Clear Sky Science · en
Entropy-mediated solidification stabilizes and enhances energetic release in amorphous energetic materials
Why making safer powerful materials matters
Explosives are designed to release energy in a split second, yet they must remain quiet and stable during storage, transport, and handling. The tension between power and safety has long forced engineers to compromise. This study explores a new way to build high-performance explosives that are both safer and more efficient by reshaping them into a glass-like, amorphous form instead of the usual crystal, offering a fresh route to next-generation energetic materials.
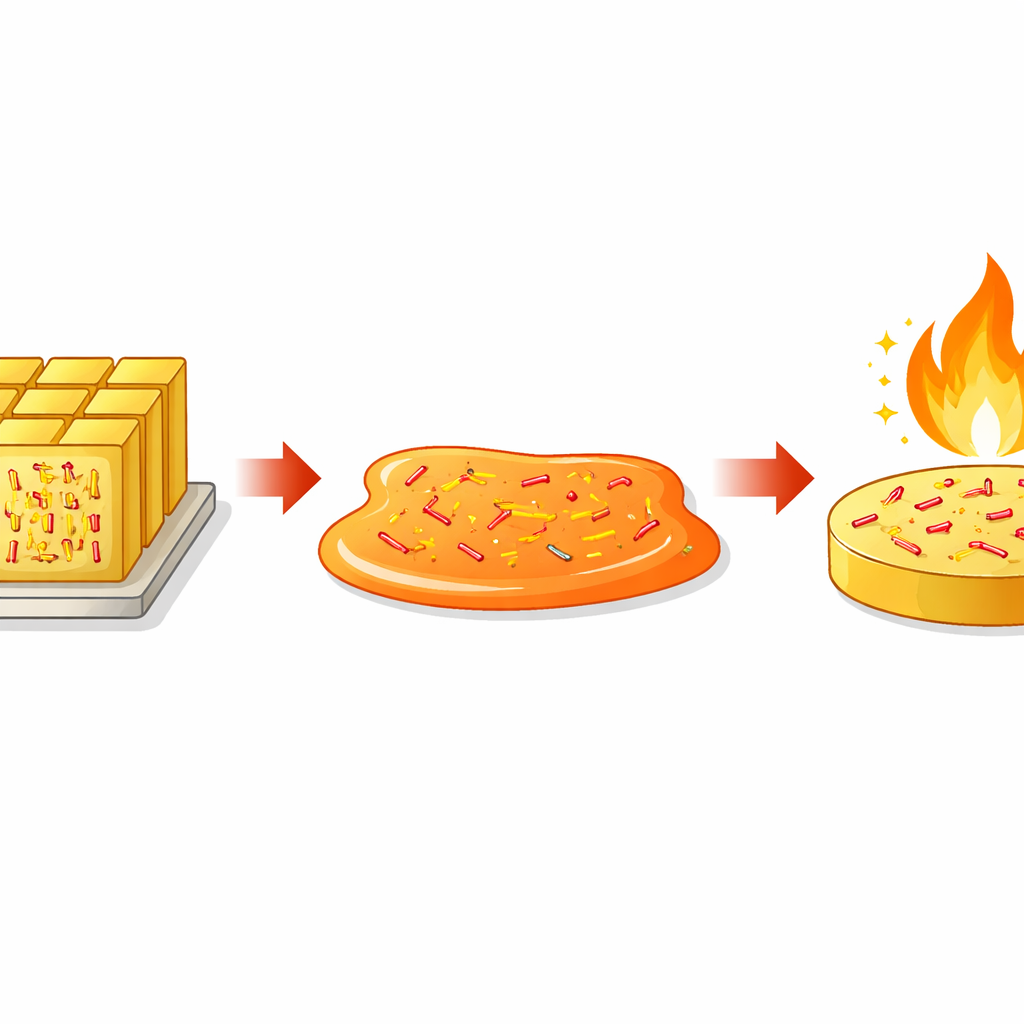
From neat crystals to disordered glass
Most traditional explosives are rigid organic molecules that crystallize easily, arranging themselves into neat, repeating lattices. That order is convenient for packing energy densely, but it also introduces weak spots, such as grain boundaries and defects, where dangerous local hot spots can form under impact or friction. The authors ask whether these same molecules could instead be frozen into a disordered, glassy solid, similar to window glass. In such an amorphous state, the molecules no longer line up in long-range patterns, potentially smoothing out weak points while preserving energy content.
Designing a molecule that refuses to crystallize
Creating a stable amorphous form from small, rigid molecules is surprisingly hard: they usually snap back into crystals as they cool. The team analyzed a series of energetic compounds and found that molecules with more three-dimensional shapes and less planarity were better at avoiding crystallization. They also discovered that having both hydrogen bond donors and acceptors helped lock molecules into disordered arrangements. Guided by these principles, they focused on an explosive called DATNBI, whose bent double-ring framework and nitro and amino groups naturally frustrate neat packing and encourage a three-dimensional hydrogen-bond network.
Freezing in disorder and keeping it stable
To make amorphous DATNBI, the researchers melted the crystalline material and then rapidly quenched it, trapping the molecules in a high-entropy, glassy state. They confirmed the loss of crystalline order using X-ray diffraction, which showed broad halos instead of sharp peaks, and found a relatively high glass transition temperature around 60 °C. Below this point, the amorphous solid remained structurally stable for at least a day, even when held just above room temperature. Microscopy revealed a smooth, dense microstructure with fewer pores and defects than the crystal, and surface measurements showed a more uniform, low-roughness surface that adheres better to other materials.
