Clear Sky Science · en
Ring-Opening Metathesis Polymerization (ROMP) of cyclic olefins: stereospecific ROMP and precision synthesis of bottlebrush polymers
Why tiny plastic combs matter
Plastics are no longer just shopping bags and soda bottles; chemists now build exquisitely shaped polymer molecules that can carry medicines, guide light, or form ultra-tough films. This article explores a powerful way to make such designer polymers, focusing on how the exact "handedness" and shape of their internal links can change how they melt, shine, and pack together. It culminates in the creation of “bottlebrush” polymers—molecules that look like microscopic bottle brushes—whose structure can be tuned with near-atomic precision.
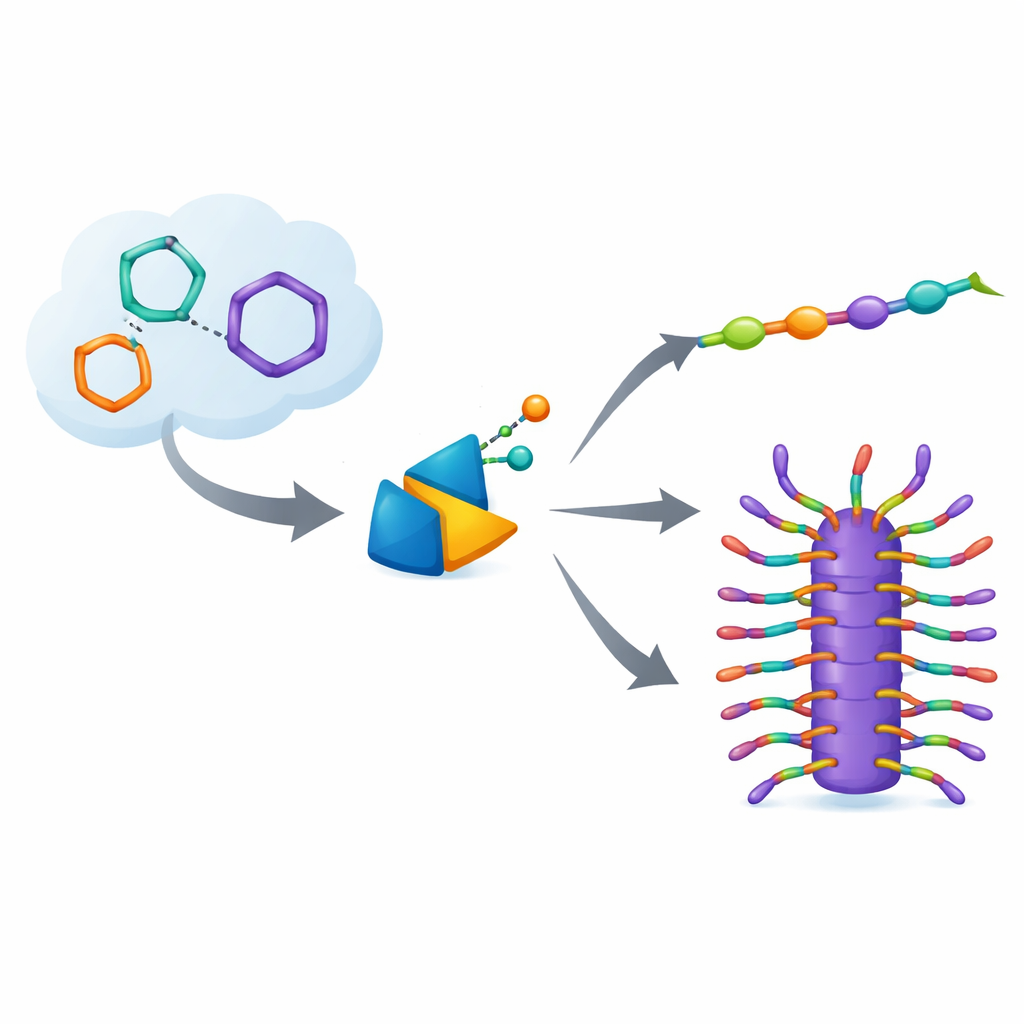
Unsnapping rings to build chains
The story begins with a reaction called ring-opening metathesis polymerization, or ROMP. Here, small ring-shaped molecules are persuaded to spring open and join end-to-end into long chains. Special metal-based catalysts, built from elements such as ruthenium, molybdenum, tungsten, vanadium, and niobium, grab a ring, cut one of its bonds, and stitch the opened piece onto a growing chain. Because many of these rings are strained, like a bent spring, opening them releases energy and drives the process forward. Under the right conditions, the reaction is “living”: chains grow in a controlled way, with little premature stopping, so chemists can pre-set how long the polymers will be and even assemble clean block structures.
Shaping chains by controlling left and right
When each ring opens, it leaves behind a carbon–carbon double bond that can adopt different shapes in space, commonly called cis and trans, and these can also be arranged in different sequences along the chain. The paper shows how carefully designing the catalyst’s surroundings—its bulky ligands and binding pockets—lets chemists favor one shape and sequence over another. For example, ruthenium complexes with specially arranged sulfur or carbene ligands can favor cis double bonds, while molybdenum and tungsten systems are tuned to give not only mostly cis links but also regular patterns (syndiotactic or isotactic) along the backbone. Vanadium and niobium catalysts go further, delivering very high cis contents even at elevated temperatures, something earlier systems struggled to achieve without falling apart.
