Clear Sky Science · en
Co-upcycling spent lithium-ion batteries and plastics into microwave absorbing materials with Ni-Co catalyst control
Turning Old Batteries and Plastic Trash into Useful Shields
Mountains of worn-out electric car batteries and plastic packaging are two of today’s most worrying waste problems. This study shows a way to tackle both at once: by transforming spent lithium-ion batteries and mixed plastic trash into a new material that can soak up unwanted microwaves. Such materials are important for reducing electronic interference and improving stealth technologies, and this approach does so while cutting pollution and recovering valuable metals.
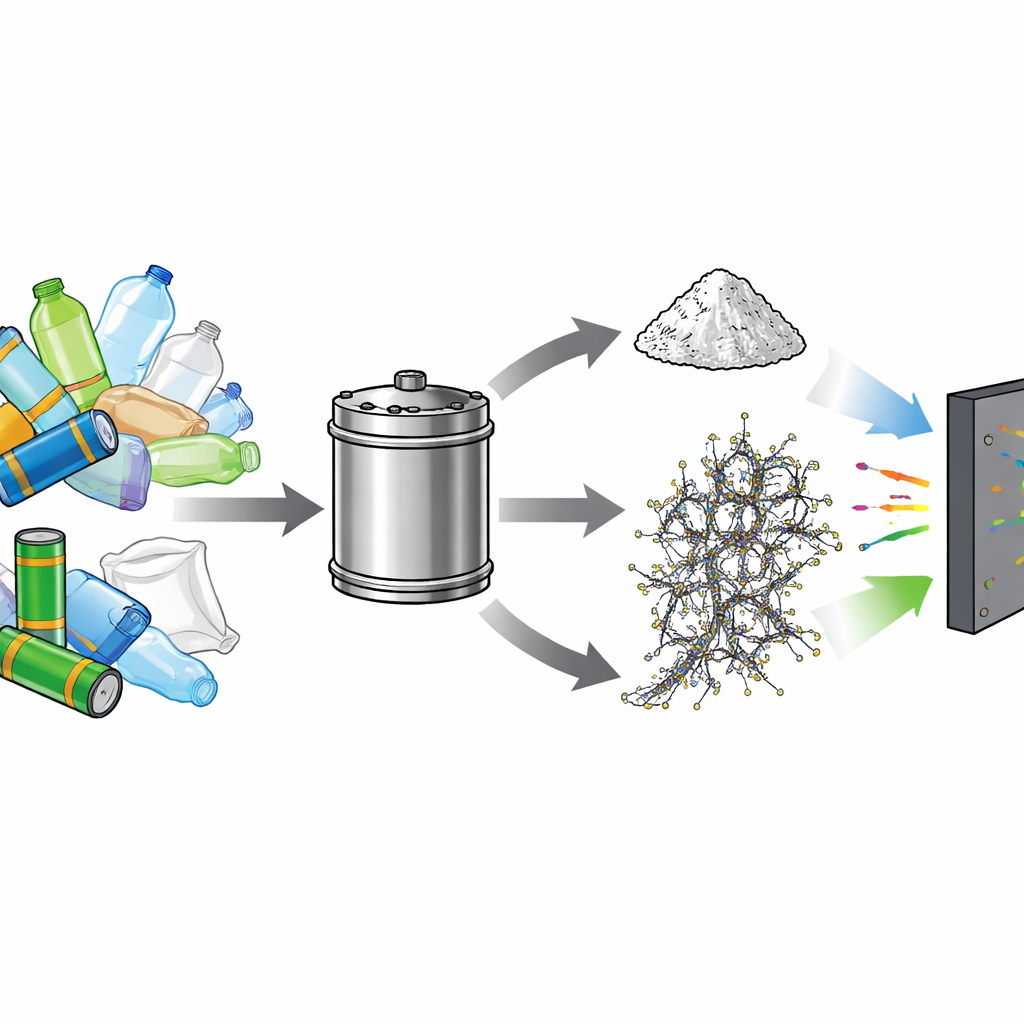
Why Battery and Plastic Waste Are Hard to Handle
Lithium-ion batteries in electric vehicles typically last only five to eight years before they must be replaced, leaving behind large volumes of used battery packs loaded with critical metals like nickel, cobalt, manganese, and lithium. At the same time, more than 380 million tons of plastic are produced each year, much of which resists decay and pollutes land and oceans. Traditional ways to deal with these wastes—such as burning plastics or smelting batteries—consume a lot of energy, release greenhouse gases and toxic fumes, and often merely recover bulk metals rather than creating higher-value products.
