Clear Sky Science · en
Solventless, ambient-pressure production of bio-based lactones over earth-abundant, mixed metal oxide catalysts for circular polyesters
Turning Plant Sugars into Better Plastics
Most plastics today start from oil and gas and leave a heavy climate footprint. This study explores a cleaner way to make key building blocks for plastics using ingredients from plants instead of fossil fuels. The researchers show how to turn simple plant‑derived liquids into ring‑shaped molecules called lactones, which can be snapped together into recyclable polyesters, using only air, modest heat, and a catalyst made from common metals.
Why Plastic Building Blocks Matter
Making plastics is not just about the final product; producing the starting molecules, or monomers, consumes vast amounts of energy and releases large quantities of greenhouse gases. Polyesters are attractive because they can be designed for easy recycling, but their monomers are often made from fossil fuels using hot, energy‑hungry processes. A more sustainable route is to start from bio‑based diols, small alcohols already produced at industrial scale from plant sugars. Converting these diols into lactones creates ideal starting rings for making circular polyesters, but existing methods typically rely on expensive precious metals and organic solvents, limiting their environmental benefits.
A Simple Recipe: Diols, Air, and a Common-Metal Catalyst
The team developed a solid catalyst composed of copper and calcium oxides arranged as a mixed material. This catalyst is able to convert a wide range of liquid diols containing four to eight carbon atoms directly into lactones without any added solvent, at temperatures below 200 °C and under normal air pressure. In the process, the diol loses hydrogen and folds into a ring while oxygen from air is incorporated, forming water as the only by‑product. The new catalyst works with linear, cyclic, and even aromatic diols, and can reach essentially complete yields when reaction conditions are tuned, simplifying the separation steps that usually add cost and energy use in chemical plants.
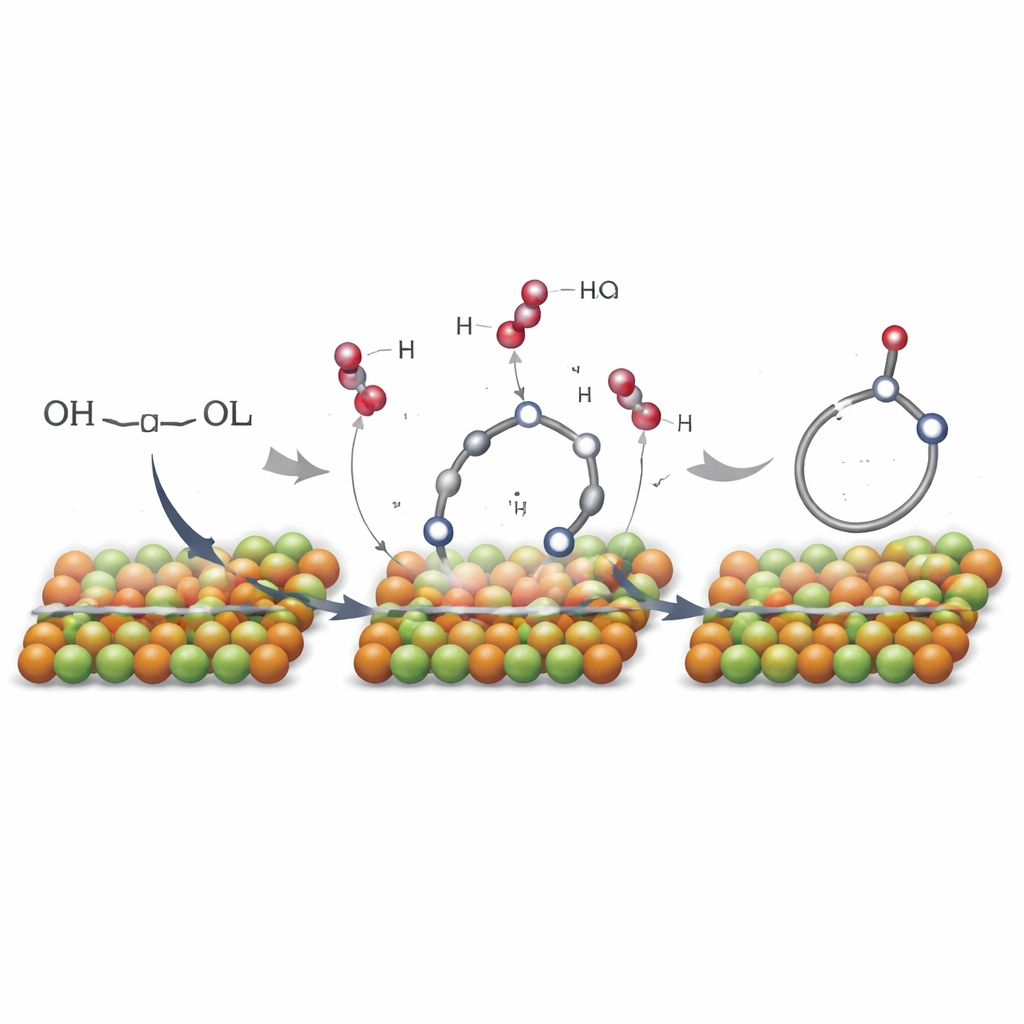
How the Catalyst Does the Job
To understand why this copper–calcium material is so effective, the researchers used several light‑based techniques that probe how atoms are arranged and how they change during the reaction. They discovered special junctions where copper and calcium share oxygen atoms. At these interfaces, copper atoms can readily shuttle between different charge states while the diol gives up hydrogen and closes into a ring. As the diol reacts, these sites briefly lose oxygen, then are re‑filled when oxygen from air lands and splits, allowing the cycle to continue. Conventional copper or calcium oxides on their own do not show this behavior under the same mild conditions, underscoring the importance of the mixed structure.
Energy, Cost, and Climate Benefits
Beyond the lab, the authors built a computer model of a plant that would convert bio‑based 1,4‑butanediol into the lactone gamma‑butyrolactone using their process. The model assumes bubble‑column reactors operated in batches, with continuous sparging of air to deliver oxygen and remove the water formed. Economic analysis suggests a minimum selling price of about $2.89 per kilogram of product, lower than the recent market average for the fossil‑based version. Life‑cycle assessment indicates that, compared with the standard petrochemical route, this bio‑based process could cut energy use by about 40 percent and greenhouse gas emissions by about 15 percent per kilogram of lactone produced, with even larger gains possible if the upstream plant sugars and energy inputs are made more sustainable.
What This Means for Future Plastics
In plain terms, this work offers a practical way to turn plant‑derived ingredients into the ring‑shaped molecules needed for next‑generation recyclable plastics, using only air, moderate heat, and a catalyst made from abundant metals instead of rare ones. The chemistry runs without added solvent, generates water as the main by‑product, and appears both cost‑competitive and less carbon‑intensive than current fossil‑based methods. If coupled with improved bio‑based feedstocks and renewable energy, this approach could substantially shrink the climate and resource footprint of polyester production and help move plastics closer to a truly circular life cycle.
Citation: Kiani, D., Rosetto, G., Ibrahim, F. et al. Solventless, ambient-pressure production of bio-based lactones over earth-abundant, mixed metal oxide catalysts for circular polyesters. Nat Commun 17, 2804 (2026). https://doi.org/10.1038/s41467-026-69362-8
Keywords: circular plastics, bio-based monomers, lactone synthesis, heterogeneous catalysis, techno-economic analysis