Clear Sky Science · en
Engineering Co-ion vacancy in dynamically reconstructed Co-based catalysts for practical anion-exchange membrane electrolysis
Turning Water into Fuel More Efficiently
Clean hydrogen is a promising fuel for a low‑carbon future, but making it from water still costs too much energy. This study explores a smart way to improve one of the weakest links in water‑splitting devices: the material that helps pull oxygen out of water. By redesigning this material at the atomic level, the researchers show how to squeeze more hydrogen from water using less electricity and with far better durability, a key step toward affordable green hydrogen.
Why Better Oxygen Helpers Matter
Industrial water‑splitting devices, especially anion‑exchange membrane water electrolyzers, are attractive because they can use inexpensive, earth‑abundant materials instead of precious metals like iridium. However, on the oxygen‑producing side of these devices, most low‑cost catalysts either work too slowly or fall apart under harsh operating conditions. A cobalt‑based compound called cobalt oxyhydroxide is one of the more promising options, but even it faces a trade‑off: activating oxygen more strongly can speed up the reaction yet also damage the material’s structure over time. The central challenge is to design a catalyst that both accelerates oxygen release and heals itself as it operates.
Creating Helpful “Missing Atoms”
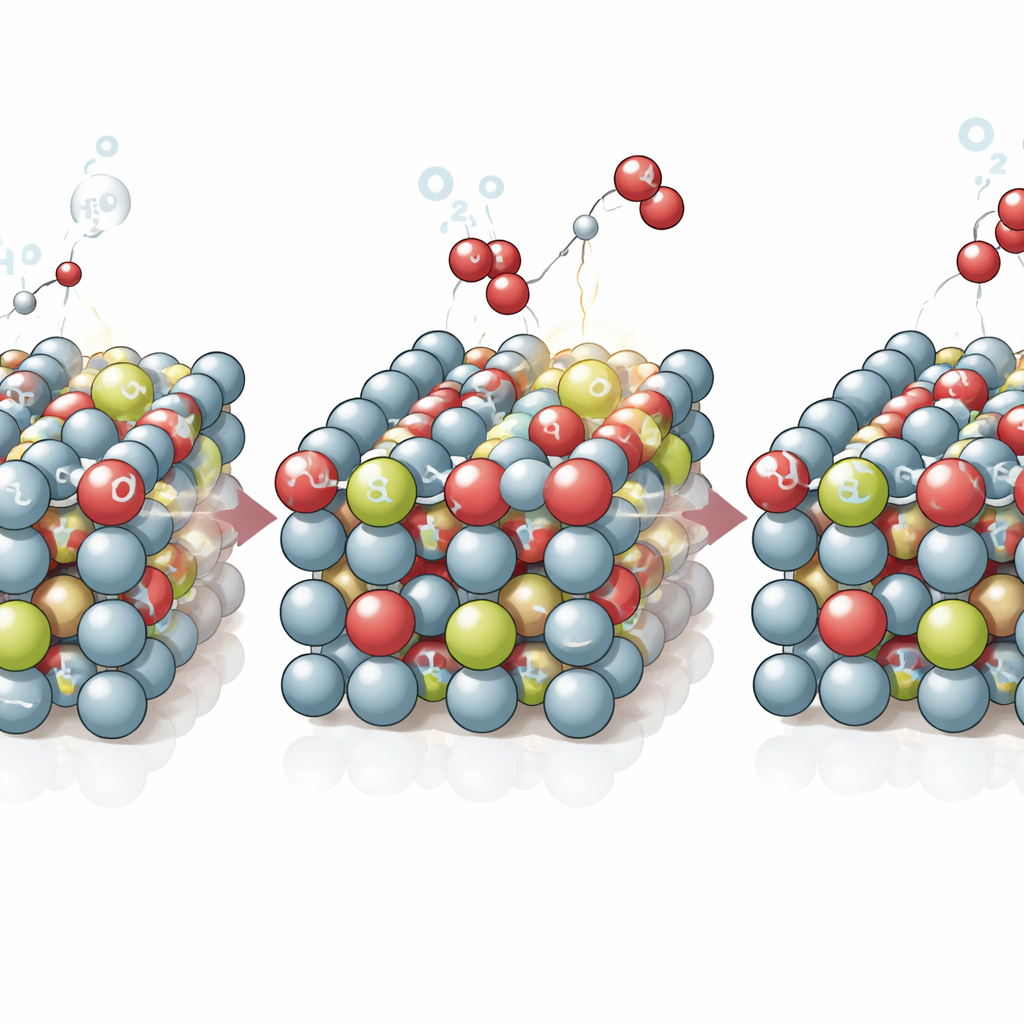
The team tackled this problem by deliberately introducing tiny imperfections—missing cobalt atoms—into thin sheets of the cobalt‑based catalyst. They first made belt‑shaped crystals of a compound containing cobalt, selenium, and a small amount of strontium. Computer calculations and X‑ray measurements showed that adding strontium weakened specific chemical bonds, making the structure easier to reorganize during operation. When these crystals were exposed to the conditions of the oxygen‑forming reaction, they transformed into cobalt oxyhydroxide nanosheets containing many well‑distributed cobalt vacancies, with strontium atoms remaining as stabilizing partners in the new structure.
How the Atomic Design Speeds the Reaction
Detailed experiments and computer simulations revealed why these intentionally missing atoms help. Around the vacancies, cobalt and oxygen share electrons more strongly, which makes it easier for oxygen within the solid itself to take part in the reaction. This activates an alternative reaction pathway in which oxygen atoms in the lattice work together with incoming water‑derived species to form oxygen gas more directly. At the same time, the altered electronic environment around the vacancies increases the attraction between the catalyst and incoming hydroxyl groups from the solution. These groups quickly refill the temporary oxygen gaps created during the reaction, preventing the structure from crumbling. In other words, the material is engineered to both release and replenish oxygen in a balanced cycle.
Performance in Realistic Devices
When tested in alkaline solution, the vacancy‑rich strontium–cobalt nanosheets generated oxygen at much higher currents and lower voltages than both plain cobalt oxyhydroxide and a commercial ruthenium oxide catalyst. Crucially, they kept this performance with almost no loss even after thousands of rapid start‑stop cycles, and the amount of cobalt lost to the solution remained very small. In a full anion‑exchange membrane electrolyzer operating at 80 °C with a commercial hydrogen‑producing electrode on the other side, the new catalyst delivered an industrial‑scale current of 3.3 amperes per square centimeter at just 2.0 volts, with lower energy use per kilogram of hydrogen than current technology targets and stable operation over 1,000 hours.
What This Means for Green Hydrogen
This work shows that carefully placing and stabilizing metal “missing spots” inside a catalyst can turn a structural weakness into a powerful design feature. By using strontium to guide the formation of cobalt vacancies that both activate oxygen and enable rapid self‑repair, the researchers created a low‑cost catalyst that is fast, efficient, and remarkably durable under realistic operating conditions. Such atom‑level engineering offers a blueprint for building the next generation of robust, high‑performance materials needed to make large‑scale green hydrogen production a practical reality.
Citation: Zhao, J., Li, X., Wang, K. et al. Engineering Co-ion vacancy in dynamically reconstructed Co-based catalysts for practical anion-exchange membrane electrolysis. Nat Commun 17, 2858 (2026). https://doi.org/10.1038/s41467-026-69547-1
Keywords: green hydrogen, water electrolysis, oxygen evolution catalyst, cobalt oxyhydroxide, defect engineering
See more on the researcher's website: https://www.xuzhaolab.com/news.php