Clear Sky Science · en
Strategies to control the geometry and symmetry around lanthanide centres for tailored luminescence and magnetism
Shaping Tiny Metals for Big Technologies
Lanthanides are a family of metals that quietly power modern life, from smartphone screens and medical scans to data storage and quantum devices. The color and magnetism they display depend not only on which element is used, but also on exactly how surrounding atoms arrange themselves around each metal ion. This paper shows how chemists can deliberately sculpt that tiny three‑dimensional environment, offering a practical roadmap for designing better materials for lighting, imaging, refrigeration, and future quantum technologies. 
The Hidden Architecture Around Lanthanide Ions
When a lanthanide ion sits inside a crystal or molecule, it is surrounded by other atoms that act like a cage. For many important compounds, eight atoms form this cage in different shapes, such as a cube, a twisted square, or a slightly rounded polyhedron. These shapes, and the symmetry with which atoms are arranged, strongly influence how the ion absorbs and emits light and how its magnetic moment behaves. The authors mined the Cambridge Structural Database, which contains over a million crystal structures, and extracted 12,670 examples where lanthanides sit in such eight‑atom environments. They then used mathematical tools to quantify how closely each cage matches six ideal shapes and how symmetric each site really is.
What the Data Reveal About Common Shapes
The survey shows that lanthanide ions rarely sit in wildly distorted surroundings. Most cages are very close to one of three low‑energy shapes that minimize crowding between ligands: the square antiprism, the dodecahedron, and the bicapped trigonal prism. Among these, the square antiprism dominates almost half the dataset and emerges as an especially favorable arrangement. Only a small fraction of structures form perfect cubes, hexagonal bipyramids, or more exotic “snub” shapes, and truly irregular cages are uncommon. The analysis also introduces a way to trace smooth transformation paths between ideal shapes, revealing, for example, how a cube can gradually twist into a square antiprism and how real compounds often sit partway along this path.
How Ligands, Size, and Temperature Steer the Shape
