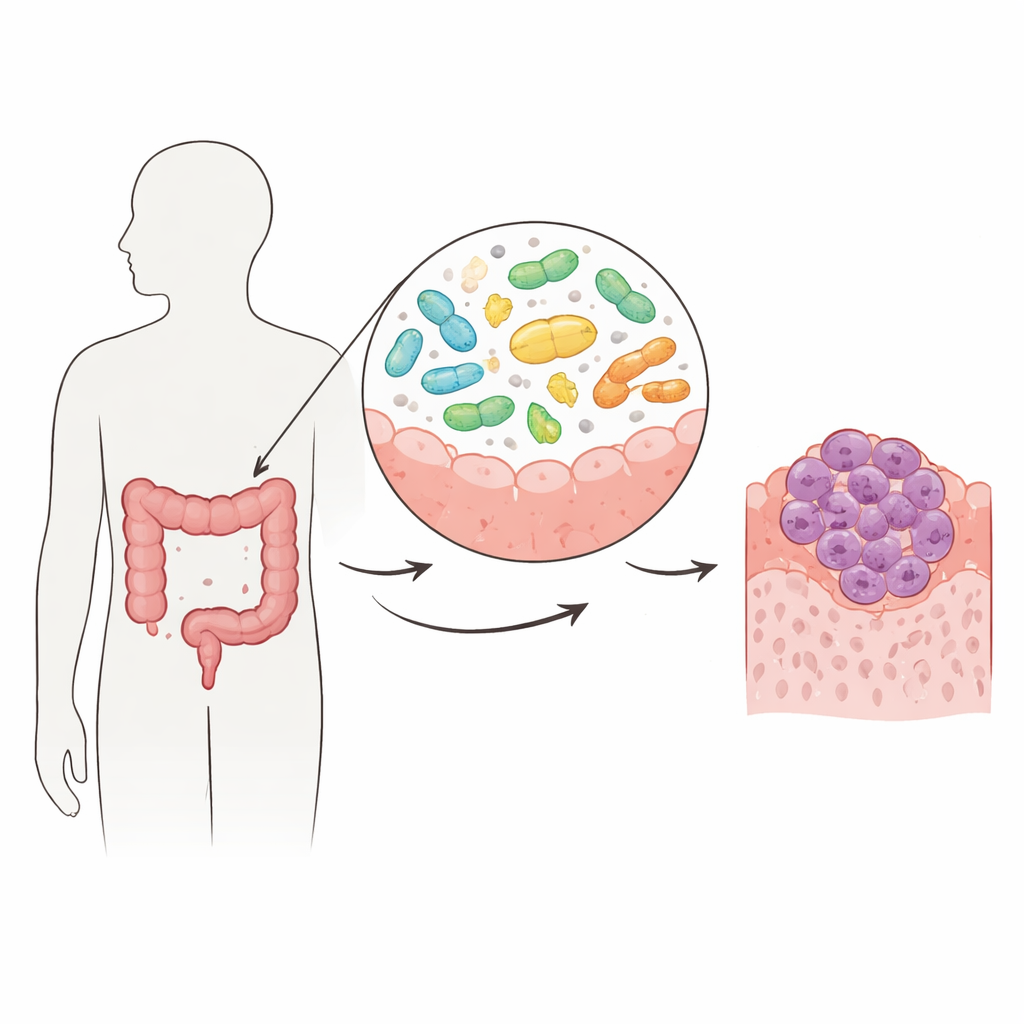
A broad look across many patient groups
To understand where archaea fit into human disease, the researchers reanalyzed nearly 3,000 previously published stool metagenomes from 19 studies across 12 countries. These datasets covered colorectal cancer, inflammatory bowel disease, type 2 diabetes, multiple sclerosis, Parkinson’s disease, schizophrenia, and early Alzheimer’s disease. Unlike older methods that detect only broad microbial groups, shotgun metagenomics allowed the team to track individual archaeal species. They applied a standardized pipeline so that differences between diseases would not simply reflect different lab techniques or sequencing platforms.
Finding a recurring archaeal signal in colon cancer
Across all these conditions, archaea in the gut were dominated by Methanobrevibacter species, especially M. smithii, which is usually regarded as a harmless resident. The team asked whether any archaeal species appeared more often in patients than in controls. In Crohn’s disease, many Methanobrevibacter species were depleted, suggesting that they may be lost when the gut is inflamed. In contrast, colorectal cancer showed the opposite trend: M. smithii and some close relatives tended to be more abundant in patients, and this pattern held across multiple independent cohorts. When the authors built a machine‑learning model to distinguish colorectal cancer from healthy microbiomes, M. smithii emerged among the top features helping to separate the two groups.
Microbial teamwork and shared fuel
M. smithii survives by using gases produced by its bacterial neighbors, but the study shows that its partnerships go far beyond this simple exchange. Using genome‑scale metabolic models, the researchers simulated how M. smithii might trade nutrients with twelve bacterial species already known to be linked to colorectal cancer, including Fusobacterium nucleatum, Bacteroides fragilis, and Escherichia coli. In every simulated pairing, the bacteria were predicted to release the organic acid succinate, which M. smithii could absorb and process. Succinate is more than just microbial fuel: in animal and cell studies, it can act as a signaling molecule that promotes tumor growth and weakens anti‑cancer immune defenses. The models also suggested extensive exchange of amino acids and vitamins, with M. smithii exporting riboflavin and consuming several amino acids that have been tied to tumor growth.
