Clear Sky Science · en
Bifidobacterium longum and prebiotic interventions restore early-life high-fat/high-sugar diet-induced alterations in feeding behavior in adult mice
Why early snacking habits may echo into adulthood
What we eat in our earliest days may quietly tune our brains and guts for years to come. This study in mice asks a question with clear human resonance: if babies are exposed to rich, high‑fat, high‑sugar foods, does it change how they eat as adults—and can friendly gut bacteria and special dietary fibers help undo the damage? By following mice from birth into adulthood, the researchers trace how an early “junk‑food” style diet reshapes gut microbes, brain cells that control appetite, and long‑term food preferences, in ways that differ between females and males.
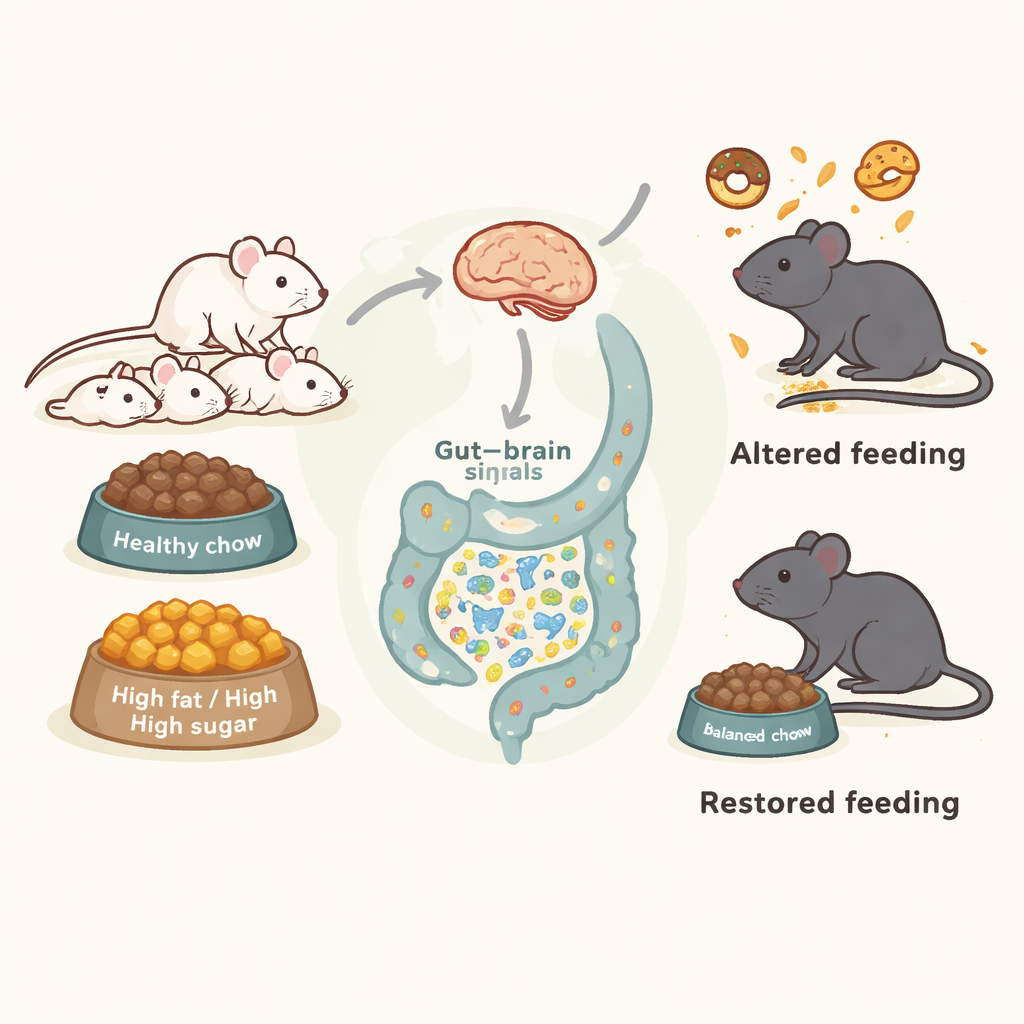
Early junk food leaves a hidden imprint
Newborn mice and their mothers were fed either a standard healthy chow or a high‑fat, high‑sugar (HFHS) diet that mimics a Western fast‑food pattern. The rich diet was given only during early life—from birth through weaning and a short period afterward—then all animals were switched back to normal chow. Despite this late “course correction” and ultimately similar adult body weights, the early HFHS exposure left lasting marks. As adults, previously exposed mice of both sexes showed a stronger preference for the tasty HFHS food when given a choice, and they engaged in more “food crumbling”—a manipulative behavior where they removed and ground food without fully eating it. These shifts suggest that early diet can prime both how appealing rich foods feel and how animals interact with them.
