Clear Sky Science · en
Genetic modifiers of APOE-ε4-associated cognitive decline
Why some brains resist memory loss
Many people carry a version of a gene called APOE-ε4 that greatly increases the chances of developing Alzheimer’s disease, yet not everyone with this gene develops serious memory problems. This puzzle hints that other genes and body systems can either cushion or worsen APOE-ε4’s impact on the aging brain. This study asks a simple but important question: among older adults who do and do not carry APOE-ε4, which other genetic factors help determine who maintains thinking skills and who declines?
Looking at thinking skills in thousands of older adults
The researchers combined data from nine long-term studies of aging, bringing together cognitive test scores and genetic information from 32,778 participants, most of whom were in their seventies. About 38% carried at least one copy of APOE-ε4. Instead of focusing only on Alzheimer’s diagnoses, the team examined three key thinking abilities—memory, language, and executive function (skills like planning and mental flexibility)—both at a single point in late life and over time as people were followed for several years. By harmonizing very different test batteries onto common scales, they could compare scores across all cohorts and look for genetic patterns linked to better or worse performance.
Finding gene changes that shape risk differently in carriers and non-carriers
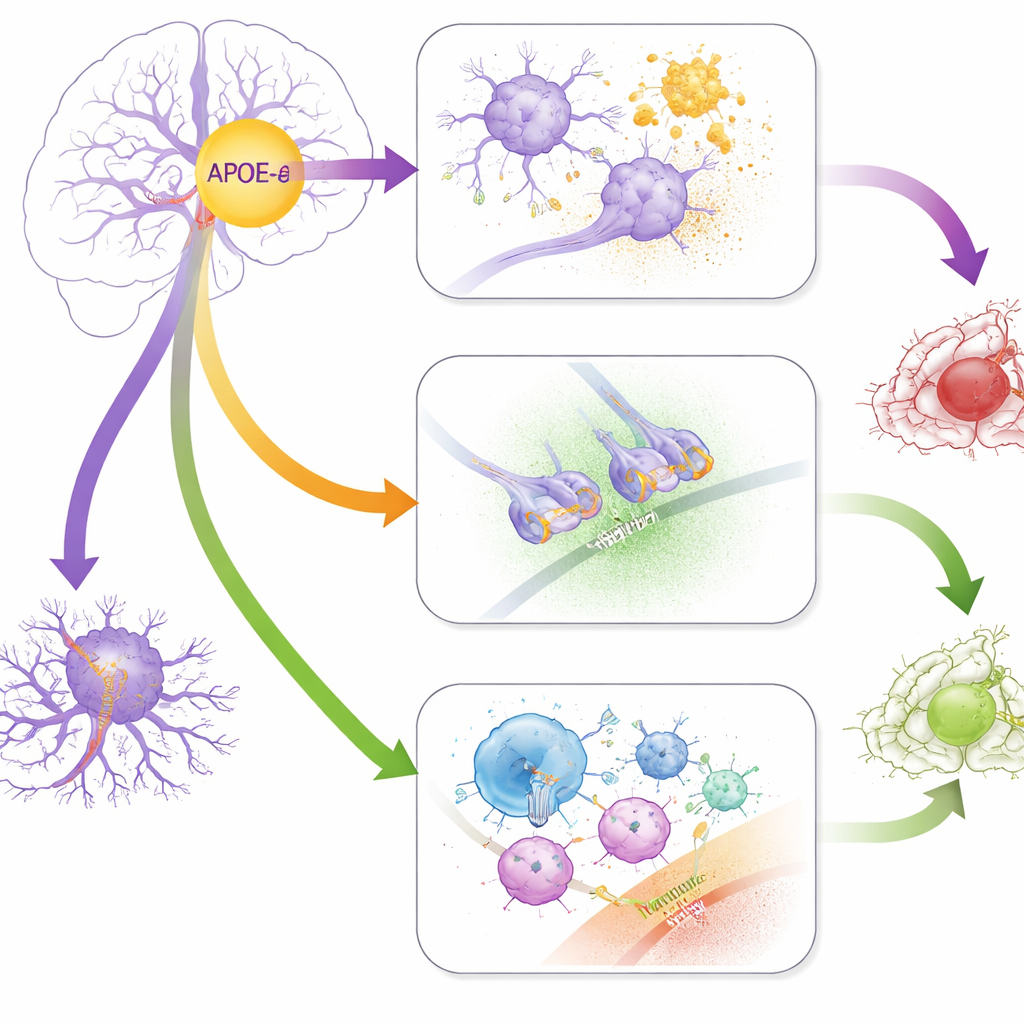
Using a genome-wide association approach, the team searched millions of genetic markers to see which ones tracked with cognitive performance, analyzing APOE-ε4 carriers and non-carriers separately and also testing for direct interactions with APOE-ε4 status. They found five regions of the genome that reached stringent statistical significance. In carriers of APOE-ε4, two regions were linked to better executive function at baseline and showed smaller but consistent benefits for language and memory. One sits near a gene called SEMA6D, which helps guide nerve fibers and appears to influence how brain immune cells clear amyloid waste. The other lies inside GRIN3A, which encodes a component of the NMDA receptor involved in communication between nerve cells and has been tied to protection from harmful overactivation of these receptors.
Gene pathways tied to brain immunity and inflammation
