Clear Sky Science · en
Electrocatalytic C(sp3)-H bond functionalization using biomass-derived electrodes
Turning Shell Waste into Useful Tools
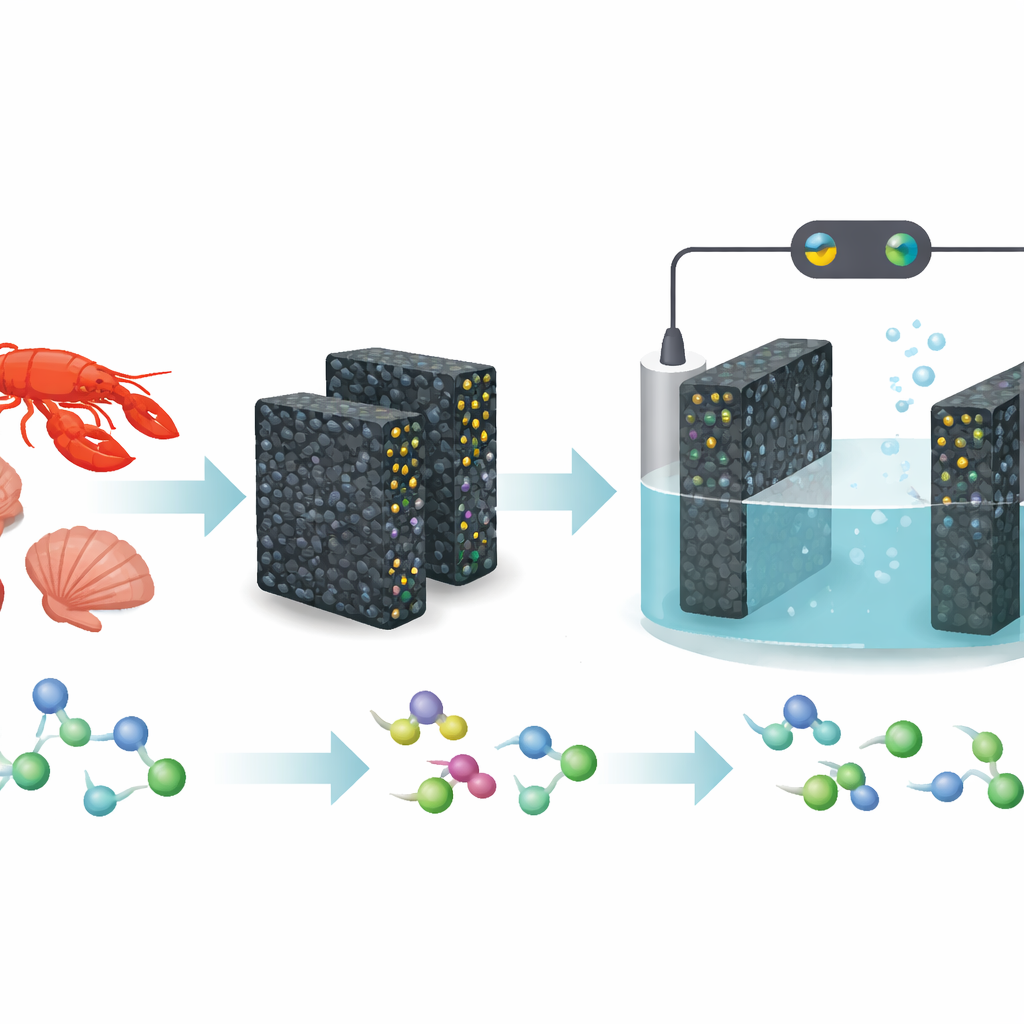
Every year, millions of tons of crab, shrimp, and lobster shells are thrown away, adding to waste and carbon emissions. This study shows how that discarded material can be transformed into high-tech electrical “sponges” that help chemists build medicines and other valuable molecules more cleanly and efficiently. By converting shell-based chitin into porous carbon packed with tiny metal particles, the researchers created new electrodes that make difficult chemical reactions easier, faster, and less polluting.
Why Everyday Molecules Are Hard to Tune
Many modern drugs and materials are built from carbon chains that look simple but are surprisingly tough to modify. The strong carbon–hydrogen bonds in these chains often resist change, so chemists usually have to go through several extra steps, using harsh reagents or high temperatures, to attach new groups such as chlorine, bromine, or oxygen-containing fragments. In recent years, chemists have turned to electricity as a cleaner way to power reactions, replacing toxic oxidants with electrons from a plug. Yet much of this progress has relied on standard metal plates or carbon sticks as electrodes, which are sturdy but not very active or selective for demanding reactions.
Building New Electrodes from Biomass
The team tackled this limitation by designing electrodes from the ground up instead of simply coating existing ones. They dissolved chitin—the structural material in shellfish exoskeletons—into a solution, turned it into a gel, freeze-dried it into a light aerogel, and then heated it to form a conductive carbon framework. Because chitin naturally contains nitrogen and oxygen atoms, it offers many anchoring points for metal species. By first loading metal ions onto tiny chitin spheres and then embedding these spheres into the gel, the researchers obtained carbon aerogels dotted with well-dispersed metal nanoparticles such as platinum, palladium, nickel, copper, and ruthenium oxide. The result is a family of “free-standing” electrodes with a high surface area, interconnected pores for fluid flow, and metal particles trapped inside microscopic channels where they cannot easily clump.
