Clear Sky Science · en
Tumor-associated CD19+ macrophages induce immunosuppressive microenvironment in hepatocellular carcinoma
Why the Body’s Defenders Sometimes Help Liver Cancer
Liver cancer is one of the deadliest cancers worldwide, in part because tumors can turn the body’s own immune system against itself. This study uncovers a surprising subset of immune cells inside liver tumors that, instead of fighting cancer, actively help it hide and grow. Understanding who these “traitor” cells are and how they work opens new paths for making immunotherapy more effective for people with hepatocellular carcinoma, the most common form of liver cancer.
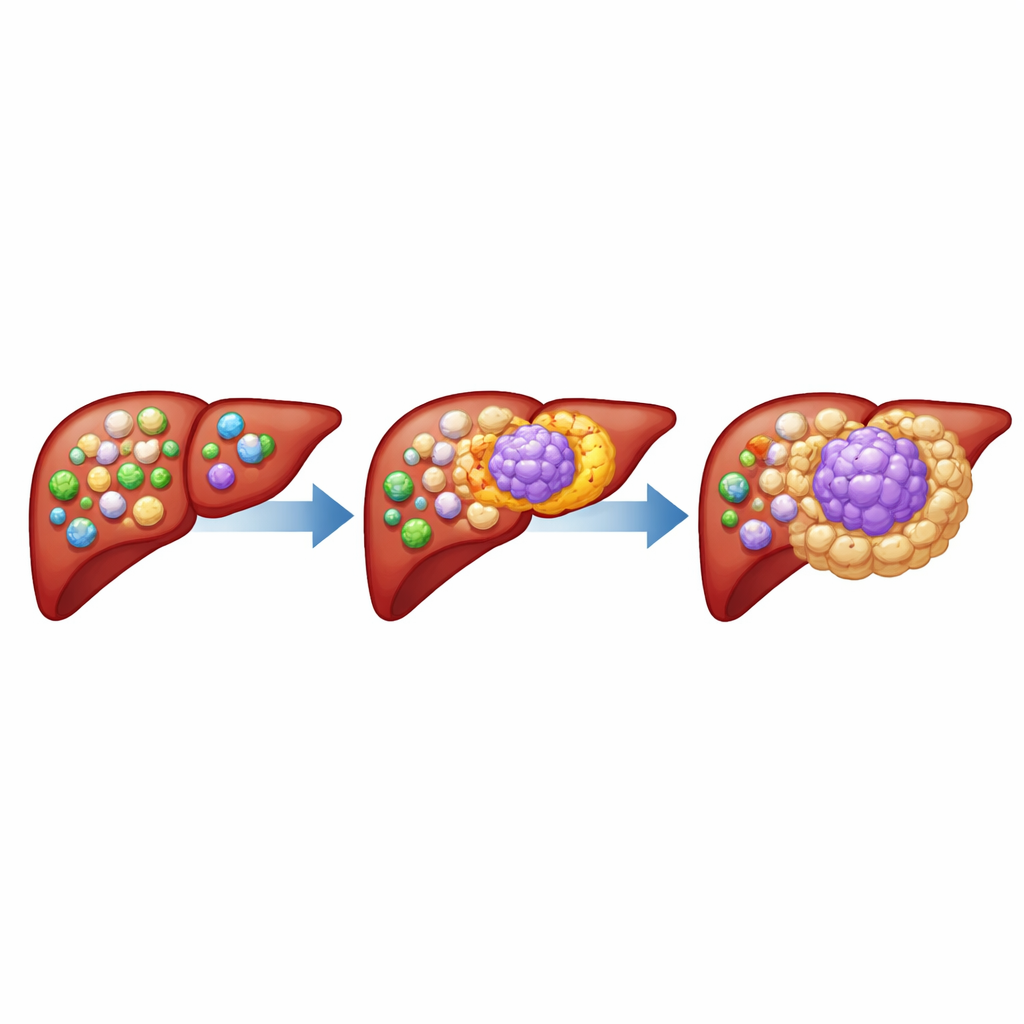
A Special Group of Immune Cells Inside Tumors
The authors focused on tumor-associated macrophages, immune cells that normally engulf germs and debris but often become corrupted inside cancers. By analyzing liver tumor samples from many patients, they discovered a distinct subgroup of macrophages that carry a surface molecule called CD19, usually found on B cells rather than macrophages. These CD19-positive macrophages were much more abundant in liver tumors than in nearby healthy liver or blood, and were also enriched in several other solid cancers. Single-cell analyses confirmed that these cells are true macrophages with a unique gene and protein profile, not mis-identified B cells.
How These Cells Shape a Tumor-Friendly Neighborhood
Patients whose tumors contained many CD19-positive macrophages tended to have larger, more aggressive cancers and shorter survival times. Tumor samples rich in these cells showed fewer cancer-killing CD8 T cells and more regulatory T cells, a pattern typical of a highly immunosuppressive environment. In mouse models, adding CD19-positive macrophages to liver cancer cells made tumors grow faster and reduced the presence of helpful T cells, while increasing other suppressive myeloid cells. Importantly, when the researchers used engineered T cells that recognize CD19 to selectively eliminate these macrophages, liver tumors shrank even in mice that lacked normal B cells, pinpointing the macrophages themselves as therapeutic targets.
