Clear Sky Science · en
Sub-5 nm high-entropy nanoalloys beyond the hume-rothery limit
Why tiny metal mixes matter
Modern electronics keep getting smaller and more powerful, but this progress comes with two big headaches: excess heat and stray electromagnetic signals that can scramble circuits. To tame both problems at once, engineers dream of ultrathin coatings that block electromagnetic interference (EMI) while also carrying heat away, much like a metal shell that is both a shield and a heat sink. This article describes a new class of ultra-small mixed-metal nanoparticles that break long-standing rules of alloy design and deliver exceptional electrical, thermal, and shielding performance in films thousands of times thinner than a human hair.
Old rules that limited metal mixing
For nearly a century, the Hume–Rothery rules have guided how different metals can be mixed into solid alloys. They state, in essence, that if the atoms are too different in size, the crystal lattice becomes unstable and the alloy tends to separate into different phases or even turn partly glassy. This size mismatch is captured by a quantity called δ: the larger the value, the harder it is to keep everything mixed. At the nanoscale, where particles are only a few billionths of a meter across, these rules bite even harder because surfaces, strain, and quantum effects make orderly packing more difficult. As a result, conventional synthesis methods have struggled to produce multi-metal nanoparticles with large size mismatches without them falling apart into multiple phases.
A new flash-forged recipe for complex metals
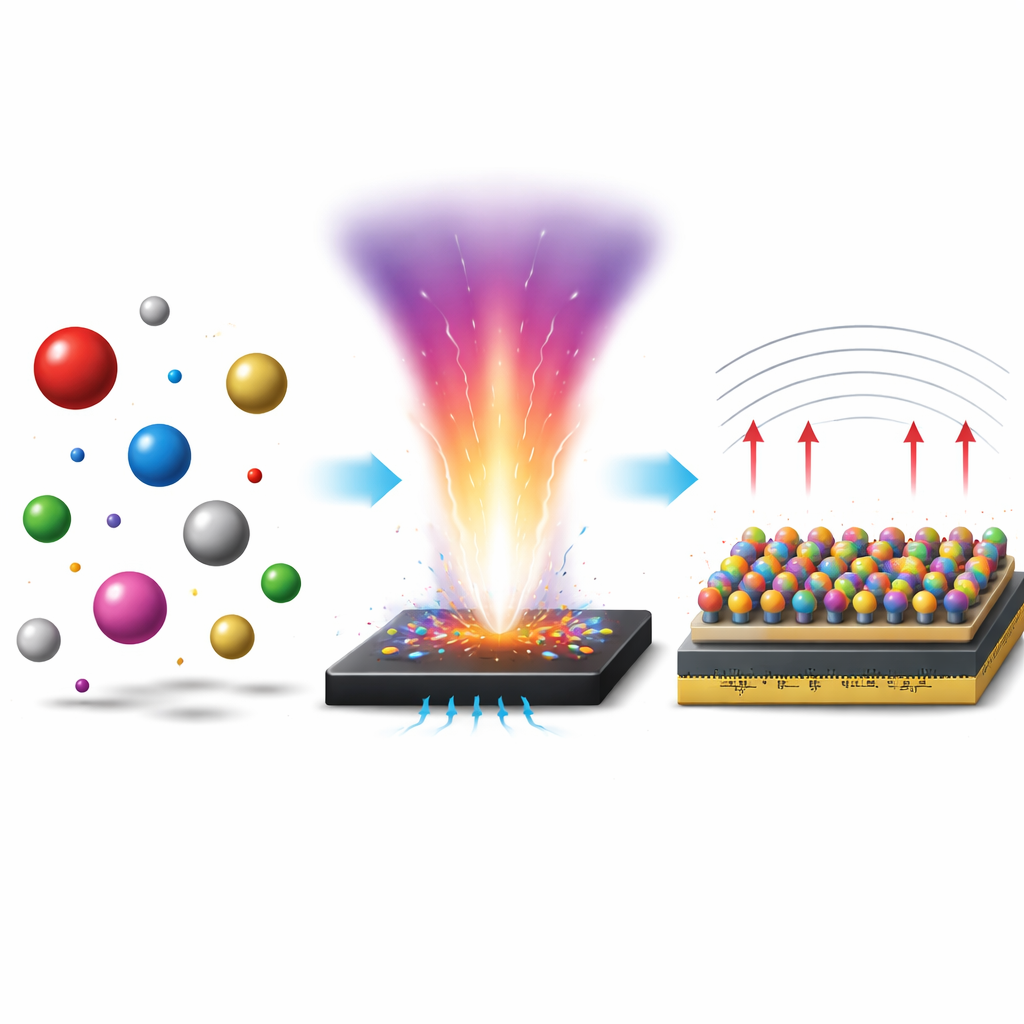
The researchers developed a new fabrication route called plasma-assisted carbothermal flash sintering (PCFS) to sidestep these limitations. They first anchor metal salts onto a carbon-based scaffold, then subject the system to extremely rapid heating and cooling cycles, with a brief plasma treatment in between. The plasma alters the electronic landscape at the surface, promoting charge transfer