Among people who did not carry APOE-ε4, different genetic players emerged. A region affecting the ITGB8 gene was associated with poorer executive function: higher ITGB8 expression may shift microglial cells and a signaling molecule called TGF-β in ways that harm thinking skills. Another signal near SLCO1A2 was linked specifically to faster decline in executive abilities, hinting that how the brain transports certain molecules may matter for long-term cognitive health. A separate region near SLC25A21 interacted directly with APOE-ε4 to influence how quickly executive function declined, affecting carriers and non-carriers in opposite directions. When the team looked at broader biological pathways, they saw evidence that, in non-carriers, genes involved in how bacteria enter cells and how immune cells engulf debris may influence late-life cognition.
Shared roots between thinking problems and immune diseases
The study also compared the genetic signatures of late-life thinking abilities with those of dozens of other traits, such as autoimmune diseases and brain structure. Strikingly, APOE-ε4 carriers showed stronger genetic links between poorer cognition and chronic inflammatory conditions, including multiple sclerosis, celiac disease, and a liver-related autoimmune disorder. They also showed opposite patterns of correlation with hippocampal volume—a brain structure vital for memory—compared with non-carriers. These results suggest that, for people with APOE-ε4, an inherited tendency toward overactive or misdirected immunity may feed into brain decline, whereas the same immune-related variants may have weaker or even different effects in others.
What this means for understanding and treating Alzheimer’s risk
Overall, the work shows that APOE-ε4 does not act alone. Other common gene variants—some affecting nerve-cell communication, others shaping how brain immune cells clear waste or respond to inflammation—can tilt the balance toward resilience or vulnerability in late life, and they often do so differently in APOE-ε4 carriers and non-carriers. For a layperson, the takeaway is that having APOE-ε4 raises risk but does not seal one’s fate: a web of additional genes and immune pathways helps determine who declines quickly, who declines slowly, and who remains relatively sharp. Mapping these modifiers opens the door to more personalized risk prediction and points to biological systems—such as microglial cleanup of amyloid, NMDA receptor regulation, and chronic inflammation—that may be promising targets for future prevention and treatment efforts.
Citation: Contreras, A.G., Walters, S., Eissman, J.M. et al. Genetic modifiers of APOE-ε4-associated cognitive decline. Nat Commun 17, 2982 (2026). https://doi.org/10.1038/s41467-026-68933-z
Keywords: APOE-ε4, Alzheimer’s disease, genetic risk, cognitive decline, brain inflammation