How the New Electrodes Power Tough Reactions
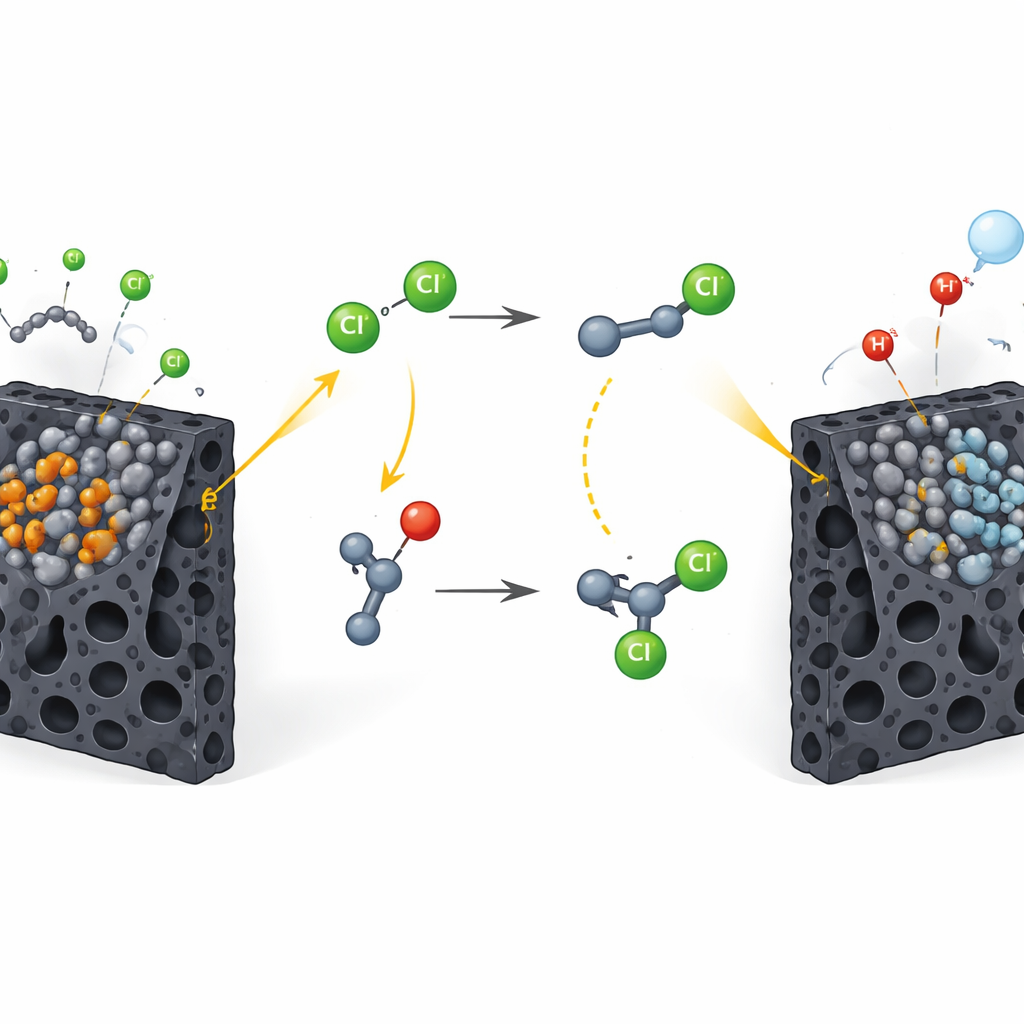
These biomass-derived electrodes showed strong performance in basic test reactions like hydrogen production and oxygen evolution, indicating excellent redox activity. The standout material was a ruthenium oxide version that excelled at oxidizing chloride ions, a common and inexpensive component of table salt and hydrochloric acid. Under an applied voltage, this electrode efficiently converted chloride into reactive chlorine radicals and, unusually, helped hold these short-lived species near its surface. Paired with a palladium-based electrode that excelled at turning protons into hydrogen gas, the system was able to transform simple alkanes into chlorinated products with high electrical efficiency while releasing hydrogen as a benign by-product. Similar strategies enabled bromination, nitration, and the formation of ether linkages between tetrahydrofuran and a wide range of alcohols, including complex natural products and sugars.
Labeling Drug Molecules with Heavy Hydrogen
The authors also showed that their electrode system can gently swap selected hydrogen atoms in drug-like molecules for deuterium, a heavier form of hydrogen. Such “heavy” versions of drugs are increasingly important in medicine, because deuterium can slow down how quickly a drug is broken down in the body. Using deuterium-containing water or alcohol as the source, the chloride-mediated process introduced deuterium into many common painkillers and other pharmacologically active structures, often with high levels of replacement. This exchange happened directly on the finished molecules, avoiding the need to rebuild them from deuterated starting materials, which are expensive and time-consuming to make.
What This Means for Greener Chemistry
Overall, the work demonstrates that carefully engineered electrodes made from waste biomass can dramatically improve the reach of electrosynthesis. By marrying porous, chitin-derived carbon with tiny metal particles, the researchers created robust, reusable tools that stabilize reactive intermediates and steer electrons into useful chemical changes instead of wasteful side reactions. Their system enables cleaner routes to halogenated compounds, ethers, and deuterium-labeled drugs, all while valorizing shell waste and using electricity as a controllable, potentially renewable power source. For non-specialists, the message is clear: smart materials design at the level of the electrode itself can help make the manufacture of everyday medicines and chemicals more sustainable.
Citation: Lu, L., Li, Y., Li, H. et al. Electrocatalytic C(sp3)-H bond functionalization using biomass-derived electrodes. Nat Commun 17, 2919 (2026). https://doi.org/10.1038/s41467-026-69274-7
Keywords: electrosynthesis, biomass-derived electrodes, chitin carbon aerogels, C–H functionalization, green chemistry