Clear Sky Science · en
Structural basis of human zinc-activated channel (ZAC) signaling and modulation
How a Trace Metal Helps Nerve Cells Talk
Zinc is best known as a nutrient in multivitamin tablets, but inside the body this metal also acts as a fast chemical messenger in the brain. The study described here uncovers, at atomic resolution, how a little-known protein called the zinc‑activated channel (ZAC) detects zinc outside cells and turns that signal into an electrical response. Understanding this special gate in the cell membrane could clarify how zinc shapes brain activity and suggest new ways to fine‑tune nerve signals in health and disease.
A Special Gate for Zinc Signals
Many rapid messages between nerve cells are carried by familiar chemicals such as serotonin or acetylcholine, which open ring‑shaped channels in the cell membrane. ZAC is a distant cousin of these receptors, but instead of responding to an organic molecule, it is switched on by metal ions such as zinc, copper and protons. ZAC is present in many human tissues, including the brain, yet is missing from standard lab animals like mice and rats, which has slowed progress. This work uses high‑resolution cryo‑electron microscopy to capture several three‑dimensional snapshots of human ZAC: in its resting form, with zinc bound, and with two different drugs that shut the channel down. Together, these structures reveal how zinc docks at the top of the channel, how ions pass through, and how blockers clamp the gate shut or wedge it open in a non‑conducting state.
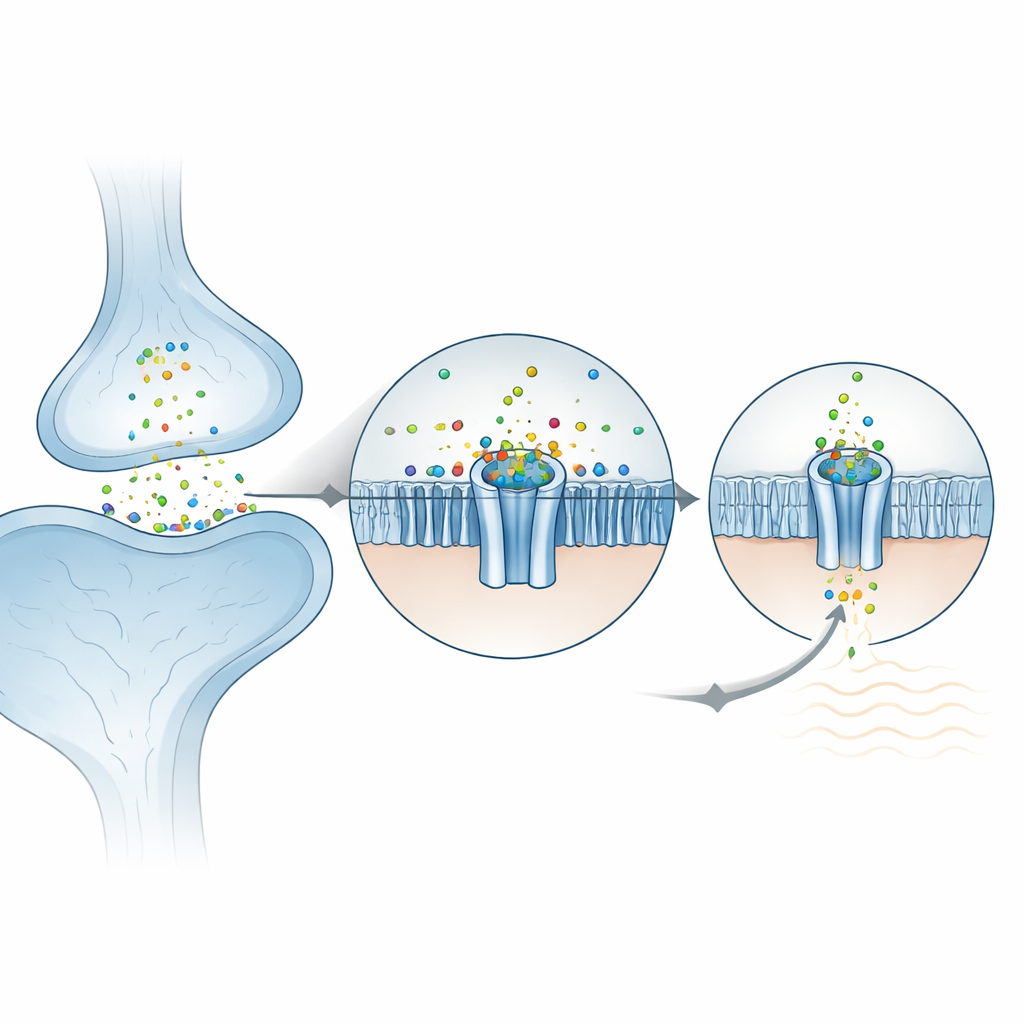
How Zinc Locks Into the Channel
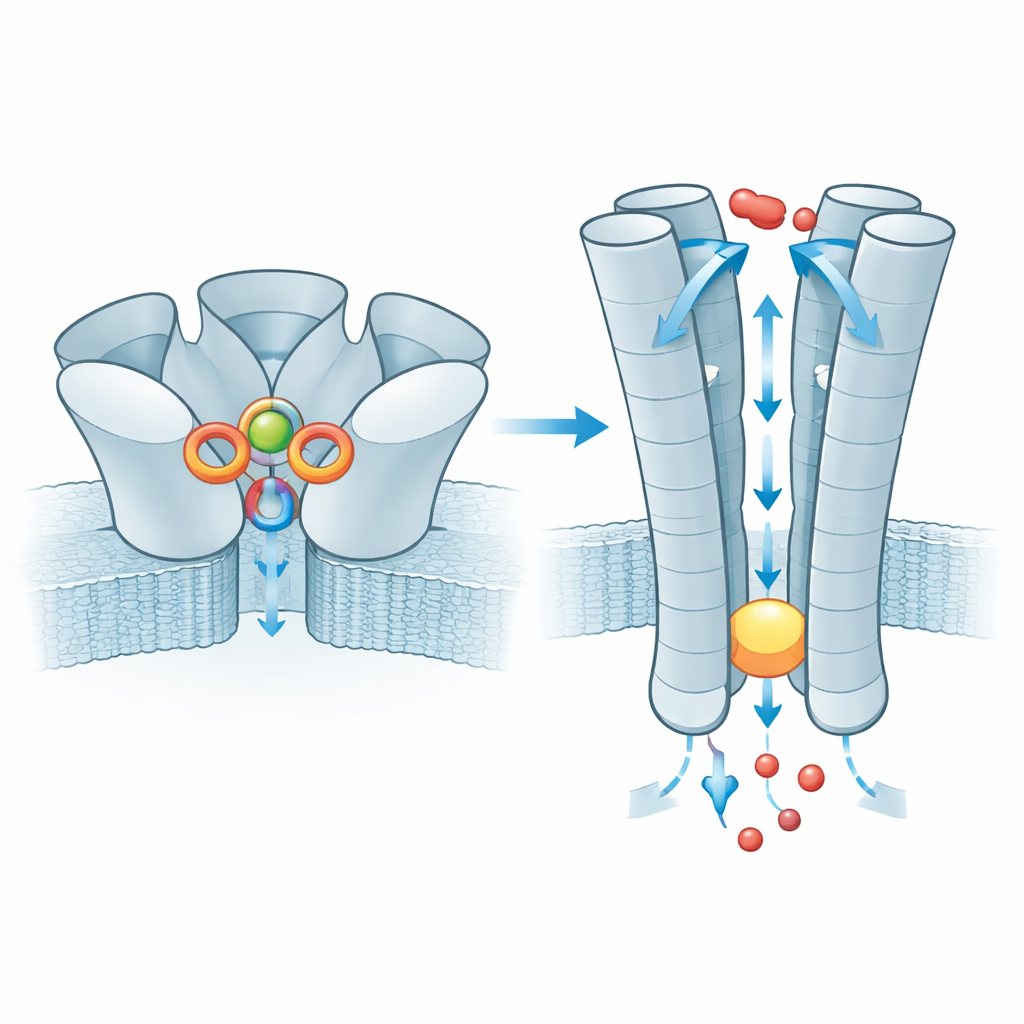
