Clear Sky Science · en
Photo-enzyme-membrane for ethylene glycol synthesis
Turning Light into Useful Chemicals
Modern society relies on chemicals made from fossil fuels, which release carbon dioxide and worsen climate change. Scientists are searching for cleaner ways to make these same products using renewable energy like sunlight and simple starting materials such as methanol, which can be made from captured carbon. This paper introduces a sunlight-powered membrane system that works together with natural enzymes to make ethylene glycol, a common ingredient in antifreeze and plastics, from simple one‑carbon molecules in a more sustainable way.
Why Ethylene Glycol Matters
Ethylene glycol is a small, two‑carbon molecule used in coolants, polyester fibers, and many everyday products. Today it is mostly produced from oil or natural gas in energy‑intensive factories. The authors aim to build a different route: a "biomanufacturing" platform that uses enzymes—the same kinds of catalysts found in living cells—and sunlight to drive the reaction. Their vision is to turn basic carbon sources such as methanol into more complex, value‑added chemicals without relying on fossil fuels or high temperatures and pressures.
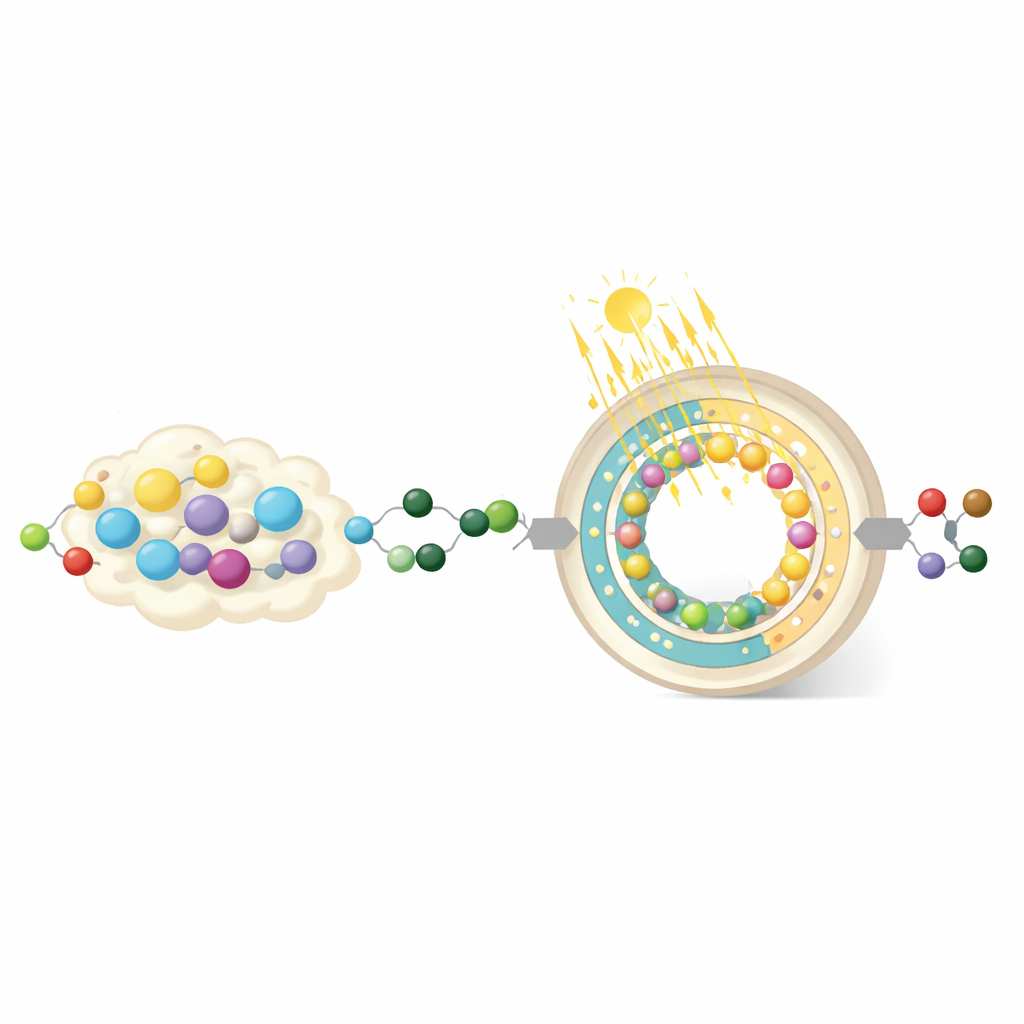
Building a Light-Driven Membrane
At the heart of the work is a tiny hollow particle called a photo‑enzyme‑membrane, or PEM. It has two main layers that cooperate. The inner layer, called the photo‑membrane, is made from an organic polymer that absorbs light and converts it into a chemical "fuel" molecule known as NADH. NADH is a natural energy carrier in biology that shuttles electrons and protons to enzymes so they can perform difficult reactions. The researchers carefully designed the membrane’s building blocks so that the movement of electrons and protons is well balanced, which is crucial for making the right form of NADH efficiently instead of wasteful by‑products.
Protecting Fragile Enzymes
The outer layer of the PEM, called the enzyme‑membrane, holds a key enzyme called alcohol dehydrogenase, which uses NADH to turn an intermediate compound into ethylene glycol. However, the inner light‑harvesting chemistry can also create highly reactive oxygen by‑products that normally damage enzymes. To prevent this, the team coats the enzyme with a carefully controlled silica shell, much like a thin glass armor. By tuning the thickness of this shell, they find a sweet spot where the harmful species are blocked or weakened, but small molecules like NADH and the reaction intermediates can still reach the enzyme. Under the best conditions, this protection boosts ethylene glycol production more than fivefold compared with an unprotected setup.