from the carbon support into the forming nanoparticles. This extra electronic disorder, combined with the ultrafast, non-equilibrium heating, allows many different metal types—including very large lanthanide atoms and small aluminum atoms—to be locked together before they can separate. By tuning the carbon surface properties and the temperature profile, the team can precisely control particle size, structure, composition, and the degree of atomic size mismatch.
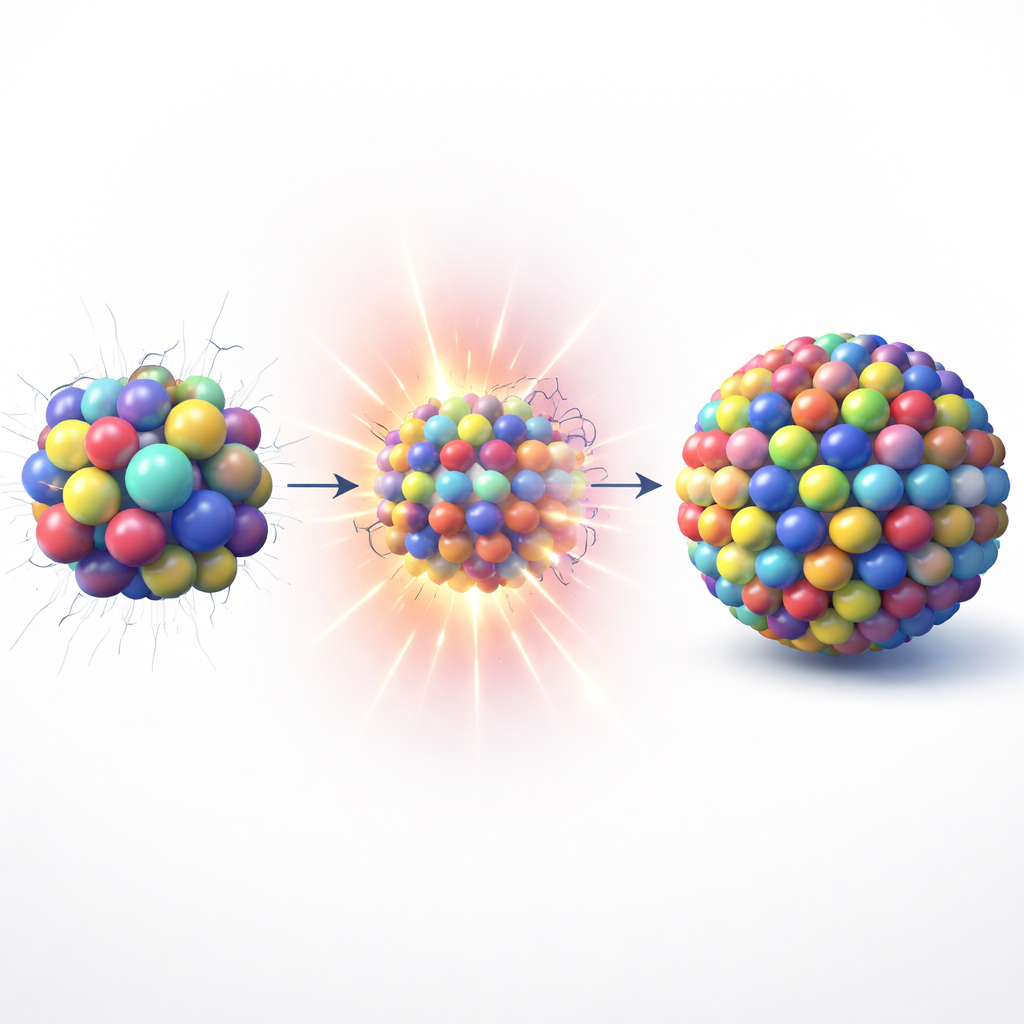
Orderly distortions inside tiny particles
At the heart of this work is a family of so‑called high-entropy alloys, in which several metals are mixed in roughly comparable amounts rather than having a single dominant ingredient. Using the PCFS method, the authors made sub‑5 nanometer particles based on iron, cobalt, and nickel, with smaller amounts of aluminum and a lanthanide element such as praseodymium. These particles achieve a very high size-mismatch parameter (δ > 15%), far beyond what is usually considered stable for such small structures. High‑resolution electron microscopy shows that the atoms are thoroughly mixed, but the lattice is not perfectly regular: it exhibits a quasi‑periodic pattern of gentle distortions that repeats in a controlled way from the surface inward. Instead of creating random defects and gaps, these “ordered distortions” relieve stress and preserve a single, well-connected metal lattice.
Unexpected boosts to conductivity and heat flow
Ordinarily, shrinking metals down to nanoparticles breaks up the continuous electronic states that carry charge and heat, making both conductivity and thermal transport worse. In these new high‑entropy particles, the opposite happens. Measurements show that their electrical conductivity rivals or exceeds that of many bulk metals and advanced carbon materials, even though the particles are only a few nanometers across. Theory and simulation suggest that the strong size mismatch and the quasi‑periodic distortions flatten the electronic energy bands and crowd many