At the outer surface of the cell, ZAC is built from five identical subunits that form a rosette. The researchers found that zinc ions settle into five equivalent pockets, each lying between two neighboring subunits in the outer domain. Surprisingly, zinc is not held by the usual “grabber” amino acids that commonly bind metals (such as histidine or cysteine). Instead, it is cradled mainly by two ring‑shaped, aromatic side chains that stabilize the positively charged ion through so‑called cation–π interactions. When these key residues were mutated in frog egg cells used for electrical recordings, the channel became unresponsive to zinc, confirming their central role. The surrounding environment is already pre‑organized, which helps explain why ZAC shows notable spontaneous activity even without zinc: the protein sits close to the tipping point between closed and open and zinc simply nudges the balance.
The Path for Ions and a Common Genetic Variant
Once zinc has bound, the effect is transmitted down to the membrane‑spanning part of ZAC, where five inner helices form the pore. In the resting state this tube narrows at a ring of leucine residues, creating a hydrophobic plug that blocks ion movement. With zinc bound, the plug widens just enough to let small positive ions such as sodium and potassium slip through, while the charged lining of the pore helps select for these ions. The team also examined a very common human variant of ZAC in which a single amino acid (threonine) is replaced by alanine in a short strand above the plug. This small change weakens an interaction network between neighboring subunits that normally helps relay zinc binding to the gate, and electrical measurements show that channels built from this variant conduct far smaller currents. The variant therefore acts as a less efficient signal converter, although its impact on human physiology remains unknown.
How Two Drugs Silence the Channel