From simple chains to molecular bottlebrushes
With ROMP under such fine control, the authors turn to more elaborate targets: bottlebrush polymers. These molecules have a main chain decorated densely with side chains, so they resemble microscopic cylindrical brushes. They can be made by first preparing side-chain-bearing building blocks (macromonomers) and then polymerizing those rings in a “grafting through” approach. Earlier versions relied mainly on ruthenium or molybdenum catalysts and already allowed precise control over molecular weight and block structure, yielding materials that self-assemble into ordered layers or reflect specific wavelengths of light. However, these older bottlebrushes usually contained mixed cis and trans bonds in their backbones, limiting how strongly the side chains could pack and how sharply their properties could be tuned.
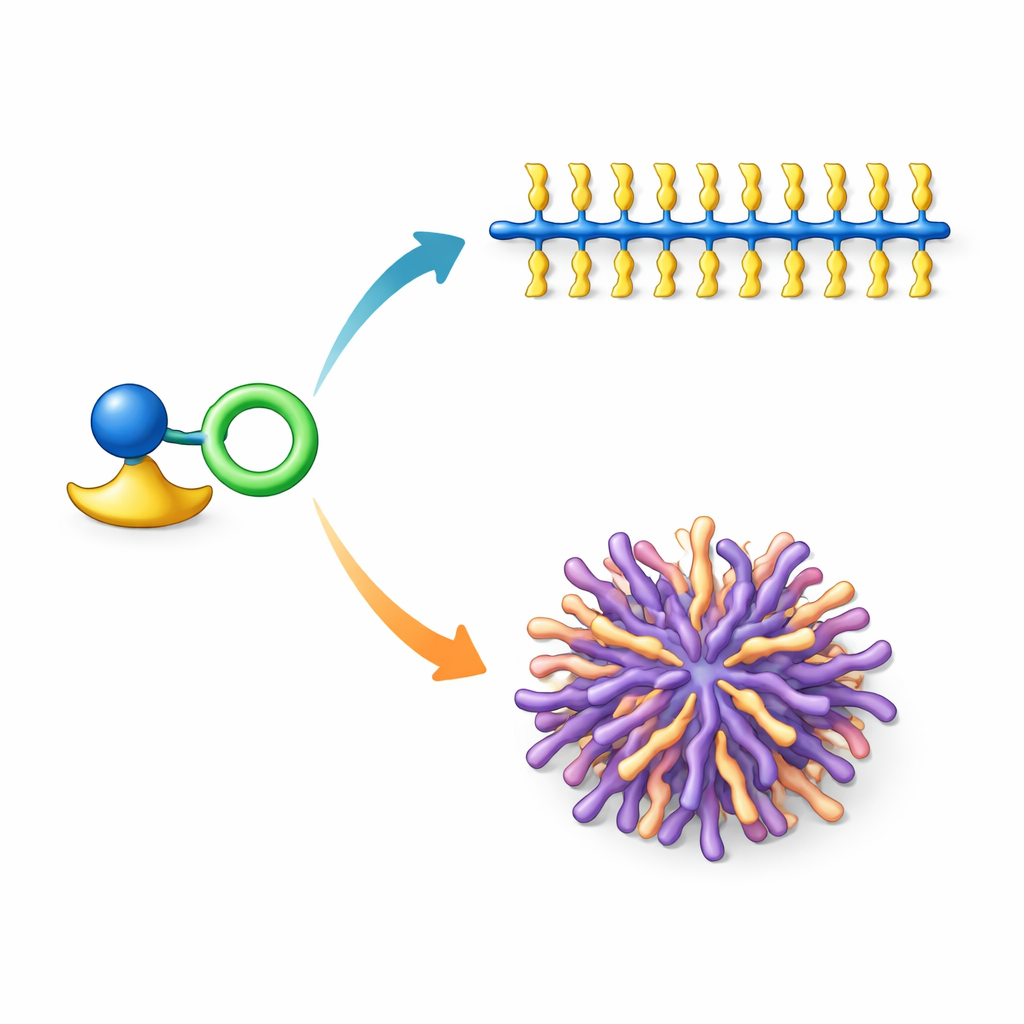
Switching backbone shape to tune material behavior
The review highlights recent breakthroughs using vanadium catalysts that can switch between making almost all-cis or mostly trans bottlebrushes simply by changing one part of the catalyst. When long, wax-like side chains are attached, the cis-rich bottlebrushes behave as semi-crystalline rods whose side chains crystallize together, while the trans-rich analogues form softer, more amorphous spherical aggregates. This same structural switch affects other functions too: when light-absorbing units such as terthiophene or pyrene are placed in the side chains, cis bottlebrushes and trans bottlebrushes show distinct melting temperatures and different light-emission patterns in films. These contrasts arise because the backbone geometry changes how closely the side chains on neighboring molecules can approach and how they interact.
Where this molecular precision can lead
To a non-specialist, these details may sound remote, but the conclusion is clear: by controlling not just which monomers are used, but exactly how each link is oriented in space, chemists can dial in the softness, melting point, and optical behavior of advanced plastics. ROMP, armed with modern catalysts, provides a toolkit for building bottlebrush polymers whose shapes and interactions are engineered from the ground up. Such control over molecular architecture could underpin future materials for flexible electronics, responsive coatings, smart drug carriers, and recyclable plastics, all designed with a level of precision that once seemed out of reach.
Citation: Nomura, K., Jaiyen, K. Ring-Opening Metathesis Polymerization (ROMP) of cyclic olefins: stereospecific ROMP and precision synthesis of bottlebrush polymers. Polym J 58, 485–509 (2026). https://doi.org/10.1038/s41428-025-01129-2
Keywords: ring-opening metathesis polymerization, stereocontrolled polymers, bottlebrush polymers, metal carbene catalysts, functional polymer materials