A self-repairing, cleaner-burning energetic glass
One striking feature of the amorphous explosive is its ability to heal small cracks when gently warmed. At around 60 °C, surface fractures closed within seconds, as molecular mobility and the hydrogen-bond network allowed the material to flow just enough to repair itself without melting. Small‑angle X-ray scattering showed that the number of tiny voids in the material dropped significantly with heating, helping to suppress hot-spot formation during mechanical insult. When heated to decomposition, the amorphous form broke down more completely than the crystal, leaving far less solid carbon residue and producing more fully oxidized gases. Kinetic analysis indicated a lower energy barrier for decomposition, and combustion tests showed faster burning and higher peak pressures, meaning the stored energy is released more rapidly and efficiently.
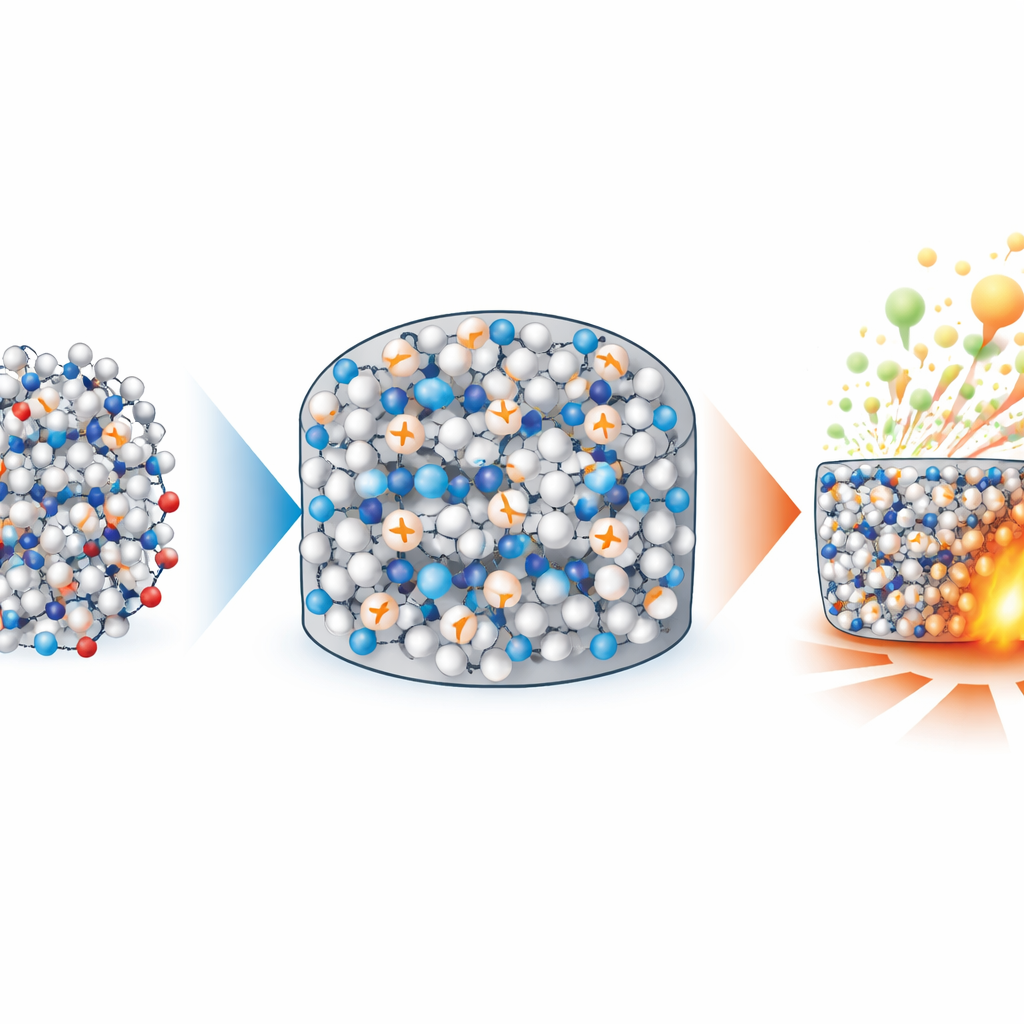
Balancing power and safety in future devices
By deliberately locking an explosive into a metastable, disordered solid, the researchers achieved an unusual combination: reduced impact and friction sensitivity together with faster, more complete energy release. This entropy-mediated solidification strategy avoids diluting the explosive with inert binders, preserving high energy density while improving processability for techniques such as hot pressing or 3D printing. Beyond this specific compound, the design rules they establish—using non-planar backbones and strong, three-dimensional hydrogen-bond networks—offer a blueprint for crafting a new generation of amorphous energetic materials and possibly other functional molecular glasses that marry robustness with high performance.
Citation: Zhou, X., Wang, Z., Huang, H. et al. Entropy-mediated solidification stabilizes and enhances energetic release in amorphous energetic materials. Nat Commun 17, 3271 (2026). https://doi.org/10.1038/s41467-026-69256-9
Keywords: amorphous energetic materials, glass-like explosives, hydrogen-bond networks, energy release efficiency, materials safety