Clear Sky Science · en
Heterobifunctional proteomimetic polymers for targeted degradation of MYC and KRAS
New tools to take down cancer’s toughest proteins
Many of the most dangerous cancer-driving proteins, like MYC and KRAS, have long been labeled “undruggable” because medicines can’t easily latch onto them. This study unveils a new kind of soft, synthetic material—called HYDRAC polymers—that can grab these slippery proteins and send them to the cell’s garbage disposal system. For readers, this work offers a glimpse of how chemistry and nanotechnology might open treatment options for cancers that have resisted traditional drugs.
A flexible scaffold instead of a single pill
Most targeted cancer drugs are small molecules that fit into pockets on a protein’s surface, much like a key in a lock. But proteins such as MYC are floppy and lack good pockets, while KRAS often mutates in ways that defeat classic drugs. The authors instead designed long, chain-like polymers built from repeating units. Along these chains they attach two kinds of components: short protein-binding segments that recognize a target such as MYC or RAS, and “degron” segments that recruit the cell’s own disposal machinery. Because many copies of each component decorate the same chain, a single HYDRAC molecule can reach out to multiple proteins and enzymes at once, something hard to achieve with a single, rigid drug molecule.
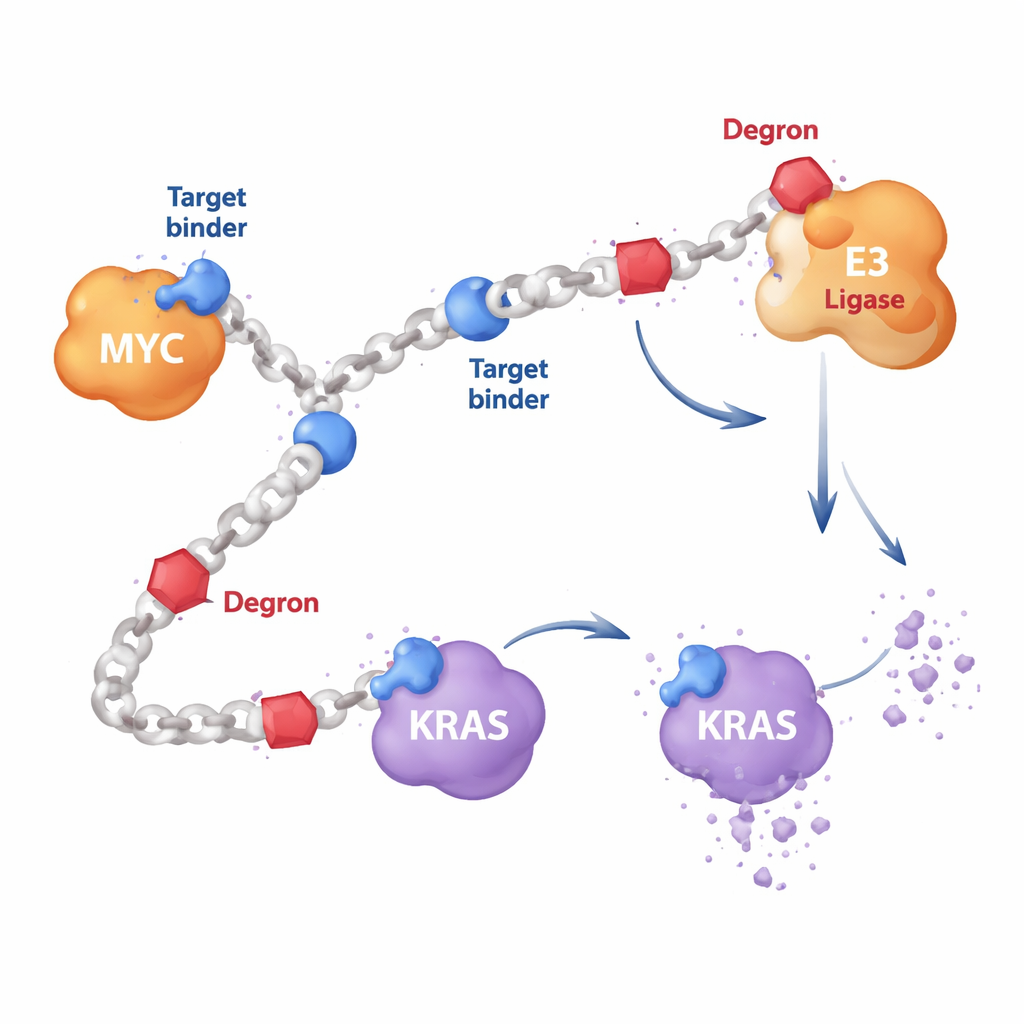
Teaching cells to chew up MYC
As a first test, the researchers focused on MYC, a master switch that drives growth in many cancers but has resisted direct attack for decades. They built HYDRAC polymers displaying a known MYC-binding peptide alongside a simple degron motif that flags proteins for destruction by the cell’s proteasome, its main protein shredder. In test-tube experiments, these polymers folded into compact, protein-like shapes and bound tightly to MYC but not to unrelated proteins. In cancer cells, the HYDRACs readily slipped inside, disrupted MYC-controlled gene programs, and triggered cell death—effects that depended on both the targeting and degron components being present on the same polymer chain.