states near the energy level where electrons conduct. This creates multiple parallel pathways for electrons and also supports more efficient vibration modes that carry heat. As a result, thin films assembled from these particles maintain high electrical and thermal conductivities over a wide temperature range.
Ultra-thin shields for future chips
To demonstrate a practical use, the team dispersed a small fraction of these nanoparticles into a silicone matrix to form a flexible coating. With only 10% by weight of particles and a thickness of about 1.8 micrometers, the film blocks roughly 99–99.9% of electromagnetic radiation in the 2–6 GHz range, which is relevant for 5G communication. This performance usually demands materials that are tens to thousands of micrometers thick. At the same time, the composite’s thermal conductivity far surpasses that of typical polymer-based heat spreaders. When applied over working graphics chips, the coating kept temperatures dramatically lower than comparable films based on more conventional alloys, indicating better heat removal as well as shielding.
What this means for everyday technology
In simple terms, the authors have found a way to mix “incompatible” metals into ultra-small particles that behave better than expected: they conduct electricity and heat extremely well and form ultra-thin layers that shield delicate electronics from electromagnetic noise. By breaking through the traditional size-mismatch limit, their method opens up a huge space of possible metal combinations and properties. This could translate into slimmer, cooler, and more reliable phones, computers, and other devices, where protective layers must be both very thin and highly effective at handling heat and electromagnetic clutter.
Citation: Du, Y., Zhou, X., Li, B. et al. Sub-5 nm high-entropy nanoalloys beyond the hume-rothery limit. Nat Commun 17, 4051 (2026). https://doi.org/10.1038/s41467-026-69681-w
Keywords: high-entropy alloys, nanoparticles, electromagnetic shielding, thermal management, advanced electronics