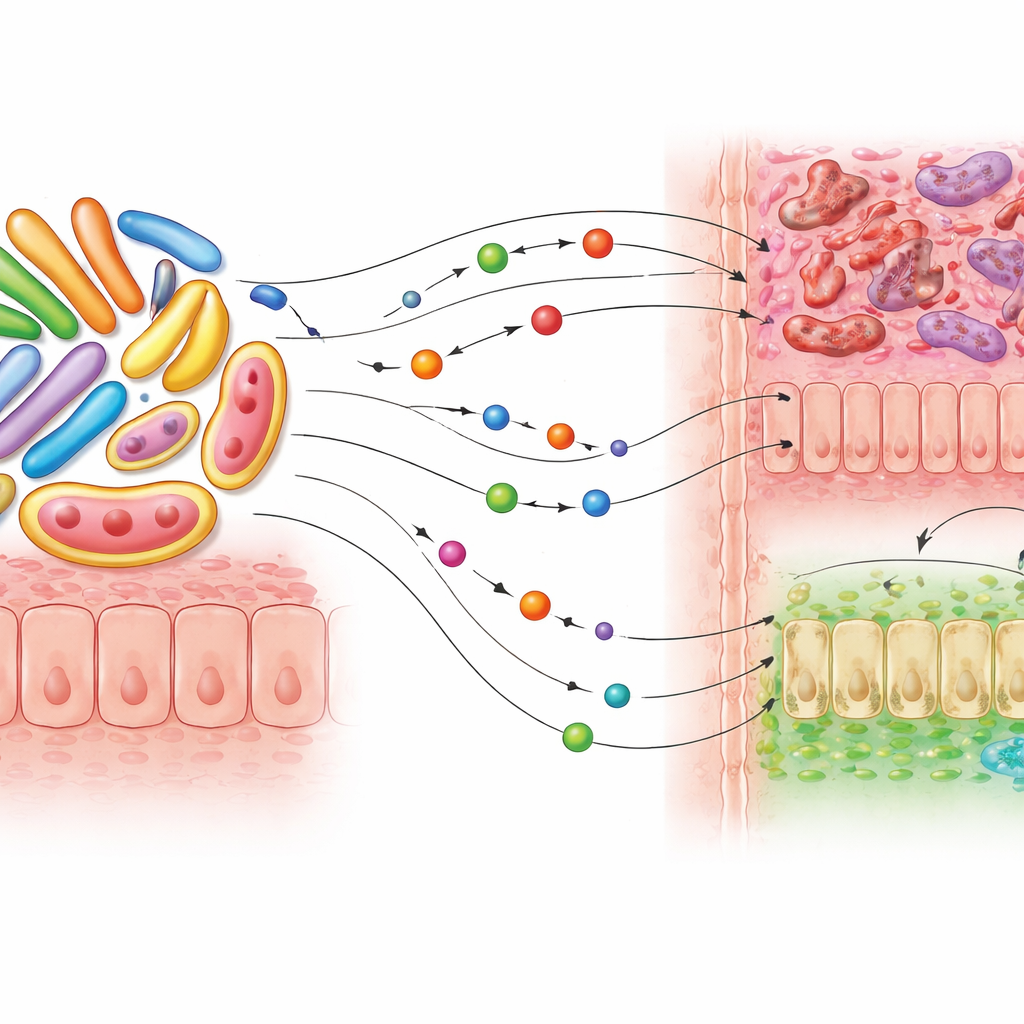
Laboratory tests of microbial partnerships
To move beyond computer predictions, the team co‑cultured M. smithii with three colorectal cancer‑associated bacteria under oxygen‑free conditions that mimic the large intestine. Microscopy showed the archaeon and bacteria growing in close contact. Growth measurements revealed that while M. smithii sometimes benefited modestly, certain bacteria, particularly F. nucleatum and E. coli, grew faster when the archaeon was present, hinting at one‑way or uneven support. Chemical profiling of the cultures confirmed that succinate levels were consistently higher in co‑cultures than in single‑species cultures, reinforcing the idea that this compound is a shared currency between partners.
A chemical mix of helpful and harmful molecules
When the researchers performed detailed mass‑spectrometry analysis on the liquid surrounding M. smithii and F. nucleatum grown together, they found a rich cocktail of small molecules. Many were amino acids and derivatives previously associated with colorectal cancer, as well as compounds involved in pathways that feed tumor growth, such as polyamine and purine metabolism. Intriguingly, the same samples also contained substances with potential anti‑tumor activity, including fatty molecules known to trigger cancer cell death or block tumor formation in experimental models. Some of these protective candidates, like certain fats and sphingolipids, were strongly linked to M. smithii itself, suggesting that archaea may supply both cancer‑supporting and cancer‑limiting molecules to the gut environment.
What this means for understanding colon cancer
Taken together, this work recasts M. smithii from a passive methane producer into an active player in colorectal cancer‑related microbial communities. Rather than acting alone, the archaeon forms dense networks with cancer‑associated bacteria, jointly shaping a chemical environment that can influence tumor growth and immune responses. The findings do not mean that M. smithii “causes” cancer, but they show that its presence, abundance, and metabolic exchanges could help tilt the gut ecosystem toward or away from disease. Future studies that track these interactions over time and in human tissues may reveal whether targeting archaeal–bacterial partnerships, or the key metabolites they trade, could open new avenues for early detection, prevention, or treatment of colorectal cancer.
Citation: Mohammadzadeh, R., Mahnert, A., Zurabishvili, T. et al. Cross-domain metabolic interactions link Methanobrevibacter smithii to colorectal cancer microbial ecosystems. Nat Commun 17, 2979 (2026). https://doi.org/10.1038/s41467-026-69711-7
Keywords: gut microbiome, archaea, colorectal cancer, microbial metabolism, Methanobrevibacter smithii