From cell culture to shrinking tumors
Inside cells, HYDRAC treatment caused MYC protein levels to fall sharply without reducing MYC’s RNA, indicating true destruction rather than simple shutdown. Blocking the proteasome or key enzymes that activate certain degradation pathways rescued MYC levels, showing that the polymers work by redirecting the cell’s own waste-disposal systems. Proteome-wide analyses revealed that very few other proteins were affected, suggesting remarkable selectivity. In mouse models of MYC-driven tumors, fluorescently labeled HYDRACs accumulated preferentially in tumors after injection and remained there for days. Repeated dosing slowed tumor growth without major weight loss or obvious tissue damage, and tumor samples showed fewer dividing cells and more signs of programmed cell death.
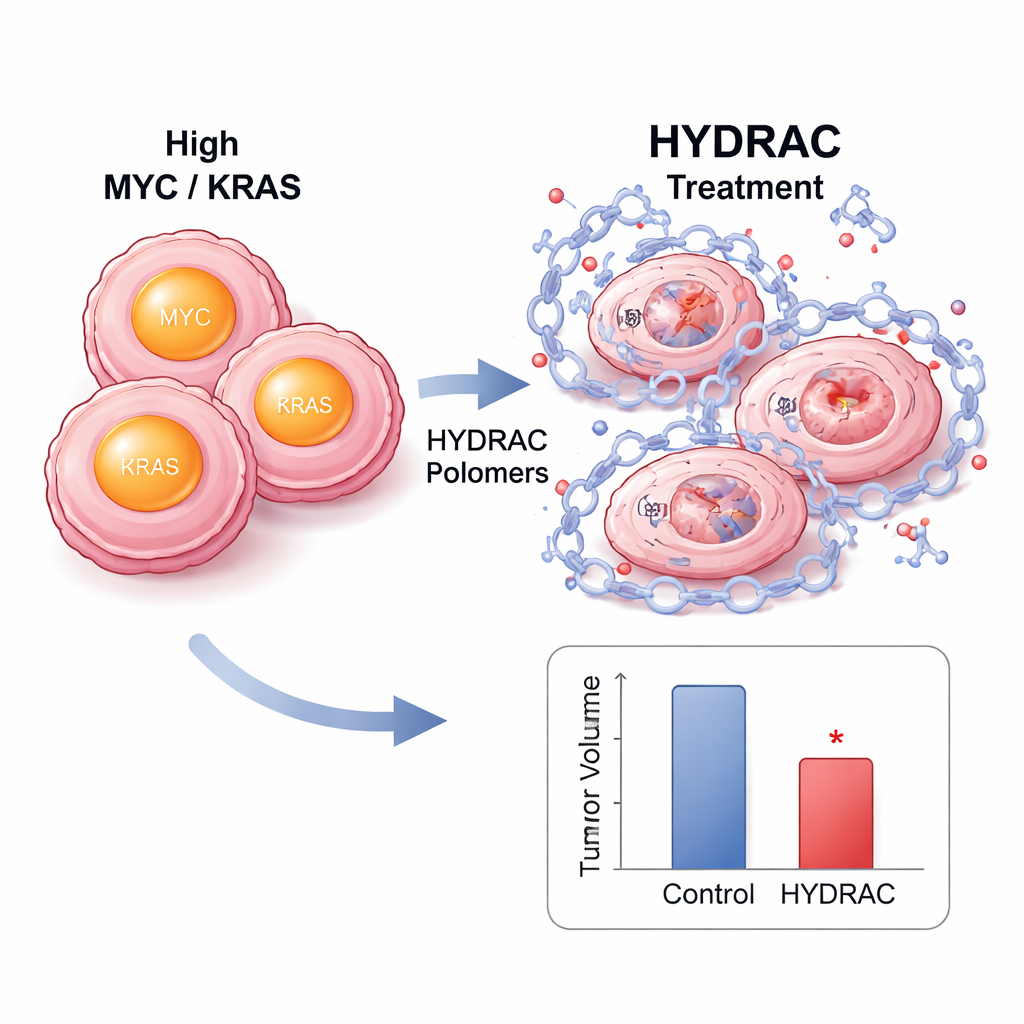
A plug-and-play platform for many disease targets
One strength of the HYDRAC approach is that its side groups can be swapped like interchangeable parts. The team replaced the original degron with other recruiting elements that call on different cellular enzymes, including those that recognize the drug thalidomide. Each version degraded MYC only when the intended enzyme was present and functional, confirming that the polymers can be tuned to use distinct cellular routes. To test generality, the researchers then built RAS-targeting HYDRACs using a peptide that recognizes RAS proteins. These constructs successfully degraded mutant KRAS in two different cancer cell lines, hinting at “pan-KRAS” potential that does not depend on a specific mutation—an important step for cancers where many KRAS variants coexist.
Why this matters for future cancer care
This work introduces HYDRACs as a new class of programmable materials that don’t just block troublesome proteins—they help cells erase them. By combining many targeting and degradation units on a flexible polymer, HYDRACs sidestep the design limits of traditional small-molecule drugs and PROTACs, which typically can only carry one of each. While much remains to be done before these materials reach the clinic, the results in MYC- and KRAS-driven models suggest that long-considered “undruggable” cancer proteins may, in fact, be vulnerable when medicine looks more like a tailored molecular brush than a single small pill.
Citation: Wang, M.M., Truica, M.I., Gattis, B.S. et al. Heterobifunctional proteomimetic polymers for targeted degradation of MYC and KRAS. Nat Commun 17, 1706 (2026). https://doi.org/10.1038/s41467-026-68913-3
Keywords: targeted protein degradation, MYC, KRAS, polymer therapeutics, cancer nanomedicine