Gut microbes as middlemen between diet and brain
The team closely tracked the gut microbiota—the trillions of microbes that live in the intestine—and found that early HFHS diet reduced bacteria from the Bifidobacterium group, which are normally abundant in early life and linked to metabolic health. The microbiome changes were accompanied by altered levels of many blood chemicals, including amino acids, bile‑related molecules, and tryptophan‑derived compounds that can influence brain function and mood. Crucially, these effects were not the same in females and males. Females showed more disruption in pathways related to arginine and tryptophan metabolism, while males showed changes in bile and steroid‑related molecules and in how their bodies sensed components of bacterial cell walls. These sex‑specific patterns imply that early diet may raise disease risk through different biochemical routes in women and men.
Brain circuits for appetite are rewired
Because appetite is coordinated in the hypothalamus, a deep brain region that integrates signals from the body, the researchers examined this structure in detail. They found sweeping, long‑lasting changes in gene activity there, especially in females, where thousands more genes were altered than in males. Within a key hypothalamic hub called the arcuate nucleus, they saw fewer cells producing POMC, a molecule that normally helps curb eating, and fewer cells carrying receptors for the hormones leptin and ghrelin, which signal fullness and hunger. Another set of inhibitory neurons marked by the molecule PNOC, and cells bearing the bacterial‑sensing receptor NOD2, were also reduced. Together, these alterations suggest that early HFHS diet dulls the brain’s ability to read signals about energy balance and gut microbes, predisposing animals to overeating or disordered feeding even after weight normalizes.
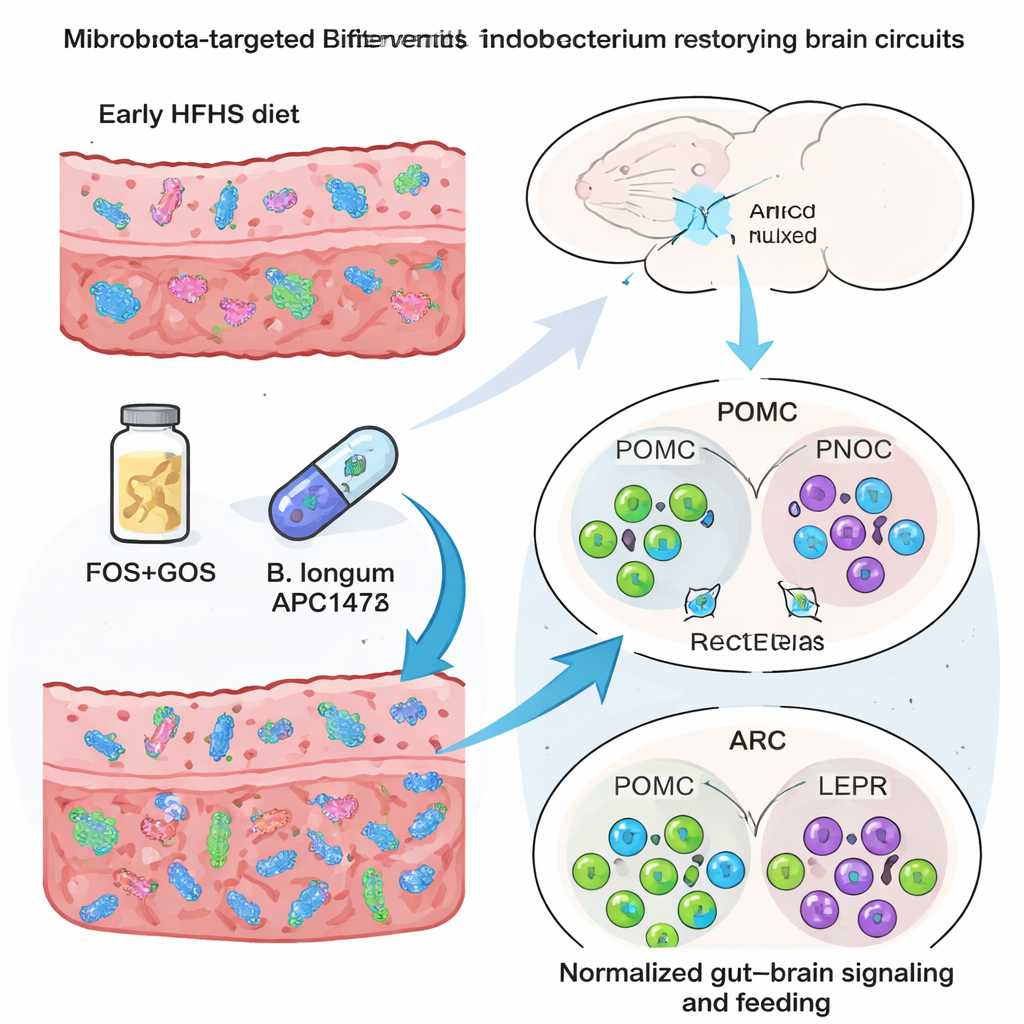
Friendly fibers and bacteria offer a partial reset