To turn statistics into design rules, the authors dissect how simple chemical features control geometry. They show that the size of the lanthanide ion itself matters: as one moves to smaller, heavier lanthanides, the increased crowding pushes ligands toward the square‑antiprismatic shape. The type of ligand and the size of its “bite” around the metal are equally crucial. Small, tight chelate rings tend to favor dodecahedral cages but inevitably introduce some distortion, while slightly larger rings better match square‑antiprismatic arrangements. Flexible ligands with longer reach can adapt to several shapes with minimal strain. In contrast, when only single‑tooth ligands are present, lanthanide ions naturally default to the three low‑energy shapes unless crystal packing or hydrogen bonding forces them into rarer geometries. Temperature usually nudges the geometry only slightly, although a few sensitive systems show noticeable reshaping as they are warmed.
Blueprints for Tailored Light and Magnetism
Because different shapes and symmetries favor different properties, the authors translate their findings into concrete strategies. Nearly perfect high‑symmetry environments, such as square antiprisms or hexagonal bipyramids, are ideal for creating robust single‑molecule magnets and spin‑based quantum bits, as they reduce unwanted quantum tunneling. Lower‑symmetry cages like dodecahedra or snub shapes relax optical selection rules, boosting the intensity and tunability of lanthanide luminescence for phosphors and upconverting nanoparticles. By choosing appropriate combinations of mono‑, bi‑, and multi‑dentate ligands, and by exploiting crystal packing and counter‑ions, chemists can selectively stabilize each of the six main geometries and fine‑tune their distortions. 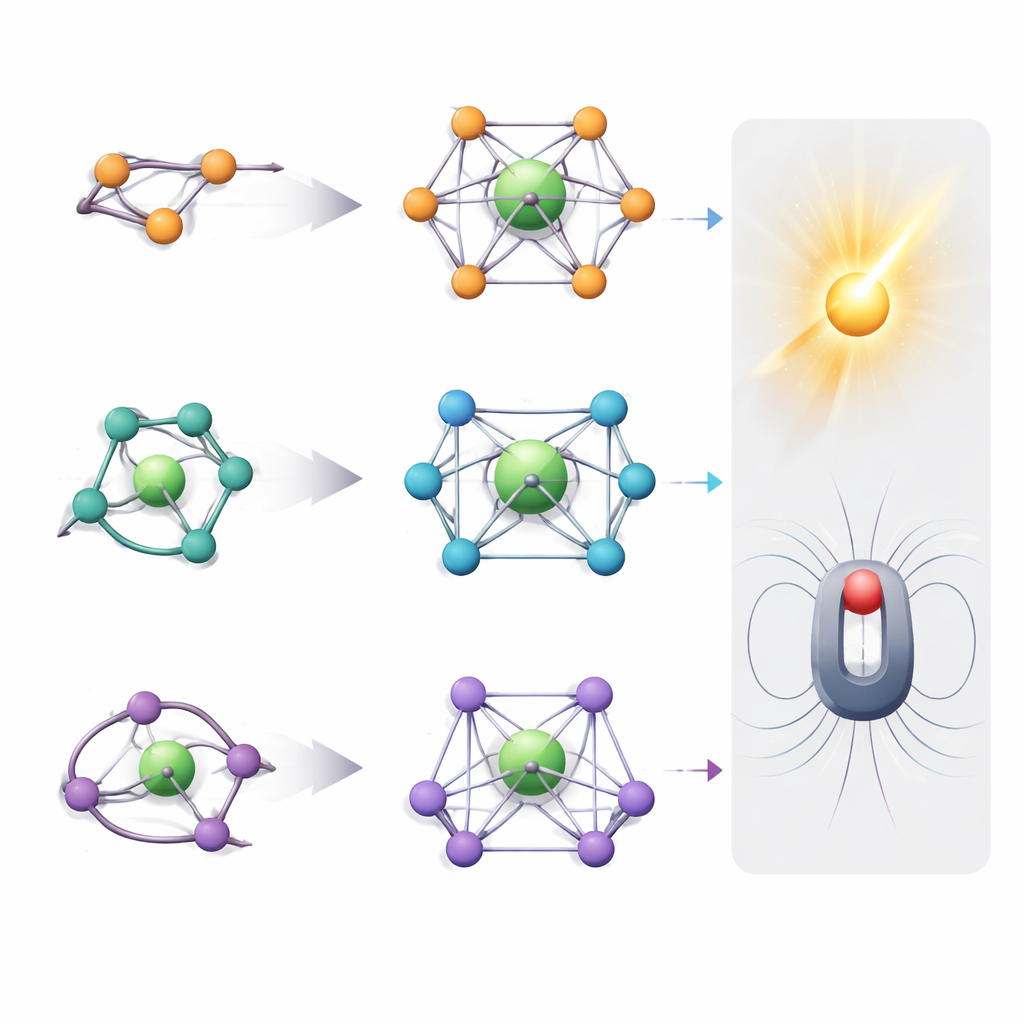
From Geometric Map to Practical Roadmap
In everyday terms, this work turns a messy catalog of crystal structures into a clear design manual. It links simple, adjustable chemical knobs—metal size, ligand shape and flexibility, and solid‑state packing—to predictable three‑dimensional cages around lanthanide ions. Those cages, in turn, govern whether a material glows brightly in a chosen color, stores magnetic information reliably, or behaves as a quantum bit. By mapping which ligand motifs lead to which geometries and symmetries, the study equips researchers to build lanthanide‑based materials with properties tailored to future needs in lighting, sensing, data storage, and quantum technologies.
Citation: Karpiuk, T.E., Leznoff, D.B. Strategies to control the geometry and symmetry around lanthanide centres for tailored luminescence and magnetism. Nat Commun 17, 2845 (2026). https://doi.org/10.1038/s41467-026-69445-6
Keywords: lanthanide coordination, molecular magnetism, luminescent materials, crystal structure analysis, quantum information