Clear Sky Science · en
A universal strategy towards self-healing materials via dynamic interfacial liquid metal coordination
Why Fixing Materials That Break Matters
From smartphone screens to wearable sensors and electric cars, modern life depends on soft plastics and rubbers that inevitably crack or tear over time. Once damaged, many of these materials must be replaced, wasting energy, money, and resources. This study introduces a clever way to make a wide range of plastics and rubbers capable of healing themselves after damage, while also carrying heat away from hot electronics like computer chips.
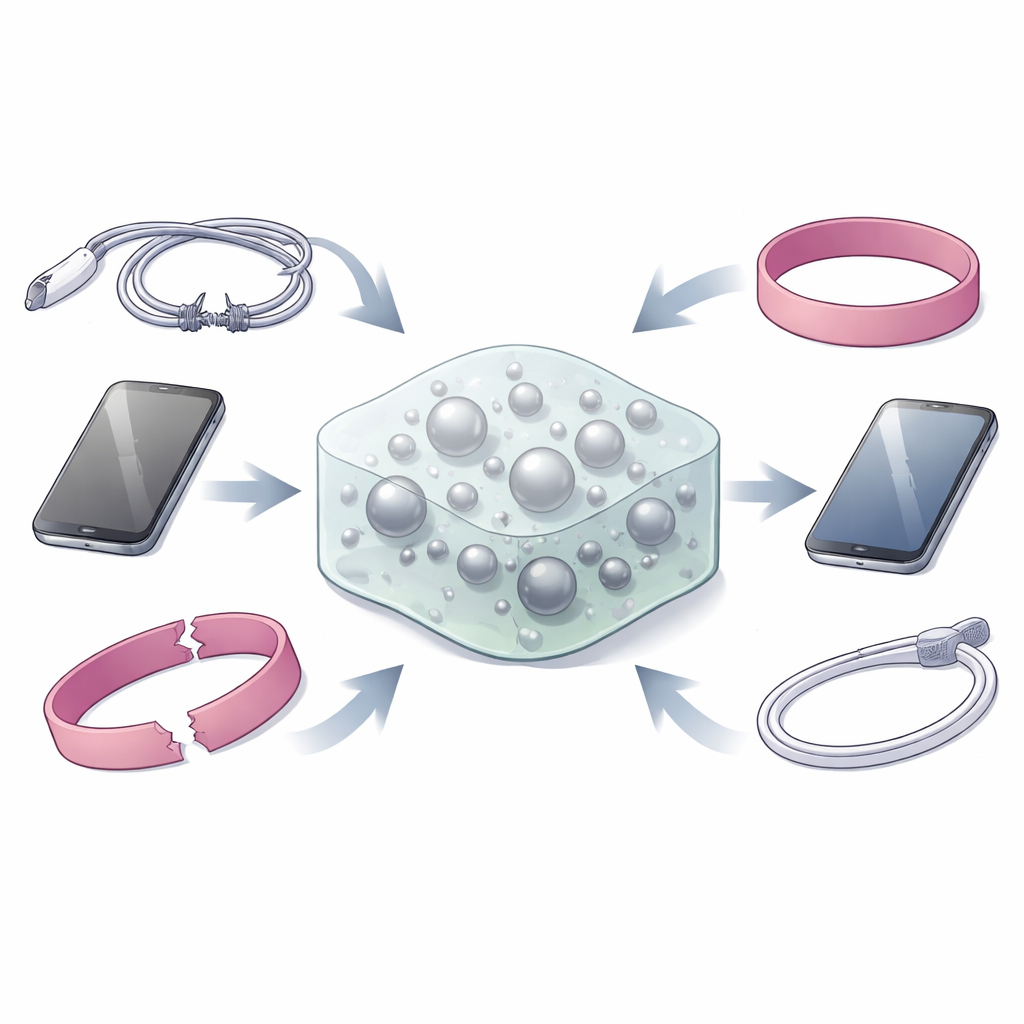
Turning Metals into Liquid Bandages
Many self-healing materials rely on reversible chemical links that can break and reform. Metal-based links are especially appealing because their strength and responsiveness can be tuned. Yet, most metal–based bonds are either too strong, locking materials into rigid, non-healable networks, or too weak to hold a solid together. The authors solve this dilemma by dissolving small amounts of “active” metals such as silver, zinc, and copper into gallium, a metal that is liquid near room temperature. The result is a multi-component liquid metal that forms tiny droplets dispersed inside a plastic or rubber. These droplets sit at the boundary between liquid and solid, creating a dynamic, mobile interface where metal atoms can still bond to the surrounding material but can also move and rearrange when the material is damaged.