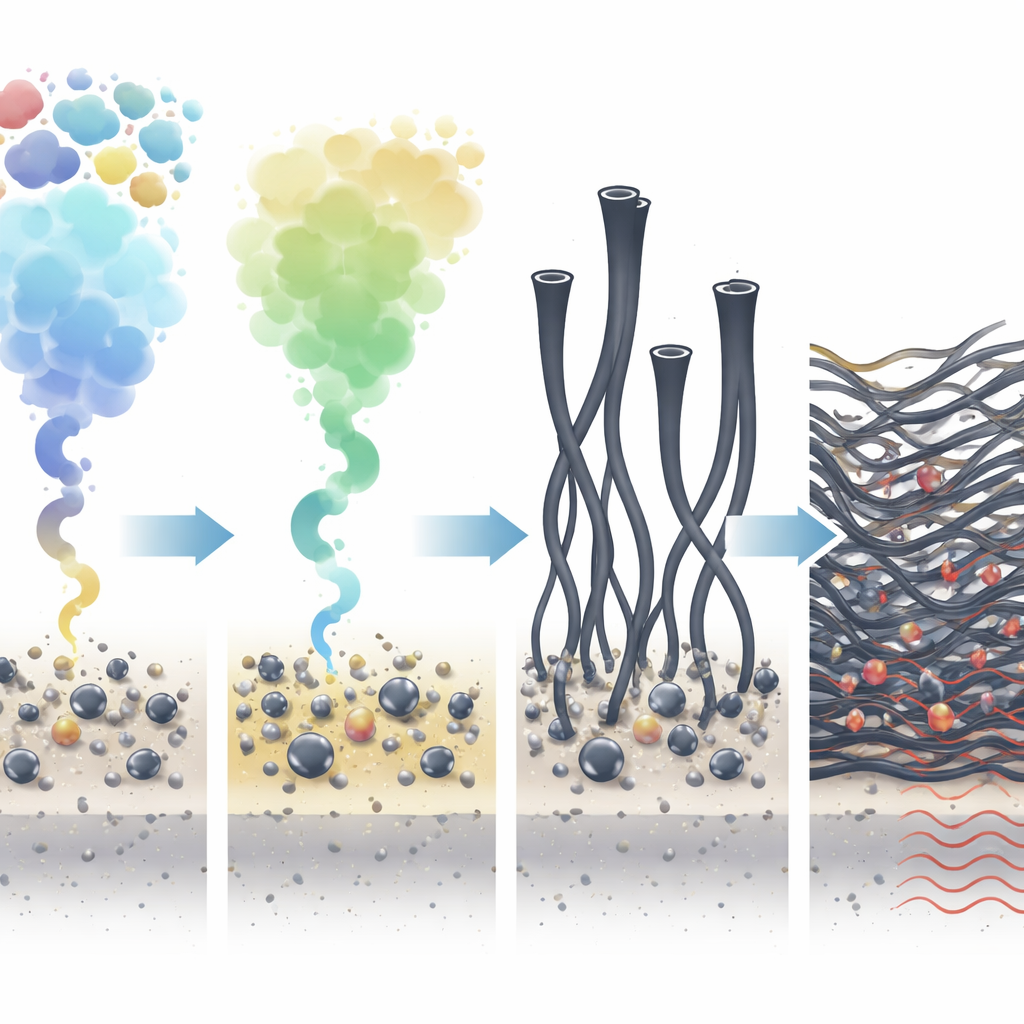
Cooking Waste Together to Make Tiny Tubes
The researchers designed a process in which shredded battery cathode powder and mixed plastics are heated together inside a closed steel reactor. A key ingredient is the common drink-bottle plastic polyethylene terephthalate, or PET, which is mixed with other plastics like polyethylene and polypropylene. When heated to around 550 °C, the plastics break down into gases that both reduce the metal oxides in the battery material and provide carbon. Nickel and cobalt atoms clump into very small particles, while lithium leaves as lithium carbonate that can later be washed out with water.
How PET Keeps the Metal Working
In many plastic-to-carbon processes, metal catalysts quickly “choke” as thick carbon layers coat their surface, stopping further reactions. Here, PET changes the mix of gases so that carbon does not simply pile up. Its breakdown produces carbon monoxide and carbon dioxide, which help strip away disordered carbon while still letting carbon-rich gases feed the growth of tidy carbon nanotubes. The lithium carbonate that forms also acts like a spacer, preventing nickel–cobalt particles from growing larger than about 100 nanometers. This size control keeps the metals highly active and guides the growth of dense forests of nanotubes intertwined with tiny metal and manganese oxide particles.
From Black Powder to Microwave Shield
After the initial “co-pyrolysis” step, the solid product is briefly heated again at about 800 °C in an inert atmosphere. This second treatment cleans off remaining fuzzy carbon and improves the ordering and electrical conductivity of the nanotubes. The final material is a lightweight composite where metal and metal-oxide particles are embedded in a conductive web of multiwalled carbon nanotubes. When tested across common radar and communication frequencies, this powder—mixed into a simple binder—shows strong microwave absorption. At a coating thickness of about 2.4 millimeters, it can absorb more than 90% of incoming waves over a 7-gigahertz-wide band, and maximum absorption depths are even higher.
Environmental and Economic Gains
Beyond lab performance, the team evaluated how this upcycling route compares with three major industrial recycling methods: high-temperature smelting, chemical leaching, and direct regeneration of cathodes. Using life cycle analysis and a battery-recycling model, they found that the co-pyrolysis approach consumes less energy and water and produces far fewer greenhouse gas emissions per kilogram of waste processed. It also avoids strong mineral acids, recovering lithium as lithium carbonate through simple water leaching. Because the end product is a high-value microwave-absorbing material, the process can, in their model, generate substantially higher profits than conventional recycling routes.
What This Means for Everyday Life
In simple terms, the study shows that yesterday’s electric car batteries and throwaway plastics can become tomorrow’s high-tech shields for managing electromagnetic waves. By smartly controlling how plastics break down and how metal particles behave during heating, the researchers turn mixed trash into a fine black powder that both recovers critical metals and performs as a strong microwave absorber. This offers a practical path to reduce waste, cut climate impact, and create advanced materials from resources that would otherwise be discarded.
Citation: Qiu, B., Hou, Y., Shi, Z. et al. Co-upcycling spent lithium-ion batteries and plastics into microwave absorbing materials with Ni-Co catalyst control. Nat Commun 17, 2822 (2026). https://doi.org/10.1038/s41467-026-69501-1
Keywords: lithium-ion battery recycling, plastic waste upcycling, carbon nanotubes, microwave absorbing materials, circular economy