The study then tested two microbiota‑targeted “rescues” given in the drinking water from birth onward: a prebiotic fiber mix (fructo‑ and galacto‑oligosaccharides, FOS+GOS) designed to feed beneficial microbes, and a specific Bifidobacterium longum strain (APC1472). Both strategies boosted either the overall Bifidobacterium population (FOS+GOS) or this particular strain (APC1472), and both blunted many of the behavioral changes caused by early HFHS diet. Food crumbling and excessive intake of palatable food were reduced, and in males, an increased liking for a calorie‑free sweetener was normalized. In the brain, both interventions restored numbers of POMC and PNOC neurons and many NOD2‑positive cells in the arcuate nucleus, with especially strong recovery in females. Yet the ways they worked differed: FOS+GOS produced broad shifts in microbiome composition and gut–brain‑related pathways, whereas B. longum APC1472 induced more targeted metabolic and brain changes with relatively modest reshaping of the overall microbial community.
What this means for future diets and therapies
For lay readers, the message is stark but hopeful. An unhealthy, high‑fat, high‑sugar diet in early life can leave deep “programming” marks on gut bacteria, blood chemistry, and brain circuits that steer appetite, and these marks persist even after outward signs like body weight have returned to normal. Females appear more vulnerable at the level of brain gene networks, while males show distinct changes in how they sense bacterial products and hormones. At the same time, carefully chosen prebiotic fibers and probiotic strains can substantially repair these hidden alterations in mice, calming disordered feeding and rebalancing gut–brain communication. Although more work is needed before translating these findings to people, the study strengthens the idea that supporting a healthy microbiome in pregnancy and early childhood—through diet and potentially targeted supplements—could help protect lifelong eating behavior and reduce the risk of obesity and related disorders.
Citation: Cuesta-Marti, C., Ponce-España, E., Uhlig, F. et al. Bifidobacterium longum and prebiotic interventions restore early-life high-fat/high-sugar diet-induced alterations in feeding behavior in adult mice. Nat Commun 17, 1653 (2026). https://doi.org/10.1038/s41467-026-68968-2
Keywords: gut microbiome, early-life nutrition, probiotics, feeding behavior, obesity risk