Beyond zinc, the study also explores how two antagonists, TTFB and d‑tubocurarine (d‑TC), shut ZAC down. TTFB is a tailor‑made small molecule that turns out to lodge deep in the membrane region, slipping between two of the pore‑forming helices just above the gate. There it interacts with a snug hydrophobic pocket and nearby polar residues, effectively bracing the helices so they cannot move into a fully open conducting state. d‑TC, an older drug once used as a muscle relaxant, acts more broadly. One d‑TC molecule binds in the same outer pocket that normally hosts zinc, mimicking zinc’s positive charge but blocking real zinc from binding properly. A second d‑TC molecule sits in the entrance of the pore itself, physically plugging the tunnel. Together, these bindings trap ZAC in a desensitized, non‑conducting conformation while the protein’s shape remains relatively open.
What the Findings Mean for Zinc and Health
This structural work shows in clear molecular detail that ZAC is a genuine zinc sensor, using an unusual aromatic cradle to detect the metal at the same type of site where other family members bind neurotransmitters. It also identifies specific interaction networks that tune how easily the channel opens and closes, and maps two distinct drug‑binding sites that can either wedge the gate shut or jam the pore. For non‑specialists, the key message is that zinc is not just a static nutrient but an active signal, and ZAC is one of its dedicated receivers. As scientists search for ZAC’s roles in the human body and in neurological disorders linked to zinc imbalance, these atomic‑level blueprints provide a roadmap for designing more precise molecules to modulate this channel and, potentially, to correct disrupted zinc signaling.
Citation: Zhou, Z., Long, Y., Chao, Y. et al. Structural basis of human zinc-activated channel (ZAC) signaling and modulation. Cell Discov 12, 23 (2026). https://doi.org/10.1038/s41421-026-00878-5
Keywords: zinc signaling, ligand-gated ion channels, neuroscience, cryo-EM structure, channel modulation