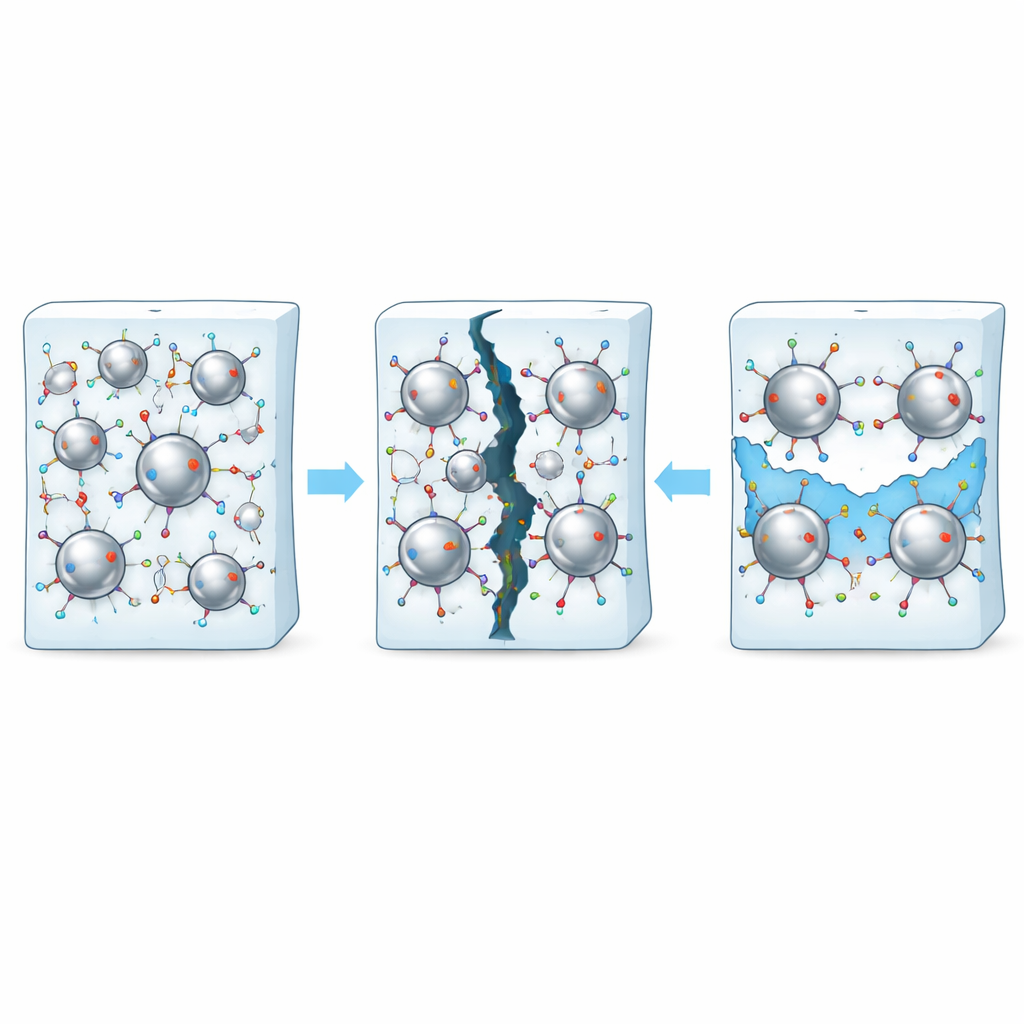
How Cracks Close Themselves
At rest, the active metal atoms at the surface of each liquid metal droplet loosely connect with chemical groups along the polymer chains, forming a network that gives the material strength. When the material is cut or stretched until it breaks, both the polymer and these interfacial connections are disrupted. Thanks to the fluid nature of the droplets, fresh metal atoms flow toward the damaged surface, renewing the interface and allowing new connections to form with nearby polymer chains. Over time, this process knits the two sides of a crack back together. In natural rubber filled with silver–gallium droplets, the healed samples recover more than 90 percent of their original mechanical performance at room temperature, far surpassing rubber alone or rubber filled only with solid silver or pure gallium.
A General Recipe for Many Plastics
The authors demonstrate that this liquid metal strategy is not limited to one particular rubber or one specific chemistry. They also embed zinc–gallium and copper–gallium droplets into acrylic-based plastics that carry imidazole groups, a common chemical motif. These composites likewise show healing efficiencies around 90 percent after being cut and left to rest, and can bear substantial loads once repaired. Microscopy, spectroscopy, and computer simulations reveal why: the active metal atoms are evenly distributed within each droplet and slightly polarized by the gallium environment, making them eager—but not too eager—to bond with sulfur or nitrogen atoms in the polymers. This balance produces bonds that are strong enough to hold, but loose enough to let the interface renew itself repeatedly.
Keeping Electronics Cool for the Long Haul
Because metals conduct heat extremely well, the team also turns these self-healing composites into thermal interface materials—thin films placed between a hot computer chip and a heat sink. Films packed with silver–gallium droplets reach thermal conductivities up to 6.8 W/m·K, far above that of the base rubber, yet remain electrically insulating. When used on a working central processing unit (CPU), these films lower the peak temperature by roughly 20–30 degrees Celsius compared with plain rubber, and they maintain this cooling performance even after repeated temperature swings between −10 and 100 degrees Celsius. Surface cracks that would normally degrade heat transfer instead gradually disappear as the film heals itself, keeping the chip safely within its operating range.
What This Means for Future Devices
In everyday terms, the study offers a broadly applicable recipe for making plastics and rubbers that can “self-stitch” after being damaged, without sacrificing strength or heat-handling ability. By using liquid metal droplets as movable anchors for metal-based bonds, the researchers convert otherwise irreversible connections into repairable ones. This universal strategy could lead to longer-lasting wearable electronics, safer batteries, and more reliable high-performance computers, while reducing waste and maintenance costs.
Citation: Li, Z., Zhang, Y., Liu, S. et al. A universal strategy towards self-healing materials via dynamic interfacial liquid metal coordination. Nat Commun 17, 2815 (2026). https://doi.org/10.1038/s41467-026-69609-4
Keywords: self-healing materials, liquid metals, polymer composites, thermal interface materials, soft electronics