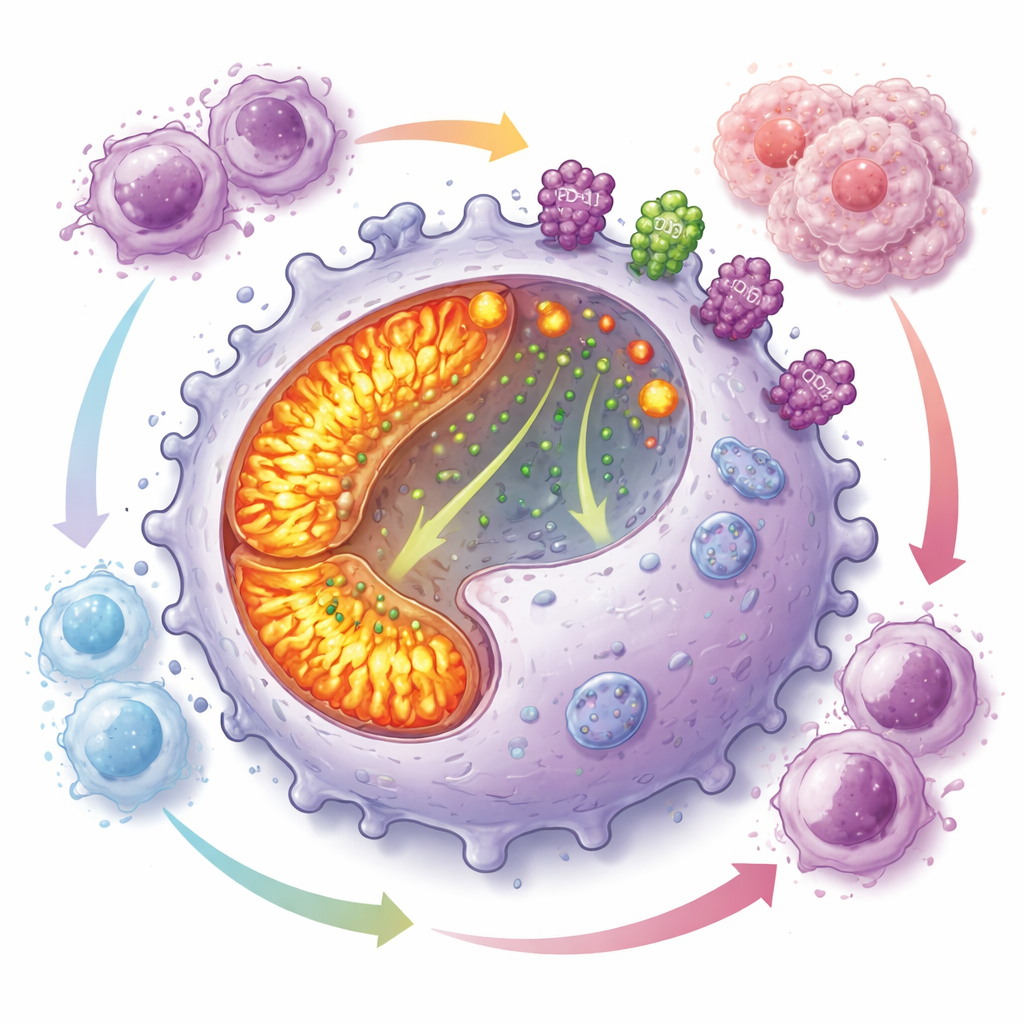
An Energy-Hungry Program That Blunts Immune Attack
Digging deeper, the team found that CD19-positive macrophages run their internal “power plants” at high gear. Their mitochondria, the organelles that generate energy, were more numerous and more active than in other macrophages, with increased oxidative phosphorylation and reactive oxygen species. Despite this energetic state, these cells had poor ability to engulf targets but divided rapidly, helping them accumulate in tumors. At the same time, they displayed high levels of PD-L1 and CD73 on their surface—two molecules known to shut down T cell responses and generate adenosine, a small molecule that powerfully dampens immunity. Co-culture experiments showed that these macrophages strongly inhibited the growth of multiple T cell types.
The Master Switch Behind the Traitor Macrophages
The researchers identified a transcription factor called PAX5 as the central driver of this harmful macrophage program. PAX5 is best known for guiding B cell development, yet here it was highly active in CD19-positive macrophages. Forcing macrophage-like cells to express PAX5 boosted mitochondrial biogenesis, increased energy production, and raised protein—but not mRNA—levels of PD-L1, CD73, and CD19. Mechanistic experiments showed that PAX5 pulls calcium into mitochondria, lowering calcium in the cell fluid and preventing a key regulator called TFEB from entering the nucleus. With TFEB held back, lysosome production and function drop, so proteins like PD-L1 and CD73 are no longer efficiently broken down and instead accumulate on the cell surface, intensifying immune suppression.
Turning Discovery into Better Treatment Options
Because these macrophages depend so heavily on CD73 activity and mitochondrial metabolism, the authors tested whether blocking these pathways could make immunotherapy work better. In mouse liver cancer, combining PD-L1 blockade with either CD73 antibodies or a CD73 inhibitor led to smaller tumors, more infiltrating T cells, and less cancer cell proliferation. Similarly, pairing PD-L1 antibodies with an inhibitor of mitochondrial oxidative phosphorylation improved tumor control and boosted immune cell entry into tumors. Strikingly, these benefits disappeared in mice whose macrophages lacked PAX5, confirming that the drug effects were tied to disabling the CD19-positive, PAX5-driven macrophage program. Together, the work suggests that selectively targeting this macrophage subset—via CD19-directed cell therapies, CD73 inhibition, or metabolic drugs—could help convert an immunologically “cold” liver tumor into one that responds much better to existing checkpoint blockade treatments.
Citation: Wang, J., Cao, W., Huang, J. et al. Tumor-associated CD19+ macrophages induce immunosuppressive microenvironment in hepatocellular carcinoma. Nat Commun 17, 3250 (2026). https://doi.org/10.1038/s41467-026-69638-z
Keywords: hepatocellular carcinoma, tumor-associated macrophages, immunotherapy, PD-L1 and CD73, mitochondrial metabolism