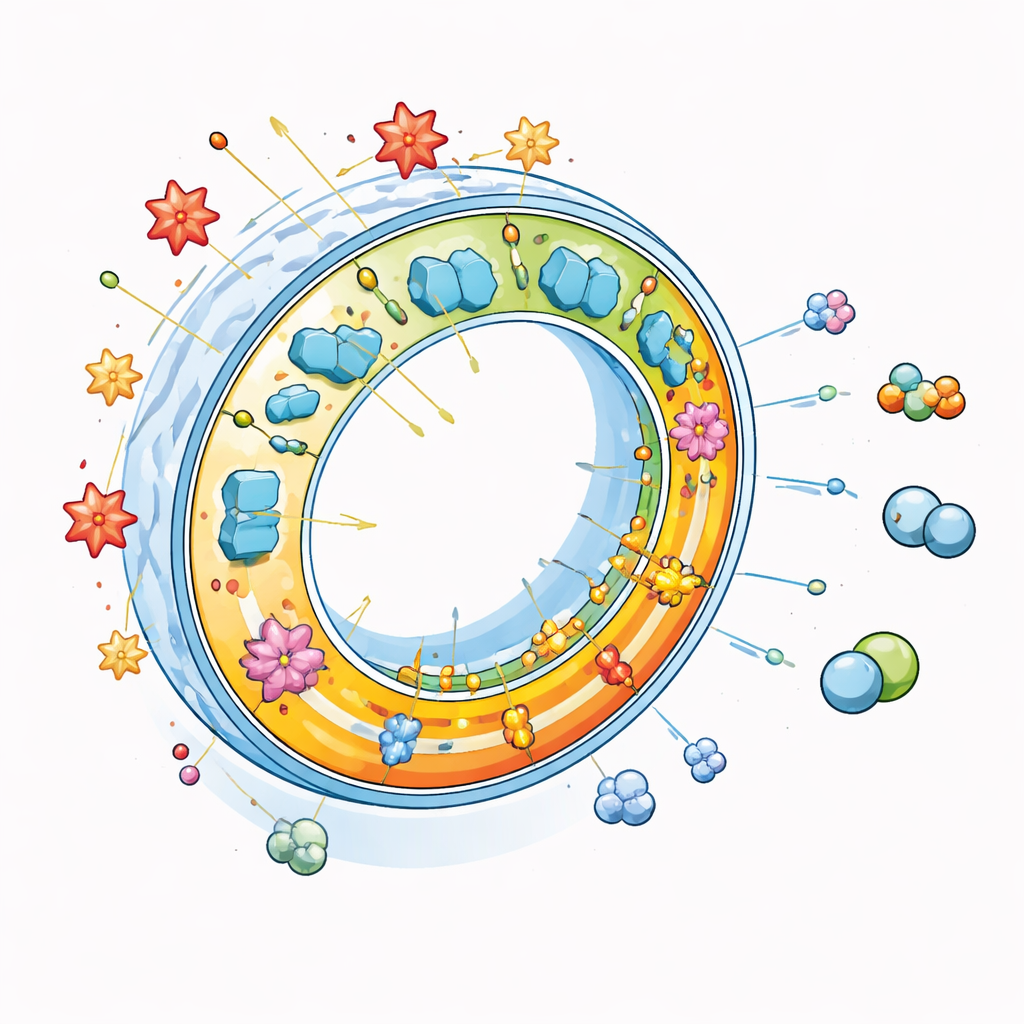
From Simple Alcohol to Valuable Product
To turn simple methanol into ethylene glycol, the authors combine the PEM with three additional enzymes in a sequence, or cascade. First, one enzyme oxidizes methanol into formaldehyde while another safely removes the hydrogen peroxide that would otherwise accumulate and cause damage. A third enzyme then links two molecules of formaldehyde into glycolaldehyde, a two‑carbon intermediate. Finally, the PEM uses sunlight to regenerate NADH and drive the last step, turning glycolaldehyde into ethylene glycol. Because methanol can harm enzymes if added all at once, the team designs a dual‑channel reactor that feeds methanol continuously and physically separates the early enzyme steps from the light‑driven membrane, while still allowing the intermediate molecules to pass between them.
A Step Toward Greener Chemical Production
In long‑running tests, the system steadily produces ethylene glycol with good efficiency while using light as the main energy input and small amounts of cofactor. Although the current output is modest compared with large industrial plants, the work demonstrates a powerful concept: carefully engineered membranes can coordinate light absorption, energy storage, and enzyme protection in a single, reusable particle. To a non‑specialist, the key message is that we can begin to mimic and extend nature’s own strategies—using sunlight, gentle conditions, and enzymes—to make important chemicals from simple carbon‑based starting materials in a cleaner and potentially more sustainable way.
Citation: Chen, Y., Sun, Y., Zhang, S. et al. Photo-enzyme-membrane for ethylene glycol synthesis. Nat Commun 17, 2814 (2026). https://doi.org/10.1038/s41467-026-69637-0
Keywords: photoenzymatic catalysis, ethylene glycol, biomanufacturing, solar-powered chemistry, multi-enzyme cascade